Structure And Function Of The Kidneys
Structure and function are closely linked in the kidneys. Consequently, an appreciation of the gross anatomic and histologic features of the kidneys is a prerequisite for an understanding of their function.
Structure Of The Kidneys
Gross Anatomy
The kidneys are paired organs that lie on the posterior wall of the abdomen behind the peritoneum on either side of the vertebral column.
In the adult human, each kidney weighs between 115 and 170 g and is approximately 11 cm long, 6 cm wide, and 3 cm thick.
The gross anatomic features of the human kidney are illustrated in 1. The medial side of each kidney contains an indentation, through which passes the renal artery and vein, nerves, and pelvis.
If a kidney were cut in half, two regions would be evident: an outer region called the cortex and an inner region called the medulla.
The cortex and medulla are composed of nephrons (the functional units of the kidney), blood vessels, lymphatics, and nerves. The medulla in the human kidney is divided into conical masses called renal pyramids.
“What is the structure and function of the kidneys?”
The base of each pyramid originates at the corticomedullary border, and the apex terminates in a papilla, which lies within a minor calyx. Minor calyces collect urine from each papilla.
The numerous minor calyces expand into two or three open-ended pouches, which are the major calyces. The major calyces in turn feed into the pelvis.
The pelvis represents the upper, expanded region of the ureter, which carries urine from the pelvis to the urinary bladder.
The walls of the calyces, pelvis, and ureters contain smooth muscle that contracts to propel the urine toward the urinary bladder.

“Understanding the role of kidneys in human physiology”
The blood flow to the two kidneys is equal to about 25% (1.25 L/min) of the cardiac output in resting individuals. However, the kidneys constitute less than 0.5% of total body weight.
As illustrated, the renal artery branches progressively to form the interlobar artery, the arcuate artery, the interlobular artery, and the afferent arteriole, which leads into the glomerular capillaries.
The glomerular capillaries come together to form the efferent arteriole, which leads into a second capillary network, the peritubular capillaries, which supply blood to the nephron.
The vessels of the venous system run parallel to the arterial vessels and progressively form the interlobular vein, arcuate vein, interlobar vein, and renal vein, which courses beside the ureter.
Ultrastructure of the Nephron
The functional unit of the kidneys is the nephron. Each human kidney contains approximately 1.2 million nephrons, which are hollow tubes composed of a single cell layer.
The nephron consists of a renal corpuscle, proximal tubule, loop of Henle, distal tubule, and collecting duct system.
The renal corpuscle consists of glomerular capillaries and Bowman’s capsule. The proximal tubule initially forms several coils, followed by a straight piece that descends toward the medulla.
The next segment is the loop of Henle, which is composed of the straight part of the proximal tubule, the descending thin limb (which ends in a hairpin turn).
The ascending thin limb (only in nephrons with long loops of Henle), and the thick ascending limb. Near the end of the thick ascending limb, the nephron passes between the afferent and efferent arterioles of the same nephron.
“How do the kidneys maintain homeostasis in the body?”
This short segment of the thick ascending limb that touches the glomerulus is called the macula densa.
The distal tubule begins a short distance beyond the macula densa and extends to the point in the cortex where two or more nephrons join to form a cortical collecting duct.
The cortical collecting duct enters the medulla and becomes the outer medullary collecting duct and then the inner medullary collecting duct.
Each nephron segment is made up of cells that are uniquely suited to perform specific transport functions.
Proximal tubule cells have an extensively amplified apical membrane (the urine side of the cell) called the brush border, which is present only in the proximal tubule of the nephron.
The basolateral membrane (the blood side of the cell) is highly invaginated. These invaginations contain many mitochondria.
In contrast, the descending and ascending thin limbs of Henle’s loop have poorly developed apical and basolateral surfaces and few mitochondria.

“Importance of studying kidney structure and function”
The cells of the thick ascending limb and the distal tubule have abundant mitochondria and extensive infoldings of the basolateral membrane.
The collecting duct is composed of two cell types: principal cells and intercalated cells. Principal cells have a moderately invaginated basolateral membrane and contain few mitochondria.
Principal cells play an important role in sodium chloride (NaCl) reabsorption and K+ secretion.
Intercalated cells, which play an important role in regulating acid-base balance, have a high density of mitochondria.
One population of intercalated cells secretes H+ (i.e., reabsorbs bicarbonate [HCO3–]) and a second population of intercalated cells secretes HCO–3.
The final segment of the nephron, the inner medullary collecting duct, is composed of inner medullary collecting duct cells.
Cells of the inner medullary collecting duct have poorly developed apical and basolateral surfaces and few mitochondria.
Except for intercalated cells, all cells in the nephron have in the apical plasma membrane a single nonmotile primary cilium that protrudes into the tubule flid.
Primary cilia are mechanosensors (i.e., they sense changes in the flow rate of tubule fluid) and chemosensors (i.e., they sense or respond to compounds in the surrounding fluid).
They initiate Ca++-dependent signaling pathways, including those that control kidney cell function, proliferation, differentiation, and apoptosis (i.e., programmed cell death).
“Common parts of the kidney explained”

