Local Anaesthetics
Local anesthetics are the drugs when given either topically or parenterally to a localized area, produce loss of sensation with or without loss of consciousness by reversibly blocking the generation and conduction of nerve impulses.
- They do not interact with the pain receptors or inhibit the release or the biosynthesis of pain mediators. The anesthesia produced by local anesthetics is without loss of consciousness or impairment of vital central cardiorespiratory functions.
- Local anesthetics block nerve conductance by binding to selective sites on the Na+ channels in the excitable membranes, thereby reducing Na+ passage (i.e., conductance) through the pores and, thus, interfering with the generation of action potentials.
- Although local anesthetics decrease the excitability of nerve membranes, they do not affect the neuron’s resting potential.
- Local anesthetics, in contrast to analgesic compounds, do not interact with the pain receptors or inhibit the release or the biosynthesis of pain mediators.
Read and Learn More Medicinal Chemistry II Notes
Characteristics of an Ideal Local Anesthetic
- The action of local anesthetic must be reversible.
- It must be non-irritating to the tissues and should not produce any secondary local reaction.
- It should have a low degree of systemic toxicity and have sufficient potency to provide
complete anesthesia. - It should have a rapid onset and be of sufficient duration to be advantageous.
- It should have sufficient penetrating properties to be effective as a topical anesthetic
- It should be relatively free from producing allergic reactionss.
- It should be stable in solution and undergo biotransformation readily within the body
- It should be either sterile or capable of being sterilized by heat without deterioration.
“What is local anesthesia? A detailed question and answers guide”
No local anesthetic fulfills all of these requirements, particularly regarding the duration of action.
Local Anesthetic Mechanism of Action
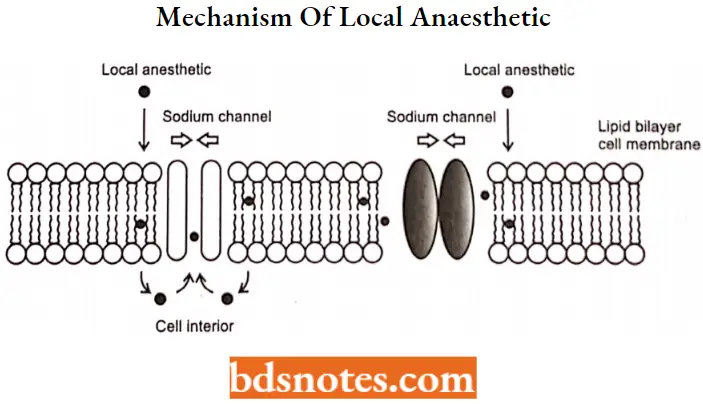
- The nerve fiber is a long cylinder surrounded by a semipermeable (allows only some substances to pass) membrane. This membrane is made up of proteins and lipids (fats). Some of the proteins act as channels, or pores, for the passage of sodium and potassium ions through the membrane.
- The movement of nerve impulses along a nerve fiber is associated with a change in the permeability of the membrane. The pores widen, and sodium ions (Na+) move to the inside of the fiber. At the same time, potassium ions (K+) diffuse out through other pores.
- The entire process is called depolarization. Immediately after the nerve impulse has passed, the pores again become smaller.
- Sodium ions (Na+) are now “pumped” out of the fiber. At the same time, potassium ions are actively transported into the fiber. The nerve membrane is then ready to conduct another impulse.
- Local anesthetics block sodium channels. When the local anesthetic binds, it blocks sodium ion passage into the cell and thus blocks the formation and propagation of the action potential.
- This blocks the transmittance of the message of “pain” or even “touch” from getting to the brain.
- The ability of a local anesthetic to block action potentials depends on the ability of the drug to penetrate the tissue surrounding the targeted nerve as well as the ability of the drug to access the binding site on the sodium channel.
“Understanding types of local anesthesia through FAQs: Q&A explained”
Structure-Activity Relationships
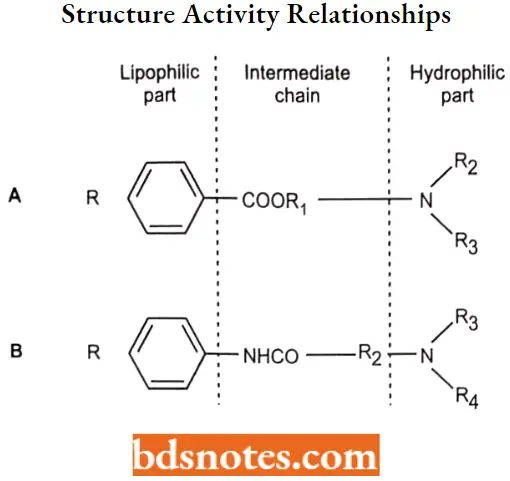
All local anesthetics contain 3 structural components:
- Lipophilic aromatic group (usually substituted)
- A connecting group that is either an ester or an amide
- An ionizable amino group (hydrophilic group)
“How does local anesthesia work in medical procedures? FAQ answered”
1. Lipophilic aromatic group
The aromatic ring adds lipophilicity to the anesthetic and helps in penetration through biological membranes. It is also having direct contact with the local anesthetic binding site on the sodium channel.
- Substituents on the aromatic ring may increase the lipophilicity of the aromatic ring. Lipophilic substituents and electron-donating substituents in the para position increased anesthetic activity.
- The lipophilic substituents are thought to both increase the ability of the molecule to penetrate the nerve membrane and increase their affinity at the receptor site.
- The electron-donating groups on the aromatic ring created a resonance effect between the carbonyl group and the ring, resulting in the shift of electrons from the ring to the carbonyl oxygen.
