Regulation Of Body Fluid Osmolality Regulation Of Water Balance
As described, water constitutes approximately 60% of the healthy adult human body. Body water is divided into two compartments (i.e., intracellular fluid and extracellular fluid [ECF]), which are in osmotic equilibrium.
“What is the regulation of body fluid osmolality?”
- Water intake into the body generally occurs orally, and the water ingested is absorbed into the ECF by the gastrointestinal tract via a mechanism similar to that which mediates water absorption by the proximal tubule.
- However, in clinical situations, intravenous infusion is an important route of water entry. Regardless of the route of entry (oral versus intravenous), water first enters the ECF and then equilibrates with the intracellular fluid. The
- kidneys are responsible for regulating water balance and under most conditions is the major route for the elimination of water from the body (Table 5-1). Other routes of water loss from the body include evaporation from the cells of the skin and respiratory passages.
- Collectively, water loss by these routes is termed insensible water loss because people are unaware of its occurrence. The production of sweat accounts for the loss of additional water.
- Water loss by this mechanism can increase dramatically in a hot environment, with exercise, or in the presence of fever. Finally, water can be lost from the gastrointestinal tract.
- Fecal water loss is normally small (~100 mL/day) but can increase dramatically with diarrhea (for example., 20 L/day in persons with cholera). Vomiting also can cause gastrointestinal water loss.
Normal Routes of Water Gain and Loss in Adults at Room Temperature (23°C):

“Understanding the role of water balance in human physiology”
Effect of Environmental Temperature and Exercise on Water Loss and Intake in Adults:
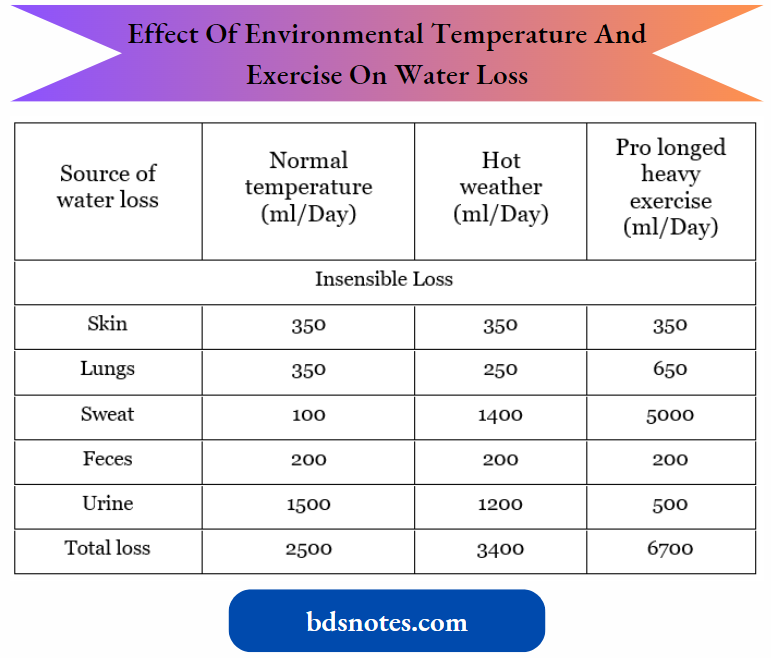
“How does the body regulate osmolality and water balance?”
Although water loss from sweating, defecation, and evaporation from the lungs and skin can vary depending on the environmental conditions or during pathologic conditions, the loss of water by these routes cannot be regulated.
- In contrast, the renal excretion of water is tightly regulated to maintain whole-body water balance. The maintenance of water balance requires that water intake and loss from the body are precisely matched. If intake exceeds losses, a positive water balance exists.
- Conversely, when intake is less than losses, a negative water balance exists.
When water intake is low or water losses increase, the kidneys conserve water by producing a small volume of hyperosmotic urine concerning plasma. - When water intake is high, a large volume of hypoosmotic urine is produced. In a healthy person, the urine osmolality (Uosm) can vary from approximately 50 to 1200 mOsm/kg H2O, and the corresponding urine volume can vary from approximately 18 to 0.5 L/day.
- It is important to recognize that disorders of water balance are manifested by alterations in the body fluid osmolality, which usually are measured by changes in
“Importance of maintaining body fluid osmolality”
When plasma osmolality (Posm) is reduced (i.e., hypos- molality), water moves from the extracellular fluid into cells, causing them to swell.
- Symptoms associated with hypo osmolality are related primarily to swelling of brain cells. For example, a rapid decrease in Posm can alter neurologic function and thereby cause nausea, malaise, headache, confusion, lethargy, seizures, and coma.
- When Posm is increased (i.e., hyperosmolality), water is lost from cells, causing them to shrink. The symptoms of an increase in Posm also are primarily neurologic and include lethargy, weakness, seizures, coma, and even death.
- The symptoms associated with changes in body fluid osmolality vary depending on how quickly osmolality is changed. Rapid changes in osmolality (i.e., over hours) are less well tolerated than changes that occur more gradually (i.e., over days to weeks).
- Indeed, when alterations in body fluid osmolality have developed over an extended period, such persons may be entirely asymptomatic.
- This situation reflects the ability of cells over time either to eliminate intracellular osmoles, as occurs with hyperosmolality, or to generate new intracellular osmoles in response to hyperosmolality and thus minimize changes in cell volume of the neurons.
- This ability has important clinical implications when treating a patient with an abnormal plasma osmolality. For example, rapid correction of the osmolality of a person who has had long-standing hyperosmolality of the body fluids can lead to the development of osmotic demyelination syndrome.
“Common mechanisms of water balance regulation explained”
The syndrome can result in paralysis of multiple muscle groups and can be fatal.

“Role of antidiuretic hormone (ADH) in osmolality regulation”
Plasma osmolality (Posm). Because the major determinant of plasma osmolality is Na+ (with its anions Cl- and HCO–3), these disorders also result in alterations in the plasma [Na+]
- When an abnormal plasma [Na+] is observed in an individual, it is tempting to suspect a problem in Na+ balance. However, the problem usually is related to water balance, not Na+ balance.
- As described, changes in Na+ balance result in alterations in the volume of the ECF, not its osmolality.
- Under steady-state conditions, the kidneys control water excretion independently of their ability to control the excretion of various other physiologically important substances such as Na+, K+, and urea
- Indeed, this ability is necessary for survival because it allows water balance to be achieved without upsetting the other homeostatic functions of the kidneys.
- This chapter discusses the mechanisms by which the kidneys maintain water balance by excreting either hypoosmotic (dilute) or hyperosmotic (concentrated) urine.
The control of arginine vasopressin (AVP) secretion and its important role in regulating the excretion of water by the kidneys are also explained.
Arginine Vasopressin
AVP, also known as antidiuretic hormone, acts on the kidneys to regulate the volume and osmolality of the urine.
- When plasma AVP levels are low, a large volume of urine is excreted (diuresis), diluting the urine.
- When plasma levels are high, a small volume of urine is excreted (antidiuresis), and the urine is concentrated.
- Illustrates the effect of AVP on the urine flow rate and osmolality. The excretion of

“How does ADH affect water reabsorption in the kidneys?”