Polycystin 1 (encoded by the PKD1 gene) and polycystin 2 (encoded by the PKD2 gene) are expressed in the membrane of primary cilia, and the PKD1/PKD2 complex mediates the entry of Ca++ into cells.
PKD1 and PKD2 are thought to play an important role in flow-dependent K+ secretion by principal cells of the collecting duct.
Increased flow of tubule fluid in the collecting duct is a strong stimulus for K+ secretion.
Increased flow bends the primary cilium in principal cells, which activates the PKD1/PKD2 Ca++ conducting channel complex, allowing Ca++ to enter the cell and increase intracellular [Ca++].
The increase in [Ca++ activates K+ channels in the apical plasma membrane, which enhances K+ secretion from the cell into the tubule fluid.
Nephrons may be subdivided into superficial and juxtamedullary types. The glomerulus of each superficial nephron is located in the outer region of the cortex.
Its loop of Henle is short, and its efferent arteriole branches into peritubular capillaries that surround the nephron segments of its own and adjacent nephrons.
This capillary network conveys oxygen and important nutrients to the nephron segments in the cortex and delivers substances to the nephron for secretion (i.e., the movement of a substance from the blood into the tubular fluid).
“Anatomy of the nephron: Structure and function”
And serves as a pathway for the return of reabsorbed water and solutes to the circulatory system. A few species, including humans, also possess very short superficial nephrons whose Henle’s loops never enter the medulla.
Autosomal dominant polycystic kidney disease (ADPKD), which is the most common inherited kidney disease, occurs in 1 in 1000 people.
Approximately 12.5 million people worldwide have ADPKD, which is caused primarily by mutations in the genes PKD1 (85% of cases) and PKD2 (~15% of cases).
The major phenotype of ADPKD is an enlargement of the kidneys related to the presence of hundreds to thousands of renal cysts that can be as large as 20 cm in diameter.
Cysts also are seen in the liver and other organs. About 50% of patients with ADPKD progress to renal failure by the age of 60 years.
Although it is not clear how mutations in PKD1 and PKD2 cause ADPKD, renal cyst formation results from defects in Ca++ uptake that lead to alterations in Ca++-dependent signaling pathways, including those that control kidney cell proliferation, differentiation, and apoptosis.
The glomerulus of each juxtamedullary nephron is located in the region of the cortex adjacent to the medulla.
In comparison with the superficial nephrons, the juxtamedullary nephrons differ anatomically in two important ways: the loop of Henle is longer and extends deeper into the medulla.
The efferent arteriole forms not only a network of peritubular capillaries but also a series of vascular loops called the vasa recta.
The vasa recta descend into the medulla, where they form capillary networks that surround the collecting ducts and ascending limbs of the loop of Henle. The blood returns to the cortex in the ascending vasa recta.
Although less than 0.7% of the blood enters the vasa recta, these vessels subserve important functions in the renal medulla, including
- Conveying oxygen and important nutrients to nephron segments,
- Delivering substances to the nephron for secretion,
- Serving as a pathway for the return of reabsorbed water and solutes to the circulatory system, and
- Concentrating and diluting the urine.
“Role of the glomerulus in kidney filtration”
Ultrastructure Of The Glomerulus
The first step in urine formation begins with the passive movement of a plasma ultrafiltrate from the glomerular capillaries into Bowman’s space.
The term ultrafiltration refers to the passive movement of fluid that is similar in composition to plasma.
Except that the protein concentration in the ultrafiltrate is lower than that in the plasma, from the glomerular capillaries into Bowman’s space.
To appreciate the process of ultrafiltration, one must understand the anatomy of the glomerulus.
The glomerulus consists of a network of capillaries supplied by the afferent arteriole and drained by the efferent arteriole.

“How does the renal tubule work in urine formation?”
During embryologic development, the glomerular capillaries press into the closed end of the proximal tubule, forming Bowman’s capsule.
As the epithelial cells thin on the outside circumference of Bowman’s capsule, they form the parietal epithelium.