- As the electronic cloud around the oxygen increases, it increases the affinity of the molecule with the receptor. When the aromatic ring was substituted with an electron-withdrawing group, the electron cloud around the carbonyl oxygen decreased and the anesthetic activity decreased.
2. Connecting group
The linker is usually an ester or an amide group along with a hydrophobic chain of various lengths. When the number of carbon atoms in the linker is increased, the lipid solubility, protein binding, duration of action, and toxicity increase.
- Esters and amides are bioisosteres having similar sizes, shapes, and electronic structures. The similarity in their structures means that esters and amides have similar binding properties and usually differ only in their stability in vivo and in vitro.
- For molecules that only differ at the linker functional groups, amides are more stable than esters and thus have longer half-lives than esters.
3. An ionizable amino group
Most local anesthetics contain tertiary nitrogen with a pKa between 7.5 and 9.5. Therefore, at physiological pH, both the cationic and neutral form of the molecule exists.
- The molecule can penetrate the nerve membrane in its neutral form and then re-equilibrate with its cationic form on the internal side of the membrane which then blocks the sodium channels.
- To keep the anesthetic soluble in commercial solutions, most preparations are acidified. To decrease pain on injection and to increase the onset of action, sodium bicarbonate is added to the commercial preparation.
- By adding sodium bicarbonate, the solution will become less acidic and more of the drug will be found in the neutral form. The neutral form will thus cross the nerve membrane quicker and have a quicker onset of action.
“Importance of studying local anesthesia for healthcare professionals: Questions explained”
Vasoconstrictors Used in Combination with Local Anesthetics
Many anesthetic preparations are commercially available combined with the vasoconstrictor epinephrine. Some anesthetics are also combined with other agents such as norepinephrine, phenylephrine, oxymetolazone, or clonidine to achieve the desired formulation.
- The epinephrine in the anesthetic solution has multiple purposes. As a vasoconstrictor, the injected epinephrine will constrict capillaries at the injection site and thus limit blood flow to the area.
- The local anesthetic will thus stay in the immediate area of injection longer and not be carried away to the general circulation. This will help keep the drug where it is needed and allow the minimal drug to be absorbed systemically.
- This will reduce the systemic toxicity from the anesthetic and increase the duration of anesthetic activity at the site of injection.
The lack of blood flow in the immediate area will also decrease the presence of metabolizing enzymes and this also increases the duration of action of the anesthetic locally.
Classification Of Local Anaesthetics
- Esters
- Esters of benzoic acid.
- For Example. Cocaine, Hexylcaine, Meprylcaine, Cyclomethycaine, Piperocaine.
- Esters of Para Amino Benzoic acid.
- For Example. Benzocaine, Butamben, Procaine, Butacaine, Propoxycaine,Tetracaine, Benoxinate.
- Anilide Amides
- For Example. Xylocaine (Lidocaine), Mepivacaine, Prilocaine, Etidocaine
- Miscellaneous
- For Example. Phenacaine, Diperodon, Dibucaine.
- Esters
- Esters of benzoic acid
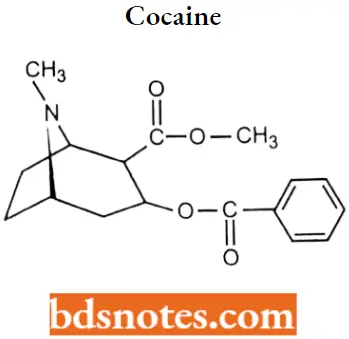
Cocaine: Cocaine is a tropane alkaloid ester isolated from the coca leaves with central nervous system(CNS) stimulation And local anesthetic activity. Cocaine was the first agent used for topical anesthesia.
“Common challenges in understanding local anesthesia effectively: FAQs provided”
Cocaine IUPAC name: methyl (1R,2R,3S,5S)-3-(benzoyloxy)-8-methyl-8-azabicyclo[3.2.1]octane-2-carboxylate.
Cocaine MOA: Cocaine is a tropane alkaloid with central nervous systems (CNS) stimulating and local anesthetic activity. Cocaine binds to and blocks the voltage-gated sodium channels in the neuronal cell membrane.
- By stabilizing neuronal membranes, cocaine inhibits the initiation and conduction of nerve impulses and produces a reversible loss of sensation.
- Cocaine binds to the dopamine, serotonin, and norepinephrine transport proteins and inhibits the reuptake of dopamine, serotonin, and norepinephrine into pre-synaptic neurons.
- This leads to an accumulation of the respective neurotransmitters in the synaptic cleft and may result in increased postsynaptic receptor activation. Its effect on dopamine levels is most responsible for the addictive properties of cocaine.
Cocaine Metabolism: Cocaine is metabolized in the liver. It is metabolized to benzoylecgonine and ecgonine methyl ester, which are both excreted in the urine. In the presence of alcohol, a further active metabolite, cocaethylene is formed and is more toxic than cocaine itself. Half-life hour.
Cocaine Uses: Cocaine is a local anesthetic indicated for the introduction of local (topical) anesthesia of accessible mucous membranes of the oral, laryngeal, and nasal cavities. It was originally used as a local anesthetic but is no longer used because of its potent addictive qualities.
Cocaine Adverse Effects: When given in high doses systemically, cocaine has mood-elevating effects that have led to its abuse potential. High doses of cocaine can be associated with toxic reactions including hyperthermia, rhabdomyolysis, shock, and acute liver injury which can be severe and even fatal.
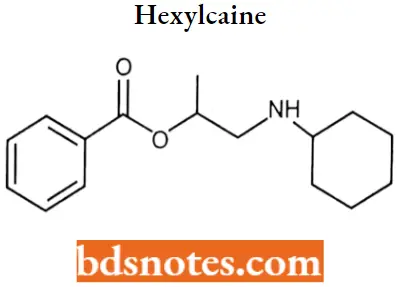
Hexylcaine: Hexylcaine also called cyclaine or osmocaine, is a short-acting local anesthetic. Hexylcaine is a benzoate ester.