As already noted, AVP does not appreciably alter the excretion of solute, which underscores the fact that AVP controls water excretion and maintains water balance without altering the excretion and homeostatic control of other substances.
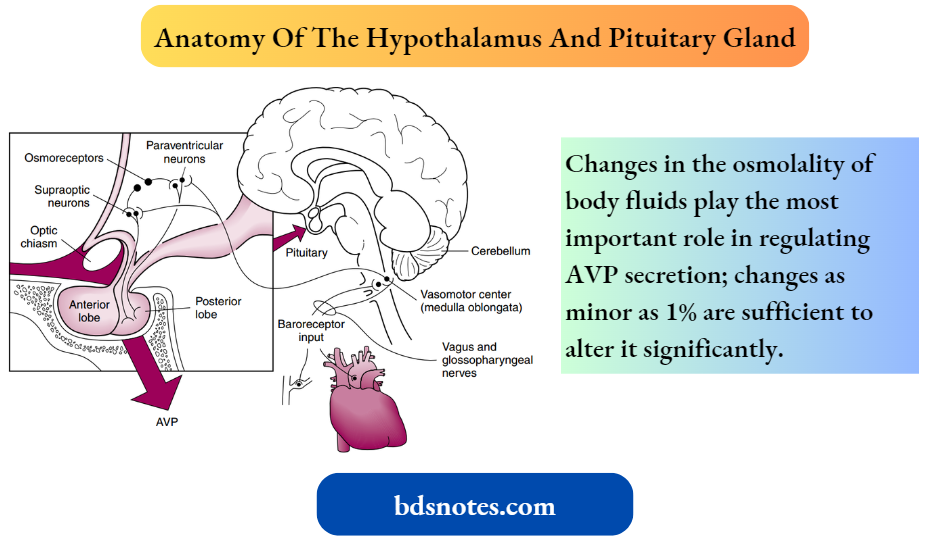
- AVP is a small peptide that is nine amino acids in length. It is synthesized in neuroendocrine cells located within the supraoptic and paraventricular nuclei of the hypothalamus.
- The synthesized hormone is packaged in granules that are transported down the axon of the cell and stored in the nerve terminals located in the neurohypophysis (posterior pituitary).
- The anatomy of the hypothalamus and pituitary gland. The secretion of AVP by the posterior pituitary can be influenced by several factors.
- The two primary physiologic regulators of AVP secretion are the osmolality of the body fluids (osmotic) and the volume and pressure of the vascular system (hemodynamic). Other factors that can alter AVP secretion include nausea
Arginine Vasopressin At The Cellular Level: The gene for arginine vasopressin (AVP) is found on chromosome 20. It contains approximately 2000 base pairs with three exons and two introns.
- The gene codes for a 145 amino acid prohormone that consists of a signal peptide, the AVP molecule, neurophysin, and a glycopeptide (copeptin).
- As the cell processes the prohormone, the signal peptide is cleaved off in the rough endoplasmic reticulum. Once packaged in neurosecretory granules, the preprohormone is further cleaved into AVP, neurophysin, and copeptin molecules.
- The neurosecretory granules are then transported down the axon to the posterior pituitary and stored in the nerve endings until released.
- When the neurons are stimulated to secrete AVP, the action potential opens Ca++ channels in the nerve terminal, which raises the intracellular [Ca++] and causes exocytosis of the neurosecretory granules.
- All three peptides are secreted in this process. Neurophysin and copeptin do not have an identified physiologic function.
- (stimulates), atrial natriuretic peptide (inhibits), and angiotensin II (stimulates). Several drugs, prescription and nonprescription, also affect AVP secretion. For example, nicotine stimulates secretion, whereas ethanol inhibits secretion.
“Impact of aldosterone on osmolality and water balance”
Osmotic Control Of Arginine Vasopressin Secretion:
Changes in the osmolality of body fluids play the most important role in regulating AVP secretion; changes as minor as 1% are sufficient to alter it significantly.
- Although the neurons in the supraoptic and paraventricular nuclei respond to changes in body fluid osmolality by altering their secretion of AVP, it is clear that separate cells exist in the anterior hypothalamus that sense changes in body fluid osmolality and regulate the activity of the AVP-secreting neurons.
- These cells, termed osmoreceptors, appear to sense changes in body fluid osmolality by either shrinking or swelling.
“Biomechanics of osmosis in body fluids explained”
The osmoreceptors respond only to solutes in plasma that are effective osmoles. For example, urea is an ineffective osmole when the function of osmoreceptors is considered.
- Thus elevation of the plasma urea concentration alone has little or no effect on AVP secretion.
- When the effective osmolality of the plasma increases, the osmoreceptors send signals to the AVP synthesizing/secreting cells located in the supraoptic and paraventricular nuclei of the hypothalamus, and AVP synthesis and secretion are stimulated.
- Conversely, when the effective osmolality of the plasma is reduced, secretion is inhibited.
- Because AVP is rapidly degraded in the plasma, circulating levels can be reduced to zero within minutes after secretion is inhibited. As a result, the AVP system can respond rapidly to fluctuations in body fluid osmolality.
Illustrates the effect of changes in plasma osmolality on circulating AVP levels. The set point of the system is the plasma osmolality value at which AVP secretion begins to increase.
- Below this set point, virtually no AVP is released. Above this set point, the slope of the relationship is quite steep and accounts for the sensitivity of this system. The set point varies among individuals and is genetically determined.
- In healthy adults, it varies from 275 to 290 mOsm/kg H2O (average ~280 to 285 mOsm/kg H2O). As described later in this chapter, the set point shifts in response to changes in blood volume and pressure.
- It also shifts during pregnancy, with the osmolality of the mother’s body fluids decreasing during the third trimester.
- The reasons for the shift of the set point during pregnancy are not completely known but likely involve hormones (for example., relaxin) whose circulating levels are elevated at this stage of pregnancy.
Hemodynamic Control Of Arginine Vasopressin Secretion:
A decrease in blood volume or pressure also stimulates AVP secretion.
The receptors responsible for this response are located in both the low-pressure (left atrium and large pulmonary vessels) and the high-pressure (aortic arch and carotid sinus) sides of the circulatory system.
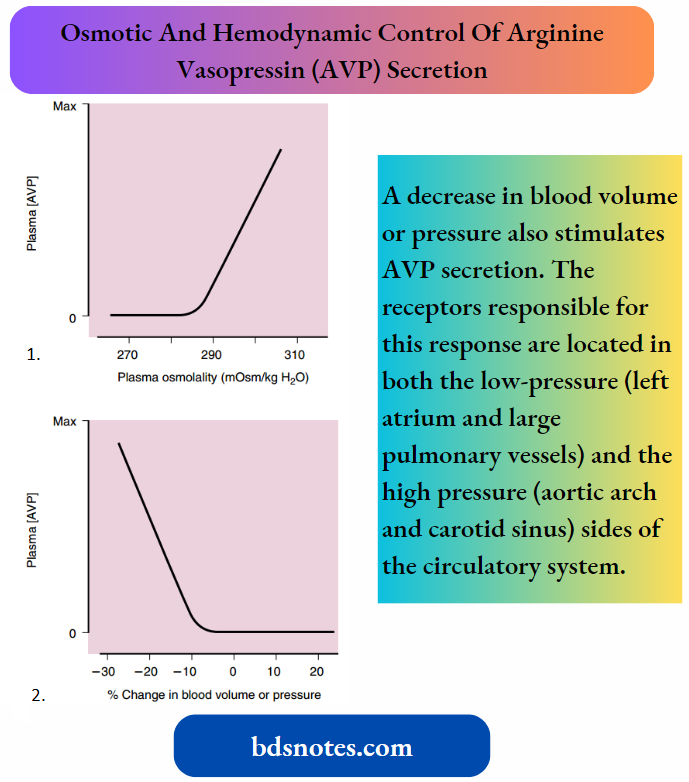
“Comparison of osmolality regulation in health vs disease”
Because the low-pressure receptors are located in the high-compliance side of the circulatory system (i.e., venous) and the majority of blood is in the venous side of the circulatory system, these low-pressure receptors can be viewed as responding to overall vascular volume.