“Structure of the renal cortex and medulla explained”
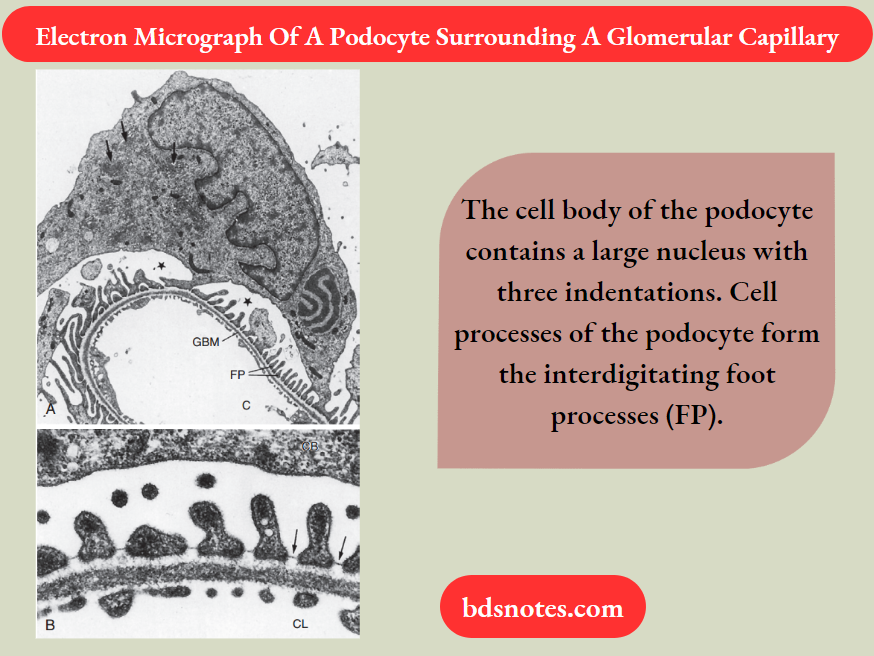
The epithelial cells in contact with the capillaries thicken and develop into podocytes, which form the visceral layer of Bowman’s capsule.
The space between the visceral layer and the parietal layer is Bowman’s space, which at the urinary pole (i.e., where the proximal tubule joins Bowman’s capsule) of the glomerulus becomes the lumen of the proximal tubule.
“Biomechanics of blood flow in the kidneys”
The endothelial cells of glomerular capillaries are covered by a basement membrane, which is surrounded by podocytes. The capillary endothelium, basement membrane, and foot processes of podocytes form the so-called filtration barrier.
The endothelium is fenestrated (i.e., it contains 700-Å holes where 1 Å = 10-10 m) and is freely permeable to water, and small solutes (such as Na+, urea, and glucose).
And small proteins but are not permeable to large proteins, red blood cells, white blood cells, or platelets.
Because endothelial cells express glycoproteins on their surface, they minimize the filtration into Bowman’s space of albumin, the most abundant plasma protein, and small plasma proteins.
In addition to their role as a barrier to filtration, the endothelial cells synthesize a number of vasoactive substances (For Example., nitric oxide, a vasodilator, and endothelin-1, a vasoconstrictor) that are important in controlling renal plasma flow.
The basement membrane, which is a porous matrix of negatively charged proteins, including type 4 collagen, laminin, the proteoglycans agrin and perlecan, and fibronectin, is also an important filtration barrier to plasma proteins.

“Role of kidneys in regulating blood pressure”
The podocytes have long fingerlike processes that completely encircle the outer surface of the capillaries.
The processes of podocytes interdigitate to cover the basement membrane and are separated by apparent gaps called filtration slits.
Each filtration slit is bridged by a thin diaphragm, the filtration slit diaphragm, which appears as a continuous structure when viewed by electron microscopy.
The filtration slit diaphragm is composed of several proteins including nephrin, NEPH-1, and podocin, along with intracellular proteins that associate with slit diaphragm proteins, including CD2-AP and α-actinin 4 (ACTN4).
Filtration slits, which function primarily as a size-selective filter, minimize the filtration of proteins and macromolecules that cross the basement membrane from entering Bowman’s space.