“Why is early learning of local anesthesia critical for patient safety? Answered”
Hexylcaine IUFAC name: 1-(cyclohexylamino)propan-2-yl benzoate.
Hexylcaine MOA: Hexylcaine acts mainly by inhibiting sodium influx through voltage-gated sodium channels in the neuronal cell membrane of peripheral nerves.
- When the influx of sodium is interrupted, an action potential cannot arise and signal conduction is thus inhibited.
- The receptor site is thought to be located at the cytoplasmic (inner) portion of the sodium channel.
Hexylcaine Metabolism: Hydrolyzed by plasma esterase to benzoic acid and other derivatives. Half-life is <10 minutes.
Hexylcaine Uses: It is a short-acting local anesthetic
Hexylcaine Overdose: Overdose can lead to headache, tinnitus, numbness and tingling around the mouth and tongue, convulsions, inability to breathe, and decreased heart functions.
Meprylcaine: Meprylcaine (also known as Epirocaine and Oracaine) is a local anesthetic with stimulant properties that is structurally related to dimethocaine.
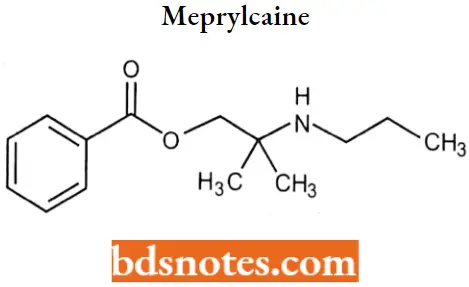
Meprylcaine IUPAC name: [2-methyl-2-(propylamino)propyl] benzoate.
Meprylcaine MOA: Meprylcaine has a relatively potent inhibitory action on the monoamine transporter and inhibits the reuptake of dopamine, norepinephrine, and serotonin
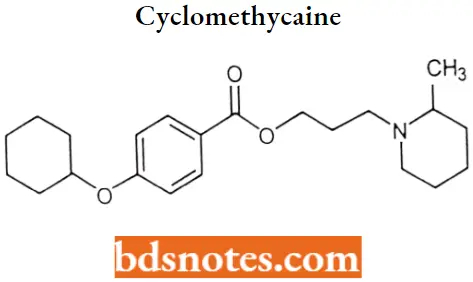
Cyclomethycaine
“Factors influencing success with local anesthesia knowledge: Q&A”
Cyclomethycaine IUPAC name: 3-(2-methylpiperidin-1-yl)propyl 4-cydohexyloxybenzoate
Cyclomethycaine MOA: It acts mainly by inhibiting sodium influx through voltage-gated sodium channels in the neuronal cell membrane of peripheral nerves. When the influx of sodium is interrupted, an action potential cannot arise and signal conduction is thus inhibited.
Piperocaine
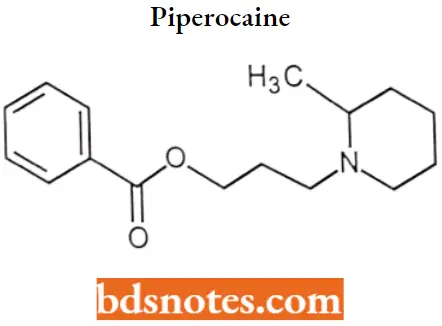
Piperocaine IUPAC name: 3-(2-methylpiperidin-1-yl) propylbenzoate.
Piperocaine MOA: It is primarily a sodium channel blocker. It acts by inhibiting sodium influx through voltage-gated sodium channels in the neuronal cell membrane of peripheral nerves due to which when the influx of sodium is interrupted, an action potential cannot arise and signal conduction is thus inhibited.
Esters of Para Amino Benzoic acid
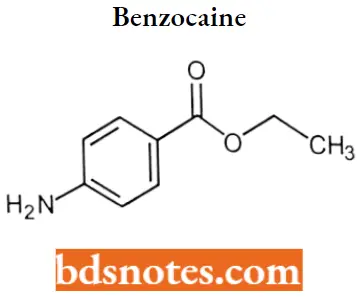
Benzocaine: Benzocaine is a unique local anesthetic becauseit does not contain a tertiary amine.
“Steps to explain types of local anesthetics: Esters vs amides: Q&A guide”
Benzocaine IUPAC name: Eethyl 4-aminobenzoate.
Benzocaine MOA: Benzocaine binds to sodium channels and reversibly stabilizes the neuronal membrane which decreases its permeability to sodium ions. Depolarization of the neuronal membrane is inhibited thereby blocking the initiation and conduction of nerve impulses.
Benzocaine Metabolism: Like most amino ester-type local anesthetics, it is easily hydrolyzed by plasma cholinesterase. The pKa of the aromatic amine is 3.5 ensuring that benzocaine is uncharged at physiological pH.
Because it is uncharged, it is not water-soluble but is ideal for topical applications. The onset of action is within 30 seconds and the duration of drug action is 10 to 15 minutes,
Benzocaine Uses: Benzocaine is used for endoscopy, bronchoscopy, and topical anesthesia. It is used topically by itself or in combination with menthol or phenol in non-prescription dosage forms such as gels, creams, ointments, lotions, aerosols, and lozenges to relieve pain or irritation caused by such conditions as sunburn, insect bites, toothache, teething, cold sores or canker sores in or around the mouth, and fever blisters.
Benzocaine Adverse effects: Toxicity can occur when the topical dose exceeds 200 to 300 mg resulting in methemoglobinemia.
Infants and children are more susceptible to this and methemoglobinemia has been reported after benzocaine lubrication of endotracheal tubes and after topical administration to treat a painful diaper rash.