- The high-pressure receptors respond to arterial pressure. Both groups of receptors are sensitive to a stretch of the wall of the structure in which they are located (for example., the cardiac atrial wall and the wall of the aortic arch) and are termed baroreceptors.
- Signals from these receptors are carried in afferent fibers of the vagus and glossopharyngeal nerves to the brainstem (solitary tract nucleus of the medulla oblongata), which is part of the center that regulates heart rate and blood pressure.
- Signals then are relayed from the brainstem to the AVP secretory cells of the supraoptic and paraventricular hypothalamic nuclei.
- The sensitivity of the baroreceptor system is less than that of the osmoreceptors, and a 5% to 10% decrease in blood volume or pressure is required before AVP secretion is stimulated.
This phenomenon is illustrated, in B. Several substances have been shown to alter the secretion of AVP through their effects on blood pressure.
- These substances include bradykinin and histamine, which lower pressure and thus stimulate AVP secretion, and norepinephrine, which increases blood pressure and inhibits AVP secretion.
- Alterations in blood volume and pressure also affect the response to changes in body fluid osmolality.
- With a decrease in blood volume or pressure, the set point is shifted to lower osmolality values and the slope of the relationship is steeper.
- In terms of survival of the individual, this means that when faced with circulatory collapse, the kidneys continue to conserve water, even though by doing so they reduce the osmolality of the body fluids.
- With an increase in blood volume or pressure, the opposite occurs. The set point is shifted to higher osmolality values, and the slope is decreased.
Arginine Vasopressin Actions On The Kidneys:
The primary action of AVP on the kidneys is to increase the permeability of the collecting duct to water.
- In addition, and notably, AVP increases the permeability of the medullary portion of the collecting duct to urea.
- Lastly, AVP stimulates sodium chloride (NaCl) reabsorption by the thick ascending limb of Henle’s loop, the distal tubule, and the cortical portion of the collecting duct.
Inadequate release of arginine vasopressin (AVP) from the posterior pituitary results in the excretion of large volumes of dilute urine (polyuria).
- To compensate for this loss of water, the individual must ingest large volumes of water (polydipsia) to maintain constant body fluid osmolality.
- If the individual is deprived of water, the body fluids become hyperosmotic. This condition is called central diabetes insipidus or pituitary diabetes insipidus.
- Central diabetes insipidus can be inherited, although this situation is rare. It occurs more commonly after head trauma and with brain neoplasms or infections.
“Mechanisms of water reabsorption in the nephron”
Persons with central diabetes insipidus have a urine-concentrating defect that can be corrected by the administration of exogenous AVP.
- The inherited (autosomal dominant) form of central diabetes insipidus is caused by a variety of mutations in the AVP gene.
- In patients with this form of central diabetes insipidus, mutations have been identified in all regions of the AVP gene (i.e., AVP, copeptin, and neurophysin).
- The most common mutation is found in the neurophysin portion of the gene. In each of these situations, defective trafficking of the peptide occurs, with abnormal accumulation in the endoplasmic reticulum.
It is believed that this abnormal accumulation in the endoplasmic reticulum results in the death of the AVP secretory cells of the supraoptic and paraventricular nuclei.
- The syndrome of inappropriate antidiuretic hormone (ADH) secretion (SIADH) is a common clinical problem characterized by plasma AVP levels that are elevated above what would be expected based on body fluid osmolality and blood volume and pressure—hence the term inappropriate ADH secretion.
- In addition, the collecting duct overexpresses water channels, thus augmenting the effect of AVP on the kidney. Persons with SIADH retain water, and their body fluids become progressively hypoosmotic.
In addition, their urine is more hyperosmotic than expected based on the low body fluid osmolality. SIADH can be caused by infections and neoplasms of the brain, drugs (for example., antitumor drugs), pulmonary diseases, and carcinoma of the lung.
- Many of these conditions stimulate AVP secretion by altering neural input to the AVP secretory cells. However, small cell carcinoma of the lung produces and secretes several peptides, including AVP.
- Recently, nonpeptide vasopressin receptor antagonists (for example., conivaptan and tolvaptan) have been developed that can be used to treat SIADH and other conditions in which AVP-dependent water retention by the kidneys occurs (for example., congestive heart failure and hepatic cirrhosis).
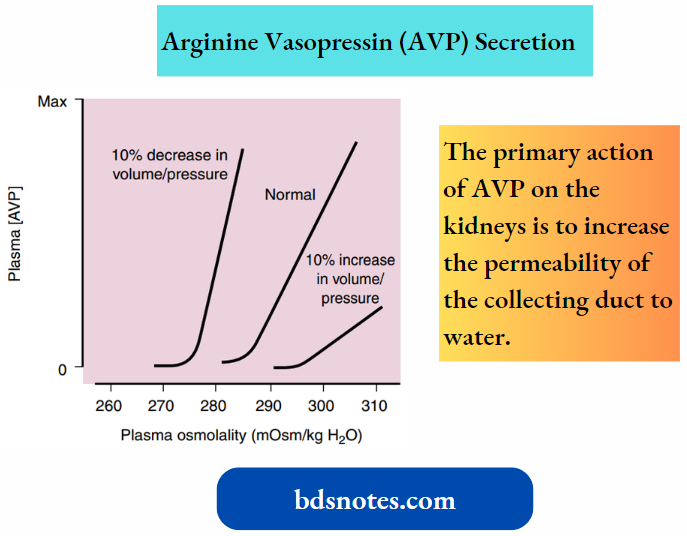
“How do the kidneys maintain osmolality balance?”