“How do kidneys control fluid and electrolyte balance?”
Nephrotic syndrome is produced by a variety of disorders and is characterized by an increase in the permeability of the glomerular capillaries to proteins and by a loss of normal podocyte structure, including effacement (i.e., thinning) of foot processes.
The augmented permeability to proteins results in an increase in urinary protein excretion (proteinuria). Thus the appearance of proteins in the urine can indicate kidney disease.
Hypoproteinemia often develops in persons with this syndrome as a result of proteinuria. In addition, generalized edema commonly is seen in
persons with nephrotic syndrome.
Mutations in several genes that encode slit diaphragm proteins, including nephrin, NEPH-1, and podocin, along with intracellular proteins that associate with slit diaphragm proteins.
Including CD2-AP and α-actinin 4 (ACTN4), or a knockout of these genes in mice, cause proteinuria and kidney disease.
For example, mutations in the nephrin gene (NPHS1) lead to abnormal or absent slit diaphragms, causing massive proteinuria and renal failure (i.e., congenital nephrotic syndrome).
In addition, mutations in the podocin gene (NPHS2) cause autosomal recessive, steroid-resistant nephrotic syndrome.
These naturally occurring mutations and knockout studies in mice demonstrate that nephrin, NEPH-1, podocin, CD2-AP, and α-actinin 4 play key roles in podocyte structure and function.
Alport syndrome is characterized by hematuria (i.e., blood in the urine) and progressive glomerulonephritis (i.e., inflammation of the glomerular capillaries) and accounts for 1% to 2% of all cases of end-stage renal disease.
Alport syndrome is caused by mutations in type 4 collagen, a major component of the glomerular basement membrane.
In about 80% of patients with Alport syndrome, the disease is X-linked with mutations in the COL4A5 gene.
Fifteen percent of patients also have mutations in type 4 collagen genes (COL4A3 and COL4A4); six have been identified, but the mode of inheritance is autosomal recessive.
The remaining 5% of patients with Alport syndrome have autosomal dominant disease that arises from heterozygous mutations in the COL4A3 or COL4A4 genes.
In persons with Alport syndrome, the glomerular basement membrane becomes irregular in thickness and fails to serve as an effective filtration barrier to blood cells and protein.
Another important component of the renal corpuscle is the mesangium, which consists of mesangial cells and the mesangial matrix.
“Impact of kidneys on acid-base regulation”

“Kidneys and their role in waste excretion”
Mesangial cells are involved in the development of immune complex-mediated glomerular disease.
Because the glomerular basement membrane does not completely surround all glomerular capillaries, some immune complexes can enter the mesangial area without crossing the glomerular basement membrane.
Accumulation of immune complexes induces the infiltration of inflmmatory cells into the mesangium and promotes the production of proinflammatory cytokines and autacoids by cells in the mesangium.
These cytokines and autacoids enhance the inflmmatory response. This inflmmatory response can lead to cell death, scarring and eventually obliterating the glomerulus.
Mesangial cells, which possess many properties of smooth muscle cells, provide structural support for the glomerular capillaries and secrete the extracellular matrix.
Exhibit phagocytic activity that removes macromolecules from the mesangium, and secretes prostaglandins and proinflammatory cytokines.
Because they also contract and are adjacent to glomerular capillaries, mesangial cells may influence the glomerular filtration rate (GFR) by regulating blood flow through the glomerular capillaries or by altering the capillary surface area.
Mesangial cells located outside the glomerulus (between the afferent and efferent arterioles) are called extraglomerular mesangial cells.
“Can kidney dysfunction affect hormone production?”
Ultrastructure Of The Juxtaglomerular Apparatus
The juxtaglomerular apparatus (JGA) is one component of an important feedback mechanism, the tubuloglomerular feedback mechanism, that is described. The following structures make up the JGA:
- The macula densa of the thick ascending limb
- The extraglomerular mesangial cells
- The renin- and angiotensin II–produce granular cells of the afferent arteriole.
The cells of the macula densa represent a morphologically distinct region of the thick ascending limb. This region passes through the angle formed by the afferent and efferent arterioles of the same nephron.
The cells of the macula densa are in contact with the extraglomerular mesangial cells and the granular cells of the afferent arterioles.
Granular cells of the afferent arterioles are derived from metanephric mesenchymal cells. They contain smooth muscle myofilaments and they manufacture, store, and release renin.
Renin is involved in the formation of angiotensin II and ultimately in the secretion of aldosterone.
The JGA is one component of the tubuloglomerular feedback mechanism that is involved in the autoregulation of renal blood flow and the GFR.
“Steps in the filtration process of the kidneys”
Innervation Of The Kidneys
Renal nerves regulate renal blood flow, GFR, and salt and water reabsorption by the nephron. The nerve supply to the kidneys consists of sympathetic nerve fibers that originate in the celiac plexus.
No parasympathetic innervation occurs. Adrenergic fibers that innervate the kidneys release norepinephrine.
The adrenergic fibers lie adjacent to the smooth muscle cells of the major branches of the renal artery (the interlobar, arcuate, and interlobular arteries) and the afferent and efferent arterioles.
Moreover, sympathetic nerves innervate the renin-producing granular cells of the afferent arterioles. Renin secretion is stimulated by increased sympathetic activity.
Nerve fibers also innervate the proximal tubule, loop of Henle, distal tubule, and collecting duct; activation of these nerves enhances Na+ reabsorption by these nephron segments.
Leave a Reply