Benzocaine Synthesis:
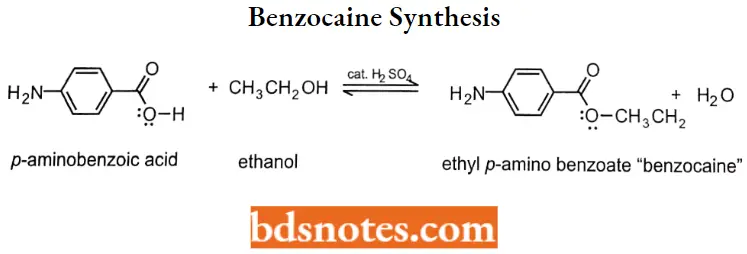
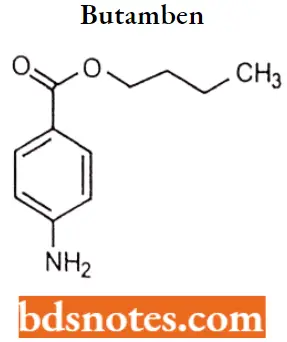
Butamben: Butamben is a local anesthetic in the form of n-butyl-p-aminobenzoate. Its structure corresponds to the standard molecule of a hydrophilic and hydrophobic domain separated by an intermediate ester found in most of the local anesthetics.
Due to its very low water solubility, butamben was considered of low usability as it is only suitable to be used as a topical anesthesia.
“Role of lidocaine in local anesthesia: Questions answered”
Butamben IUPAc name: Butyl 4-aminobenzoate.
Butamben MOA: Butamben acts by inhibiting the voltage-gated calcium channels in dorsal root ganglion neurons. It is reported as well that butamben is an inhibitor of the sodium channels and a delayed rectifier of potassium currents.
All the effects of butamben are performed in the root ganglion neurons which suggests that the related anesthetic effect may be caused by the reduced electrical excitability.
Butamben Metabolism: It is hydrolyzed via cholinesterase for the formation of inert metabolites.
Butamben Uses: Butamben was indicated for the treatment of chronic pain due to its long-duration effect. It is also indicated as a surface anesthetic for skin and mucous membranes and for the relief of pain and pruritus associated with anorectal disorders.
Butamben Toxicity: In studies, the most common effect was related to the generation of a prolonged effect. It was also shown in preclinical trials to produce tissue necrosis and neuritis.
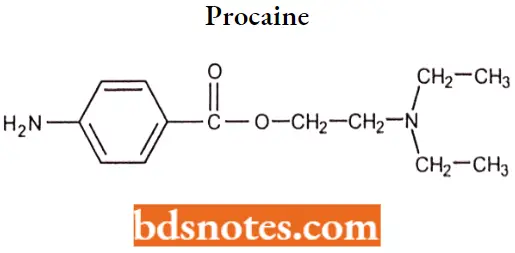
Procaine: Procaine was synthesized to overcome the chemical instability of cocaine. pKa of procaine is 8.9; it has low lipid solubility and the ester group is unstable in basic solutions. It has a slow onset and a short duration of action.
Procaine is available in concentrations ranging from 25% to 10% with pHs adjusted to 5.5 to 6.0 for chemical stability. Procaine is also included in some formulations of Penicillin G to decrease the pain of intramuscular injection.
“How do amide anesthetics differ from ester anesthetics? FAQ explained”
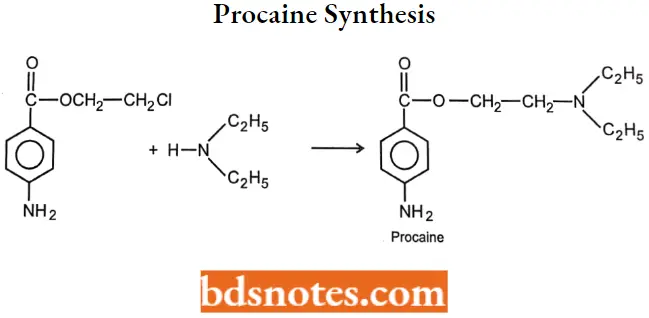
Procaine IUPAC name: 2-(diethylamino)ethyl 4-aminobenzoate.
Procaine MOA: Procaine acts mainly by inhibiting sodium influx through voltage-gated sodium channels in the neuronal cell membrane of peripheral nerves.
- When the influx of sodium is interrupted, an action potential cannot arise and signal conduction is thus inhibited. The receptor site is thought to be located at the cytoplasmic (inner) portion of the sodium channel.
- Procaine has also been shown to bind or antagonize the function of N-Methyl-D-aspartate (NMDA) receptors as well as nicotinic acetylcholine receptors and the serotonin receptor-ion channel complex.
Procaine Metabolism: Metabolism- Procaine is very quickly metabolized in the plasma by cholinesterases and in the liver via ester hydrolysis by a pseudocholinesterase. The in vitro elimination half-life is approximately 60 seconds.
Procaine Uses: Procaine is an anesthetic agent indicated for the production of local or regional anesthesia, particularly for oral surgery. Procaine (like cocaine) has the advantage of constricting blood vessels which reduces bleeding.
It is used for infiltration anesthesia, peripheral nerve block, and spinal block. Procaine has also been investigated as an oral entry inhibitor in the treatment of experienced HIV patients.
Procaine Synthesis:
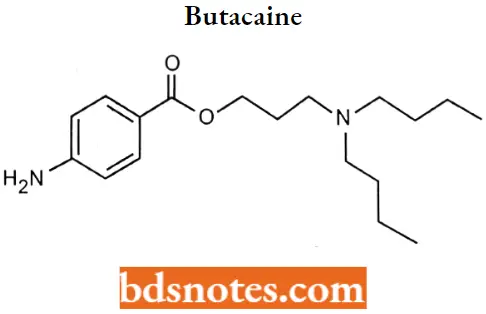
Butacaine
“Early warning signs of gaps in understanding local anesthesia basics: Common questions”
Butacaine IUPAC name: 4-aminobenzoic acid 3-(dibutylamino) propyl ester.