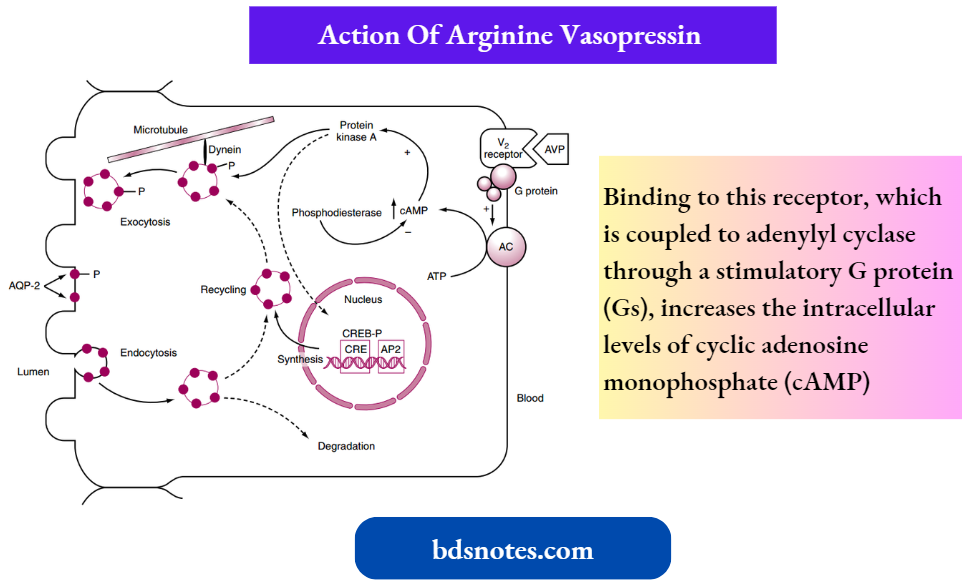
The actions of AVP on the water permeability of the collecting duct have been studied extensively. AVP binds to a receptor on the basolateral membrane of the cell. This receptor is termed the V2 receptor (i.e., vasopressin 2 receptor).
- Binding to this receptor, which is coupled to adenylyl cyclase through a stimulatory G protein (Gs), increases the intracellular levels of cyclic adenosine monophosphate (cAMP).
- The rise in intracellular cAMP activates protein kinase A, which ultimately increases the number of aquaporin (AQP)-2 water channels in the apical membrane of the cell and the synthesis of more AQP-2.
- With the removal of AVP, the number of AQP-2 water channels in the apical membrane is reduced, thereby rendering the membrane impermeable to water.
- Because the basolateral membrane is freely permeable to water because of the presence of AQP-3 and AQP-4 water channels, any water that enters the cell through apical membrane water channels exits across the basolateral membrane, resulting in net absorption of water from the tubule lumen.
“Role of the loop of Henle in concentrating urine”
AVP also increases the permeability of the terminal portion of the inner medullary collecting duct to urea. This increase in permeability results in an increase in urea reabsorption and an increase in the osmolality of the medullary interstitial fluid.
- The inner medullary collecting duct expresses two different urea transporters (UTs: UT-A1 and UT-A3. UT-A1 is found in the apical membrane and UT-A3 is found in the basolateral membrane.
- AVP, acting through adenylyl cyclase and the cAMP/protein kinase A cascade, increases the permeability of the apical membrane to urea.
- This increase in permeability is associated with phosphorylation of UT-A1 and UT-A3. Increasing the osmolality of the interstitial fluid of the renal medulla also increases the permeability of the collecting duct to urea.
- This effect is mediated by the phospholipase C pathway and involves protein kinase C phosphorylation. Thus this effect is separate from and additive to that of AVP.
- AVP also stimulates the reabsorption of NaCl by the thick ascending limb of Henle’s loop and by the distal tubule and cortical segment of the collecting duct.
- It is thought that stimulation of thick ascending limb NaCl transport, in particular, may help maintain the hyperosmotic medullary interstitium that is necessary for the absorption of water from the medullary portion of the collecting duct (discussed later in this chapter).
Arginine Vasopressin At The Cellular Level
The gene for the V2 receptor is located on the X chromosome. It codes for a 371-amino-acid protein that is in the family of receptors that have seven membrane-spanning domains and are coupled to heterotrimeric G proteins.
- As binding of AVP to its receptor on the basolateral membrane activates adenylyl cyclase.
- The increase in intracellular cyclic adenosine monophosphate (cAMP) then activates protein kinase A, which results in phosphorylation of aquaporin (AQP)-2 water channels, which reduces the endocytic removal of AQP-2 from the apical membrane and also results in increased transcription of the AQP-2 gene through activation of a cAMP response element.
- AVP also increases the rate of insertion of vesicles containing AQP-2 into the apical membrane by facilitating their movement along microtubules driven by the molecular motor dynein.
Once near the apical membrane, proteins called SNAREs interact with vesicles containing AQP-2 and facilitate the fusion of these vesicles with the membrane.
- The net addition of AQP-2 to the apical membrane, resulting from reduced endocytosis and increased insertion, allows more water to enter the cell driven by the osmotic gradient (lumen osmolality < cell osmolality).
- The water then exits the cell across the basolateral membrane through AQP-3 and AQP-4 water channels, which are constitutively present in the basolateral membrane.
- When the V+ receptor is not occupied by AVP, clathrin-mediated endocytosis of AQP-2 is enhanced and the exocytic insertion of AQP-2 is reduced, which decreases the total number of AQP-2 channels in the apical membrane, rendering the apical membrane once again impermeable to water.
Recently, persons have been found who have activating (gain-of-function) mutations in the V2 receptor gene. Thus the receptor is constitutively activated even in the absence of AVP.
- These persons have laboratory findings similar to those seen in the syndrome of inappropriate antidiuretic hormone secretion (SIADH), including reduced plasma osmolality, hyponatremia (reduced plasma [Na+]), and urine more concentrated than would be expected from the reduced body fluid osmolality.
- However, unlike persons with SIADH, in whom circulating levels of AVP are elevated and thus responsible for water retention by the kidneys, these persons have undetectable levels of AVP in their plasma. This new clinical entity has been termed “nephrogenic syndrome of inappropriate antidiuresis.”
- The collecting ducts of some persons do not respond normally to arginine vasopressin (AVP). These persons cannot maximally concentrate their urine and consequently have polyuria and polydipsia.
“Impact of glomerular filtration rate (GFR) on water balance”
This clinical entity is termed nephrogenic diabetes insipidus to distinguish it from central diabetes insipidus. Nephrogenic diabetes insipidus can result from several systemic disorders and, more rarely, occurs as a result of inherited disorders.
- Many of the acquired forms of nephrogenic diabetes insipidus are the result of decreased expression of aquaporin-2 (AQP-2) in the collecting duct.
- Decreased expression of AQP-2 has been documented in the urine-concentrating defects associated with hypokalemia, lithium ingestion (some degree of nephrogenic diabetes insipidus develops in 35% of persons who take lithium for bipolar disorder), ureteral obstruction, a low-protein diet, and hypercalcemia. The inherited forms of nephrogenic diabetes insipidus reflect mutations in the AVP receptor (V2 receptor) gene or the AQP-2 gene.
- Approximately 90% of hereditary forms of nephrogenic diabetes insipidus are the result of mutations in the V2 receptor gene, with the other 10% being the result of mutations in the AQP-2 gene.
Because the gene for the V2 receptor is located on the X chromosome, these inherited forms are X-linked. Most of these mutations result in trapping of the receptor in the endoplasmic reticulum of the cell; only a few cases result in the surface expression of a V2 receptor that does not bind AVP.
- The gene coding for AQP-2 is located on chromosome 12 and is inherited as both an autosomal recessive and an autosomal dominant defect. As noted in Chapters 1 and 4, aquaporins exist as homotetramers.