Butacaine MOA: It blocks sodium ion channels thereby preventing the entry of sodium ions into nerve cells. When the influx of sodium is interrupted, an action potential cannot arise and signal conduction is thus inhibited.
Propoxycalne: Propoxycaine is a local anesthetic of tire ester type that has a rapid onset of action and a longer duration of action than procaine hydrochloride
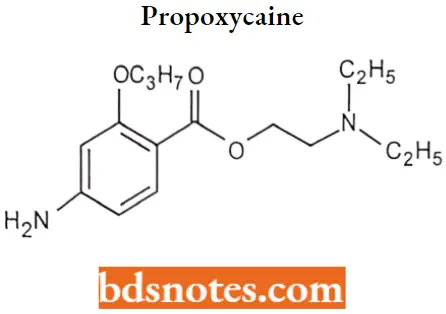
Propoxycalne IUPAC name: 2-(diethylamino)ethyl4-anrino-2-propoxybenzoate.
Propoxycalne MOA: Propoxycaine is a para-aminobenzoic acid ester with local anesthetic activity. Propoxycaine binds to and blocks voltage-gated sodium channels, thereby inhibiting the ionic flux essential for the conduction of nerve impulses. This results in a loss of sensation.
Propoxycalne Metabolism: This drug is hydrolyzed in both the plasma and the liver by plasma esterases.
Propoxycalne Uses: It was used beginning in the 1950s during dental procedures. This medication was used in combination with procaine to aid in anesthesia during dental procedures.
Used in combination with procaine, it was the only dental local anesthetic available in cartridge form. It has been combined with procaine to accelerate its onset of action and provide longer-lasting anesthetic effect.
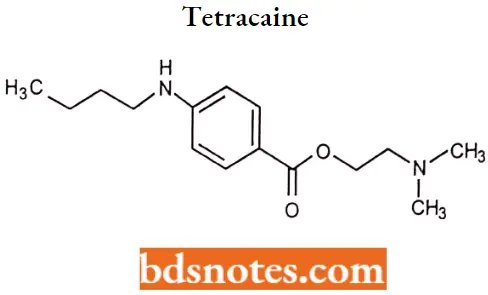
Tetracaine: Tetracaine is an ester local anesthetic currently available in combination with lidocaine as a cream and patch.
“Asymptomatic vs symptomatic effects of ignoring local anesthesia principles: Q&A”
Tetracaine IUPAC name: 2-(dimethylamino) ethyl 4-(butylamino)benzoate.
Tetracaine MOA: Tetracaine is an ester-type anesthetic and produces local anesthesia by blocking the sodium ion channels involved in the initiation and conduction of neuronal impulses.
Tetracaine Metabolism: It is rapidly hydrolyzed by plasma esterases to the primary metabolites: para-aminobenzoic acid and diethylaminoethanol. The activity of both metabolites is unspecified.
Tetracaine Uses: The combination of lidocaine and tetracaine patch is indicated for local dermal analgesia for superficial dermatological procedures and superficial venous access. The combination of lidocaine and tetracaine cream is intended to provide topical local analgesia for superficial dermatological procedures.
Tetracaine Adverse Effects: The most common adverse effects with the combination cream are localized reactions such as erythema (47%), skin discoloration (16%), and edema (14%).
Systemic adverse events were less common, occurring at a rate of <1%, and included vomiting, headache, dizziness, and fever. Similar to other amide and ester anesthetics, CNS excitation and/or depression may occur.
Benoxinate (Oxybuprocaine): Oxybuprocaine is a benzoate ester in which 4-amino-3-butoxybenzoic acid and 2-(diethylamino) ethanol have combined to form the ester bond. It may be less irritating than tetracaine, and the onset and duration of action are similar to tetracaine.
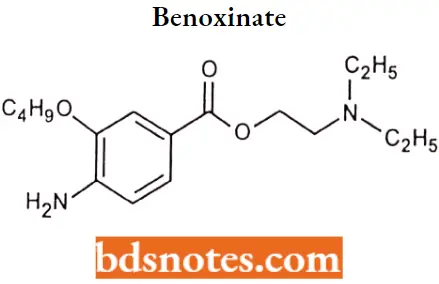
Benoxinate IUPAC name: 4-Amino-3-butoxy-benzoic acid 2-diethylamino-ethyl ester.
Benoxinate MOA: Oxybuprocaine binds to sodium channel and reversibly stabilizes the neuronal membrane which decreases its permeability to sodium ions. Depolarization of the neuronal membrane is inhibited thereby blocking the initiation and conduction of nerve impulses.
Benoxinate Uses: Oxybuprocaine is the name of a local anesthetic, which is used especially in ophthalmology and otolaryngology. It is an ester-based local anesthetic.
Anilide amides derivatives
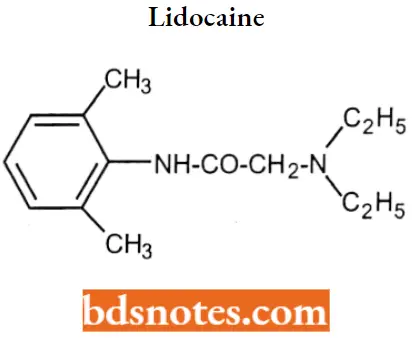
Lidocaine: A local anesthetic and cardiac depressant used as an anti-arrhythmic agent. Lidocaine is the most commonly used amino amide-type local anesthetic.
- Lidocaine is very lipid soluble and, thus, has a more rapid onset and a longer duration of action than most amino ester types local anesthetics, such as procaine and tetracaine.
- It can be administered parenterally (with or without epinephrine) or topically either by itself or in combination with prilocaine or etidocaine as a eutectic mixture.
“Can targeted interventions improve outcomes using local anesthesia knowledge? FAQs provided”
Lidocaine IUPAC name: 2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide.
Lidocaine MOA: Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby affecting local anesthetic, action.
- Lidocaine alters signal conduction in neurons by blocking the fast voltage-gated sodium (Na+) channels in the neuronal cell membrane that are responsible for signal propagation.
- With sufficient blockage the membrane of the postsynaptic neuron will not depolarize and will thus fail to transmit an action potential.
- This creates the anesthetic effect by not merely preventing pain signals from propagating to the brain but by aborting their birth in the first place.
Lidocaine Metabolism: Absorption of lidocaine will be decreased with the addition of epinephrine to the local anesthetic. Lidocaine is primarily metabolized by de-ethylation of the tertiary nitrogen to form monoethylglycinexylidide. (MEGX).
Lidocaine Uses: For the production of local or regional anesthesia. It is also frequently used as a class IB antiarrhythmic agent for the treatment of ventricular arrhythmias, both because it binds and inhibits Na+ channels in the cardiac muscle and because of its longer duration of action than amino ester-type local anesthetic.
Mepivacaine: Mpivacaine has a reasonably rapid onset (more rapid than that of procaine) and medium duration of action (shorter than that of procaine) and is marketed under various trade names including Carbocaine and Polocaine.
It is supplied as the hydrochloride salt of the racemate, which consists of R(-)-mepivacaine and S(+)-mepivacaine in equal proportions. These two enantiomers have markedly different pharmacokinetic properties.
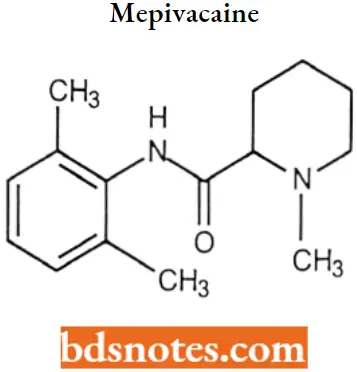
Mepivacaine IUPAC name: N-(2,6-dimethylphenyl)-l-methylpiperidine-2-carboxamide.
Mepivacaine MOA: Mepivacaine blocks the generation and the conduction of nerve impulses, presumably by increasing the threshold for electrical excitation in the nerve, slowing the propagation of the nerve impulse, and reducing the rate of rise of the action potential.
In general, the progression of anesthesia is related to the diameter, myelination, and conduction velocity of affected nerve fibers. Clinically, the order of loss of nerve function is as follows: pain, temperature, touch.
Mepivacaine Metabolism: Rapidly metabolized in the liver by CYP3A4 and CYP1A2 with only a small percentage of the anesthetic (5 percent to 10 percent) being excreted, unchanged in the urine.
The major metabolic biotransformations of mepivacaine are N-dealkylation (to give the N-demethylated compound 2′,6′-pipecoloxylidide) and aromatic hydroxylations. These metabolites are excreted as their corresponding glucuronides.
Mepivacaine Uses: Mepivacaine is used to provide regional analgesia and anesthesia by local infiltration, peripheral nerve block, and epidural and caudal blocks.
- The pharmacologic and toxicologic profile of mepivacaine is quite similar to that of lidocaine, except that mepivacaine is less lipophilic (logDpH 7.4 = 1.95) and has a slightly longer duration of action but lacks the vasodilator activity of lidocaine.
- For this reason, it serves as an alternate choice for lidocaine when the addition of epinephrine is not recommended in patients with hypertensive vascular disease.
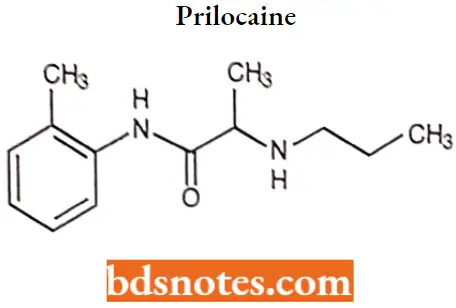
Prilocaine: Prilocaine is a local anesthetic that is similar pharmacologically to lidocaine: Prilocaine hydrochloride is a water-soluble salt available as a solution for nerve block or infiltration in dental procedures.
The pKa of the secondary amine is 7,9 and commercial preparations have a pH of 5.0 to 5.6. Prilocaine has only one ortho substitution on the aromatic ring, making it more susceptible to amide hydrolysis and giving it a shorter duration.
“Differential applications of topical vs injectable local anesthesia: Questions answered”
Prilocaine IUPAC name: N-(2-methylphenyiV2-(propylamino)propanamide.
Prilocaine MOA: Prilocaine acts on sodium channels on the neuronal cell membrane, limiting the spread of seizure activity and reducing seizure propagation. The antiarrhythmic actions are mediated through effects on sodium channels in Purkinje fibers.
Prilocaine Metabolism: Prilocaine is metabolized in both the liver and the kidney and excreted via the kidney.
Prilocaine Uses: Currently, it is used most often for infiltration anesthesia in dentistry. Prilocaine is used for intravenous regional anesthesia as the risk of CNS toxicity is low because of its quick metabolism.
Etidocaine: Etidocaine is a local anesthetic with rapid onset and long action, similar to BUPIVACAINE.