- This homotetramer formation explains the difference between the two forms of nephrogenic diabetes insipidus. In the recessive form, heterozygotes produce both normal AQP-2 and defective AQP-2 molecules.
- The defective AQP-2 monomer is not delivered to the plasma membrane, and thus the homotetramers that do form contain only normal AQP-2 molecules.
- Accordingly, mutations in both alleles would be required to produce nephrogenic diabetes insipidus.
- In the autosomal dominant form, the defective monomers can form tetramers with normal monomers, as well as defective monomers. However, these tetramers cannot be delivered to the plasma membrane.
“How does the renal system respond to dehydration?”
Thirst
In addition to affecting the secretion of AVP, changes in plasma osmolality and blood volume or pressure lead to alterations in the perception of thirst.
- When body fluid osmolality is increased or the blood volume or pressure is reduced, a person perceives thirst. Of these stimuli, hypertonicity is the more potent.
- An increase in plasma osmolality of only 2% to 3% produces a strong desire to drink, whereas decreases in blood volume and pressure in the range of 10% to 15% are required to produce the same response.
- As already discussed, people have a genetically determined threshold for AVP secretion (i.e., a body fluid osmolality above which AVP secretion increases).
- Similarly, people have a genetically determined threshold for triggering the sensation of thirst. However, the thirst threshold is higher than the threshold for AVP secretion.
- On average, the threshold for AVP secretion is approximately 285 mOsm/kg H2O, whereas the thirst threshold is approximately 295 mOsm/kg H2O. Because of this difference, thirst is stimulated at a body fluid osmolality at which AVP secretion is already stimulated.
- The neural centers involved in regulating water intake (the thirst center) are located in the same region of the hypothalamus involved with regulating AVP secretion.
- However, it is not certain if the same cells serve both functions. Indeed, the thirst response, like the regulation of AVP secretion, occurs only in response to effective osmoles (for example., NaCl).
- Even less is known about the pathways involved in the thirst response to decreased blood volume or pressure, but it is believed that the pathways are the same as those involved in the volume- and pressure-related regulation of AVP secretion.
- Angiotensin II, acting on cells of the thirst center (subfornical organ), also evokes the sensation of thirst. Because angiotensin II levels are increased when blood volume and pressure are reduced, this effect of angiotensin II contributes to the homeostatic response that restores and maintains the body fluids at their normal volumes.
- The sensation of thirst is satisfied by the act of drinking even before sufficient water is absorbed from the gastrointestinal tract to correct the plasma osmolality.
- Oropharyngeal and upper gastrointestinal receptors appear to be involved in this response. However, relief of the thirst sensation by these receptors is short-lived, and thirst is completely satisfied only when the plasma osmolality or blood volume or pressure is corrected.
- It should be apparent that the AVP and thirst systems work in concert to maintain water balance. An increase in plasma osmolality evokes drinking and, through AVP action on the kidneys, the conservation of water.
- Conversely, when the plasma osmolality is decreased, thirst is suppressed and, in the absence of AVP, renal water excretion is enhanced.
- However, most of the time fluid intake is dictated by cultural factors and social situations, which is especially the case when thirst is not stimulated. In this situation, maintaining a normal body fluid osmolality relies solely on the ability of the kidneys to excrete water.
- How the kidney accomplishes this task is discussed in detail in the following sections of this chapter.
“Disorders of osmolality and water balance explained”
Renal Mechanisms For Dilution And Concentration Of The Urine
Under normal circumstances, the excretion of water is regulated separately from the excretion of solutes.
- For this separate regulation to occur, the kidneys must be able to excrete urine that is either hypoosmotic or hyperosmotic concerning the body fluids.
- This ability to excrete urine of varying osmolality in turn requires that solute be separated from water at some point along the nephron. As discussed, the reabsorption of solute in the proximal tubule results in the reabsorption of a proportional amount of water.
- Hence solute and water are not separated in this portion of the nephron. Moreover, this proportionality between proximal tubule water and solute reabsorption occurs regardless of whether the kidneys excrete dilute or concentrated urine.
- Thus the proximal tubule reabsorbs a large portion of the filtered solute and water but does not produce dilute or concentrated tubular fluid. The loop of Henle, in particular the thick ascending limb, is the major site where solute and water are separated.
- Thus the excretion of both dilute and concentrated urine requires normal function of the loop of Henle.
With adequate access to water, the thirst mechanism can prevent the development of hyperosmolality. Indeed, it is this mechanism that is responsible for the polydipsia seen in response to the polyuria of both central and nephrogenic diabetes insipidus.
“Role of osmolality regulation in diagnosing diseases”
- Water intake also is influenced by social and cultural factors. Thus persons ingest water even in the absence of the thirst sensation. Normally the kidneys can excrete this excess water because they can excrete up to 18 L/day of urine.
- However, in some instances, the volume of water ingested exceeds the kidneys’ capacity to excrete water, especially over short periods. When this situation occurs, the body fluids become hypoosmotic.
- An example of how water intake can exceed the capacity of the kidneys to excrete water is found in long-distance runners.
- A study of participants in the Boston Marathon found that hyponatremia developed in 13% of the runners during the race.
This finding reflected the practice of some runners of ingesting water, or other hypotonic drinks, during the race to remain “well hydrated.”
- In addition, water is produced from the metabolism of glycogen and triglycerides used as fuels by the exercising muscle.
- Because throughout the race they ingested and generated more water through metabolism than their kidneys were able to excrete, hyponatremia developed. In some racers, the hyponatremia was severe enough to elicit the neurologic symptoms described previously.
- Throughout the popular media, one can find articles urging us to drink eight 8-oz glasses of water a day (the 8 × 8 recommendation). Drinking this volume of water is said to provide innumerable health benefits.
- As a result, it seems that everyone now has a water bottle as his or her constant companion.
- Although ingesting this volume of water over a day (approximately 2 L) does not harm most persons, no scientific evidence exists to support the beneficial health claims ascribed to the 8 x 8 recommendation. t Indeed, most persons get adequate amounts of water through the foods they ingest and the fluids taken with those meals.
- The maximum amount of water that can be excreted by the kidneys depends on the amount of solute excreted, which in turn depends on food intake.
“How does dehydration affect osmolality?”
For example, with maximally dilute urine (urine osmolality [Uosm] = 50 mOsm/kg H2O), the maximum urine output of 18 L/day is achieved only if the solute excretion rate is 900 mmol/day.
Uosm = Solute excretion/Volume excreted 50 mOsm/kg H2O = 900 mmol/18 L
- If solute excretion is reduced, as commonly occurs in elderly people with reduced food intake, the maximum urine output decreases.
- For example, if solute excretion is only 400 mmol/day, a maximum urine output (at Uosm = 50 mOsm/kg H2O) of only 8 L/day can be achieved. Thus persons with reduced food intake have a reduced capacity to excrete water.
The excretion of hypoosmotic urine is relatively easy to understand. The nephron simply must reabsorb solute from the tubular fluid and not allow water reabsorption to occur as well.
- The reabsorption of solute without concomitant water reabsorption occurs in some portions of the descending limb and along the entire ascending limb of Henle’s loop.
- Under appropriate conditions (i.e., in the absence of AVP), the distal tubule and collecting duct also dilute the tubular fluid.
- The excretion of hyperosmotic urine is more complex and thus more difficult to understand. This process in essence involves removing water from the tubular fluid without solute.
“Complications of overhydration and hyponatremia”
Because water movement is passive, driven by an osmotic gradient, the kidney must generate a hyperosmotic compartment that then reabsorbs water osmotically from the tubular fluid.
- The compartment in the kidney where this reabsorption occurs is the interstitial space of the renal medulla.
- It has long been recognized that Henle’s loop is associated with the kidneys’ ability to excrete hyperosmotic urine.
- Indeed, only birds and mammals can excrete hyperosmotic urine, and among vertebrates, only the avian and mammalian kidneys have loops of Henle.
Moreover, some animals, such as desert rodents, have extremely long loops of Henle and excrete urine with an osmolality that can exceed 5000 mOsm/kg H2O.
- This extraordinary ability to concentrate the urine allows the animals to survive without the need to drink water because they obtain sufficient water in the food (for example., seeds) that they ingest.
- For more than 50 years our understanding of how the loop of Henle can generate a hyperosmotic environment within the renal medulla was focused on the process of countercurrent multiplication.
- By this process, solute (principally NaCl) is reabsorbed without water from the ascending limb of Henle’s loop into the surrounding medullary interstitium.
This reabsorption decreases the osmolality in the tubular fluid and raises the osmolality of the interstitium at this point.
- The increased osmolality of the interstitium then causes water to be reabsorbed from the descending limb of Henle’s loop, thus increasing the tubular fluid osmolality in this segment.
- Thus at any point along the loop of Henle, the fluid in the ascending limb has an osmolality less than fluid in the adjacent descending limb. This osmotic difference was termed the single effect.
- Because of the countercurrent flow of tubular fluid in the descending (fluid flowing into the medulla) and ascending (fluid flowing out of the medulla) limbs, this single effect could be multiplied.
“Techniques for monitoring osmolality levels”
The multiplication of this single effect results in an osmotic gradient within the medullary interstitium, where the tip of the papilla has an osmolality of 1200 mOsm/kg H2O, compared with 300 mOsm/kg H2O at the corticomedullary junction.
- Although it is simple in concept, it is now clear that countercurrent multiplication cannot fully explain the process by which the loop of Henle generates a hyperosmotic medullary interstitium.
Given our evolving understanding, specifically of the urine-concentrating mechanism, what follows is a simplified explanation that highlights several key concepts:
- Urine is concentrated by the AVP-dependent reabsorption of water from the collecting duct.
- Reabsorption of NaCl from the ascending limb of Henle’s loop generates a high [NaCl] in the medullary interstitium (up to 600 mmol/L at the tip of the papilla), which then drives water reabsorption from the collecting duct.
- Urea accumulates in the medullary interstitium (up to 600 mmol/L), which allows the kidneys to excrete urine with the same high urea concentration. This phenomenon allows large amounts of urea to be excreted with relatively little water.
Summarizes the essential features of the mechanisms whereby the kidneys excrete either dilute or concentrated urine.
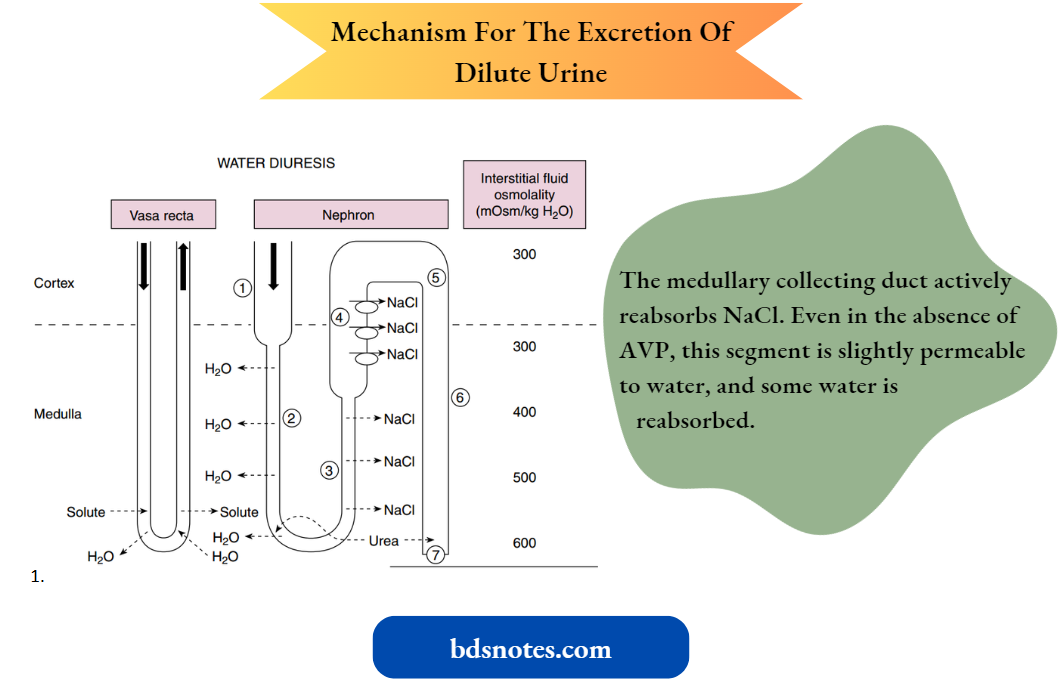
First, how the kidneys excrete dilute urine (water diuresis) when AVP levels are low or zero is considered. The following numbers refer to those encircled.
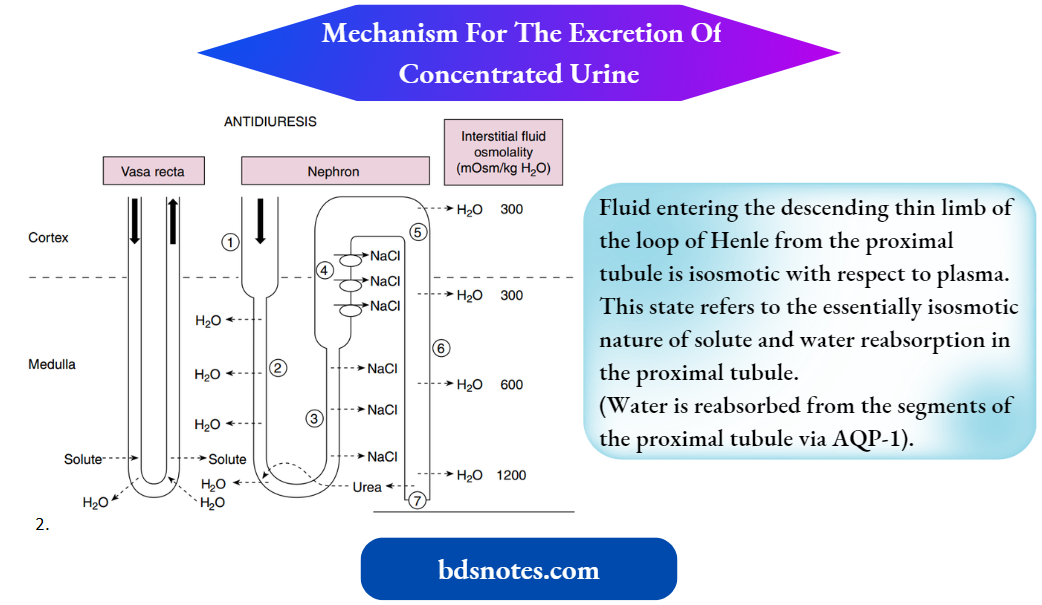
1. Fluid entering the descending thin limb of the loop of Henle from the proximal tubule is isosmotic concerning plasma.
- This state reflects the essentially isosmotic nature of the solute and water reabsorption in the proximal tubule. (Note: Water is reabsorbed from the segments of the proximal tubule via AQP-1).