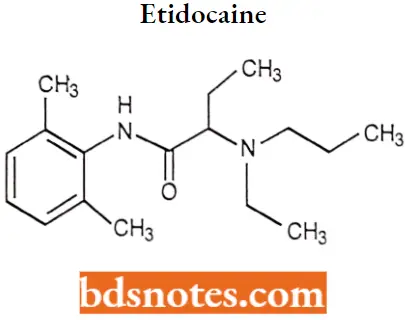
Etidocaine IUPAC name: N-(2,6-dimethylphenyl)-2-[ethyl(propyl)amino]butanamide.
Etidocaine MOA: Etidocaine is the most potent amino amide local anesthetic. Etidocaine differs from lidocaine by the addition of an alkyl chain and the extension of one ethyl group on the tertiary amine to a butyl group.
The additional lipophilicity gives etidocaine a quicker onset, longer half-life, and an increased potency compared with lidocaine. The tertiary nitrogen pKa is 7.74, which is similar to lidocaine’s pKa (7.8)
Etidocaine Uses: It is used for epidural anesthesia, topical anesthesia, and for peripheral nerve or plexus block. Etidocaine blocks large fast-conducting neurons quicker than the sensory neurons. It is given by injection during surgical procedures and labor and delivery.
Etidocaine Adverse effect: Etidocaine has the same potential for cardiac toxicity as bupivacaine.
Miscellaneous
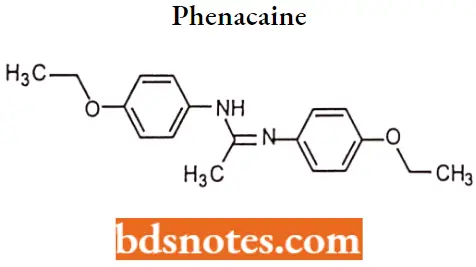
Phenacaine
“Steps to apply local anesthesia knowledge in clinical practice: Diagnosis vs treatment: Q&A guide”
Phenacaine IUPAC name: N,N’-bis(4-ethoxyphenyl)ethanimidamide.
Phenacaine MOA: It is a sodium channel blocker. It blocks sodium ion passage into the cell and thus blocks the formation and propagation of the action potential.
Phenacaine Use: It is a local anesthetic approved for ophthalmic use.
Diperodon
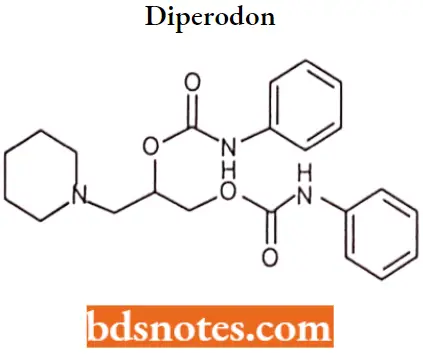
Diperodon IUPAC name: [2-(phenylcarbamoyloxy)-3-piperidin-1-ylpropyl] N-phenylcarbamate.
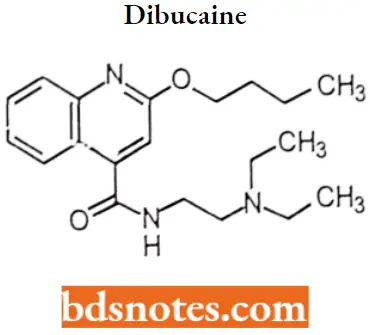
Dibucaine: Dibucaine is a topical amide anesthetic available in over-the-counter creams and ointments used to treat minor conditions such as sunburns and hemorrhoids. Dibucaine is a quinoline derivative and amino amide with anesthetic activity.
Dibucaine is a Standardized Chemical Allergen. The physiologic effect of dibucaine is through Increased Histamine Release, and Cell-mediated Immunity, The chemical classification of dibucaine is Allergens.
“Role of nerve blocks in surgical procedures: Questions answered”
Dibucaine IUPAC name: 2-butoxy-N-[2-(diethylamino)ethyl]quinoline-4-carboxamide.
Dibucaine MOA: Dibucaine reversibly binds to and inactivates sodium channels in the neuronal cell membrane.
Inhibition of sodium channels prevents the depolarization of nerve cell membranes and inhibits subsequent propagation of impulses along the course of the nerve, thereby limiting the excitation of nerve endings. This results in loss of sensation.
Dibucaine Metabolism: Metabolites of dibucaine identified in the urine of rats, rabbits, and humans included hydroxylated metabolites of the quinoline ring, monohydroxylated and dihydroxylated metabolites of the O-alkyl side chain (2-and 3-position), and the N-de-ethylated dibucaine metabolite.
Dibucaine Uses: It is a local anesthetic of the amide type now generally used for surface anesthesia. It is one of the most potent and toxic of the long-acting local anesthetics and its parenteral use is restricted to spinal anesthesia.
Dibucaine Toxicity: It is highly toxic when taken orally, inducing seizures, coma, and death in several children who accidentally ingested it.
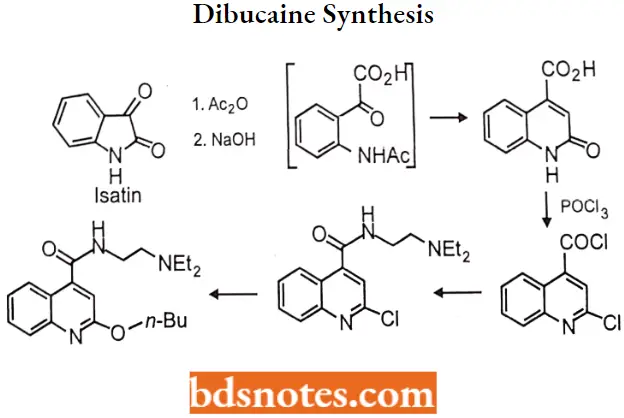
Dibucaine Synthesis:
“How does epinephrine prolong the effects of local anesthesia? FAQ explained”
Local Anaesthetics Multiple Choice Questions And Answers
Question 1. Local anesthetics produce:
- Analgesia, amnesia, loss of consciousness
- Blocking pain sensation without loss of consciousness
- Alleviation of anxiety and pain with an altered level of consciousness
- A stupor or somnolent state
Answer: 2. Blocking pain sensation without loss of consciousness
Question 2. Which one of the following drugs has an allergenic effect?