2. Depending on the nephron type (i.e., short-looped nephrons versus long-looped nephrons, some water will be reabsorbed by the thin descending limb.
- Importantly, this water reabsorption is limited to the outer medulla and the outermost portion of the inner medulla.
- By con-fining water reabsorption to these outer portions of the medulla, less water is added to the deepest part of the inner medullary interstitial space, thus preserving the hyperosmolality of this region of the medulla.
3. In the inner medulla, the terminal portion of the descending thin limb and all of the thin ascending limb are impermeable to water.
- These same nephron segments express the Cl– transporter ClC-K1, which mediates Cl– reabsorption with Na+ following passively via the paracellular pathway.
- This passive reabsorption of NaCl without concomitant water reabsorption begins the process of diluting the tubular fluid.
“Pathophysiology of osmolality imbalances explained”
“Emerging research on osmolality regulation mechanisms”
4. The thick ascending limb of the loop of Henle also is impermeable to water and actively reabsorbs NaCl from the tubular fluid thereby diluting it further.
- Dilution occurs to such a degree that this segment often is referred to as the diluting segment of the kidney.
- Fluid leaving the thick ascending limb is hypoosmotic concerning plasma (approximately 150 mOsm/kg H2O).
5. The distal tubule and cortical portion of the collecting duct actively reabsorb NaCl. In the absence of AVP, these segments are not permeable to water (i.e., AQP-2 is not present in the apical membrane of the cells).
- Thus when AVP is absent or present at low levels (i.e., decreased plasma osmolality), the osmolality of tubule fluid in these segments is reduced further because NaCl is reabsorbed without water.
- Under this condition, fluid leaving the cortical portion of the collecting duct is hypo-motif concerning plasma (approximately 50-100 mOsm/kg H2O).
6. The medullary collecting duct actively reabsorbs NaCl. Even in the absence of AVP, this segment is slightly permeable to water, and some water is reabsorbed.
7. The urine has an osmolality as low as approximately 50 mOsm/kg H2O and contains low concentrations of NaCl. The volume of urine excreted can be as much as 18 L/day or approximately 10% of the glomerular filtration rate (GFR).
- Next, how the kidneys excrete concentrated urine (antidiuresis) when plasma osmolality and plasma AVP levels are high is considered. The following numbers refer to those encircled.
- These steps are similar to those for the production of dilute urine. An important point in understanding how concentrated urine is produced is to recognize that while reabsorption of NaCl by the ascending thin and thick limbs of the loop of Henle dilutes the tubular fluid, the reabsorbed NaCl accumulates in the medullary interstitium and raises the osmolality of this compartment.
- The accumulation of NaCl in the medullary interstitium is crucial for the production of urine hyperosmotic to plasma because it provides the osmotic driving force for water reabsorption by the medullary collecting duct.
- As already noted, AVP stimulates NaCl reabsorption by the thick ascending limb of Henle’s loop.
- This action is thought to maintain the medullary interstitial gradient at a time when water is being added to this compartment from the medullary collecting duct, which would tend to dissipate the gradient.
“Case studies on osmolality and water balance outcomes”
Because of NaCl reabsorption by the ascending limb of the loop of Henle, the fluid reaching the collecting duct is hypoosmotic concerning the surrounding interstitial fluid.
Thus an osmotic gradient is established across the collecting duct.
- In the presence of AVP, which increases the water permeability of the last half of the distal tubule and the collecting duct by increasing the number of AQP-2 water channels in the luminal membrane of the cells, water diffuses out of the tubule lumen, and the tubule fluid osmolality increases.
- This diffusion of water out of the lumen of the collecting duct begins the process of urine concentration.
The maximum osmolality that the fluid in the distal tubule and cortical portion of the collecting duct can attain is approximately 290 mOsm/kg H2O (i.e., the same as plasma), which is the osmolality of the interstitial fluid and plasma within the cortex of the kidney.
- As the tubular fluid descends deeper into the medulla, water continues to be reabsorbed from the collecting duct, increasing the tubular fluid osmolality to 1200 mOsm/kg H2O at the tip of the papilla.
- The urine produced when AVP levels are elevated has an osmolality of 1200 mOsm/kg H2O and contains high concentrations of urea and other non-absorbed solutes. The urine volume under this condition can be as low as 0.5 L/day.
- Under most conditions, a relatively constant volume of tubular fluid is delivered to the AVP-sensitive portions of the nephron (late distal tubule and collecting duct).
Plasma AVP levels then determine the amount of water that is reabsorbed by these segments. When AVP levels are low, a relatively small volume of water is reabsorbed by these segments, and a large volume of hypoosmotic urine is excreted (up to 10% of the filtered water).
- When AVP levels are high, a large volume of water is reabsorbed by these same segments, and a small volume of hyperosmotic urine is excreted (<1% of filtered water).
- During antidiuresis, most of the water is reabsorbed in the distal tubule and cortical and outer medullary portions of the collecting duct.
- Thus a relatively small volume of fluid reaches the inner medullary collecting duct, where it is then reabsorbed.
This distribution of water reabsorption along the length of the collecting duct (i.e., cortex > outer medulla > inner medulla) allows for the maintenance of a hyperosmotic interstitial environment in the inner medulla by minimizing the amount of water entering this compartment.
Renal Mechanisms At The Cellular Level:
Water movement across the various segments of the nephron occurs through water channels.
- The proximal tubule and portions of some thin descending limbs of Henle’s loop are highly permeable to water, and these segments express high levels of AQP-1 in both the apical and basolateral membranes.
- The vasa recta also are highly permeable to water and express AQP-1. AQP-7 and AQP-8 also are expressed in the proximal tubule.
- As already discussed, AQP-2 is responsible for arginine vasopressin (AVP)-regulated water movement across the apical membrane of principal cells of the late distal tubule and collecting duct, and AQP-3 and AQP-4 are responsible for water movement across the basolateral membrane.
- Mice lacking the AQP-1 gene have been created. These mice have a urine-concentrating defect with increased urine output. Several persons have been found who also lack the normal AQP-1 gene.
- Interestingly, these persons do not have polyuria. However, when challenged by water deprivation, they can concentrate their urine to only approximately half of what is seen in a healthy person.
“Global prevalence of osmolality disorders”
Role Of Urea
As noted, a hyperosmotic renal medullary interstitium is critically important in concentrating the urine and provides the driving force for the reabsorption of water from the collecting duct.