- Lidocaine
- Procaine
- Dibucaine
- Bupivacaine
Answer: 3. Dibucaine
Question 3. Most local anesthetic agents consist of:
- Lipophilic group
- Intermediate chain
- Amino group
- All of the above
Answer: 4. All of the above
Question 4. Which one of the following groups is responsible for the duration of the local anesthetic action?
- Intermediate chain
- Lipophilic group
- Ionizable group
- All of the above
Answer: 1. Intermediate chain
“Early warning signs of complications from ignoring local anesthesia protocols: Common questions”
Question 5. Which one of the following groups is responsible for the potency of local anesthetics?
- Ionizable group
- Intermediate chain
- Lipophilic group
- All of the above
Answer: 3. Lipophilic group
Question 6. The primary mechanism of action of local anesthetic is _________.
- Activation of ligand-gated potassium channels
- Blockade of voltage-gated sodium channels
- Stimulation of voltage-gated N-type calcium channels
- Blockade the GABA-gated chloride channels
Answer: 2. Blockade of voltage-gated sodium channels
Question 7. Which one of the following local anesthetics is an ester of benzoic acid?
- Lidocaine
- Procaine
- Ropivacaine
- Cocaine
Answer: 4. Cocaine
Question 8. Local anesthetics are:
- Weak bases
- Weak acids
- Salts
- None of the above
Answer: 1. Weak bases
Question 9. For therapeutic application local anesthetics reasons of:
- Less toxicity and higher potency
- Higher stability and greater lipid solubility
- Less local tissue damage and more potency
- More stability and greater water solubility
Answer: 4. More stability and greater water solubility
“Asymptomatic vs symptomatic effects of outdated local anesthesia practices: Answered”
Question 10. Which of the following statements is not correct for local anesthetics?
- In a tissue, they exist either as an uncharged base or as a cation
- A charged cationic form penetrates biological membranes more readily than an uncharged form
- Local anesthetics are much less effective in inflamed tissues
- Low pH in inflamed tissues decreases the dissociation of nonionized molecules
Answer: 2. A charged cationic form penetrates biological membranes more readily than an uncharged form
Question 11. Which of the following anesthetic agents shows antiarrhythmic action?
- Lidocaine
- Bupivacaine
- Procaine
- Etidocaine
Answer: 1. Lidocaine
Local Anaesthetics Short Questions And Answers
Question 1. Define Local Anesthetics
Answer:
Local anesthetics are the drugs when given either topically or parenterally to a localized area, produce loss of sensation with or without loss of consciousness by reversibly blocking the generation and conduction of nerve impulses.
Question 2. Write the characteristics of an ideal Local Anesthetic
Answer:
The following are the characteristics of an ideal Local Anesthetic.
- The action of local anesthetic must be reversible.
- It must be non-irritating to the tissues and should not produce any secondary local reaction
- It should have a low degree of systemic toxicity and have sufficient potency to provide complete anesthesia.
- It should have a rapid onset and be of sufficient duration to be advantageous.
- It should have sufficient penetrating properties to be effective as a topical anesthetic.
- It should be relatively free from producing allergic reactions.
- It should be stable in solution and undergo biotransformation readily within the body.
- It should be either sterile or capable of being sterilized by heat without deterioration.
“Can preventive measures reduce risks of local anesthesia side effects? FAQs provided”
Question 3. Write the structural requirements for Local Anesthetics
Answer:
Following are the structural requirements for Local Anesthetic.
- Lipophilic aromatic group (usually substituted)
- A connecting group that is either an ester or an amide
- An ionizable amino group (hydrophilic group).
Question 4. Draw the structures of Propoxycaine and Butamben
Answer:
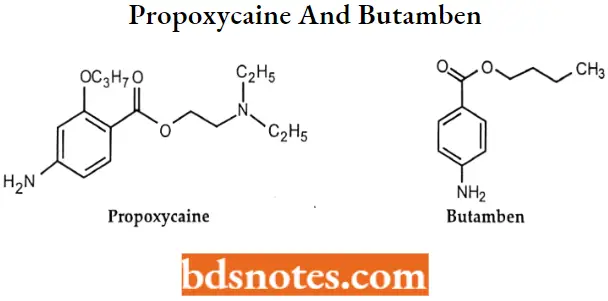
Question 5. Write the mechanism of action of local anesthetics.
Answer:
Local anesthetics block sodium channels. When the local anesthetic binds, it blocks sodium ion passage into the cell and thus blocks the formation and propagation of the action potential.
- This blocks the transmittance of the message of “pain” or even “touch” from getting to the brain.
- The ability of a local anesthetic to block action potentials depends on the ability of the drug to penetrate the tissue surrounding the targeted nerve as well as the ability of the drug to access the binding site on the sodium channel.
“Differential applications of short-acting vs long-acting local anesthetics: Q&A”
Question 6. Write the uses of Lidocaine and Tetracaine
Answer:
Lidocaine– For the production of local or regional anesthesia. Lidocaine is also frequently used as a class IB antiarrhythmic agent for the treatment of ventricular arrhythmias.
Tetracaine -The combination of lidocaine and tetracaine patch is indicated for local dermal analgesia for superficial dermatological procedures and superficial venous access. The combination of lidocaine and tetracaine cream is intended to provide topical local analgesia for superficial dermatological procedures.
Leave a Reply