- The principal solutes within the renal medullary interstitium are NaCl and urea, but the concentration of these solutes is not uniform throughout the medulla (i.e., a gradient exists from cortex to papilla).
- Other solutes also accumulate in the medulla (for example., ammonium [NH+4] and K+), but the most abundant solutes are NaCl and urea. For simplicity, this discussion assumes that NaCl and urea are the only solutes.
- At the junction of the medulla with the cortex, the interstitial fluid has an osmolality of approximately 300 mOsm/kg H2O, with virtually all osmoles attributable to NaCl. The concentrations of both NaCl and urea increase progressively with increasing depth into the medulla.
When maximally concentrated urine is excreted, the medullary interstitial fluid osmolality is approximately 1200 mOsm/kg H2O at the papilla.
- Of this value, approximately 600 mOsm/ kg H2O is attributed to NaCl, and 600 mOsm/kg H2O is attributed to urea. As described later, NaCl is an effective osmole in the inner medulla and thus is responsible for driving water reabsorption from the medullary collecting duct. T
- The high urea concentration of the medullary interstitial fluid allows this solute to be excreted at a high concentration (600 mmol/L) in a small volume of urine, thus limiting the amount of water that otherwise would be needed to excrete the daily load of bread
- The medullary gradient for NaCl results from the accumulation of NaCl reabsorbed by the segments of Henle’s loop (see the previous discussion).
- Urea accumulation within the medullary interstitium is more complex and occurs most effectively when hyperosmotic urine is excreted (i.e., antidiuresis). When dilute urine is produced, especially over extended periods, the osmolality of the medullary interstitium declines.
- This reduced osmolality is almost entirely caused by a decrease in the concentration of urea. This decrease reflects washout by the vasa recta (discussed in a later section of this chapter) and diffusion of urea from the interstitium into the tubular fluid within the medullary portion of the collecting duct.
(Note: The cortical and outer medullary portions of the collecting have a low permeability to urea, whereas the inner medullary portion has a relatively high permeability because of the presence.)
- Urea is not synthesized in the kidney but is generated by the liver as a product of protein metabolism. It enters the tubular fluid via glomerular filtration.
- Approximately half of this filtered urea is reabsorbed by the proximal tubule. During antidiuresis, water reabsorption by the cortical and outer medullary portions of the collecting duct leads to an increase in the urea concentration of the tubular fluid.
- When this fluid reaches the portion of the inner medullary collecting duct that expresses UT-A1 and UT-A3, urea is reabsorbed.
- The reabsorption of urea is further enhanced by the high levels of AVP, which increase the expression of the UTs. Some of this reabsorbed urea is secreted into thin descending limbs of Henle’s loops via UT-A2, and some enter vasa recta via UT-B.
The urea that is secreted into the descending thin limbs of Henle’s loops is then trapped in the nephron until it again reaches the medullary collecting duct, where it can reenter the medullary interstitium.
- Thus urea recycles from the interstitium to the nephron and back into the interstitium. This process of recycling facilitates the accumulation of urea in the medullary interstitium, where it can attain a concentration at the tip of the papilla of 600 mmol/L.
- It is the high concentration of urea in the interstitial fluid that prevents the diffusion of urea out of the lumen of the inner medullary collecting duct into the interstitium, thereby facilitating urea excretion in the urine.
- As described, the hyperosmotic medulla is essential for concentrating the tubular fluid within the collecting duct.
- Because water reabsorption from the collecting duct is driven by the osmotic gradient established in the medullary interstitium, urine can never be more concentrated than that of the interstitial fluid in the papilla.
- Thus any condition that reduces the medullary interstitial osmolality impairs the ability of the kidneys to maximally concentrate the urine.
However, because the inner medullary collecting duct is highly permeable to urea, especially in the presence of AVP, urea cannot drive water reabsorption across this nephron segment (i.e., urea is an ineffective osmole).
- Instead, the urea in the tubular fluid and medullary interstitium equilibrate and a small volume of urine with a high concentration of urea is excreted.
- It is the medullary interstitial NaCl concentration that is responsible for reabsorbing water from the inner medullary collecting duct and thereby concentrating the nonurea solutes (for example., NH+ salts, K+ salts, and creatinine) in the urine.
At The Cellular Level
The expression of the urea transporter (UTA1) in the inner medullary collecting duct is increased by arginine vasopressin (AVP) via a cyclic adenosine monophosphate–mediated mechanism. UTA1 expression also is increased by hyperosmolality.
- This effect is mediated by changes in intracellular Ca++ and protein kinase C activity. Thus the effects of AVP and hyperosmolality are separate and additive.
- The expression of UTA3 and UTA2 also is increased by AVP. Knockout mice have been created that lack the UTA1/UTA3 collecting duct transporters, the UTA2 thin descending limb transporter, or the UTB vasa recta transporter.
- All of these animals have some degree of impairment of urinary concentration. Humans with genetic loss of UTB exhibit a similar urinary concentrating defect as the knockout mouse model.
Vasa Recta Function
The vasa recta, the capillary networks that supply blood to the medulla, are highly permeable to solute and water. As with the loop of Henle, the vasa recta form a parallel set of hairpin loops within the medulla.
- Not only do the vasa recta bring nutrients and oxygen to the medullary nephron segments but, more importantly, they also remove the water and solute that is continuously added to the medullary interstitium by these nephron segments.
- The ability of the vasa recta to maintain the medullary interstitial gradient is flow-dependent. A substantial increase in vasa recta blood flow dissipates the medullary gradient. Alternatively, decreased blood flow reduces oxygen delivery to the nephron segments within the medulla.
- Because the transport of salt and other solutes requires oxygen and adenosine triphosphate, reduced medullary blood flow decreases salt and solute transport by nephron segments in the medulla. As a result, the medullary interstitial osmotic gradient cannot be maintained, which also reduces the ability to concentrate the urine.
Assessment Of Renal Diluting And Concentrating Ability
Assessment of renal water handling includes measurements of urine osmolality and the volume of urine excreted. The range of urine osmolality is from 50 to 1200 mOsm/kg H2O.
- The corresponding range in urine volume is 18 to as little as 0.5 L/day. These ranges are not fixed, but they vary from person to person and, as noted previously, depend on the amount of water ingested and lost from nonrenal routes, as well as the amount of solute excreted.
- As emphasized in this chapter, the ability of the kidneys to dilute or concentrate the urine requires the separation of solute and water.
- This separation of solute and water in essence generates a volume of water that is “free of solute.” When the urine is dilute, solute-free water is excreted from the body.
- When the urine is concentrated, solute-free water is returned to the body (i.e., conserved).
- The concept of free water clearance (CH2O) provides a way to calculate the amount of solute-free water generated by the kidneys, either when dilute urine is excreted or when concentrated urine is formed.
- As its name denotes, CH2O is directly derived from the concept of renal clearance discussed.
“Complications of ignoring osmolality issues”
To calculate CH2O, the clearance of total solute by the kidneys must be calculated. This clearance of total solute (i.e., osmoles, whether effective or ineffective) from plasma by the kidneys is termed the osmolar clearance (Cosm) and can be calculated as follows:
⇒ \(\mathrm{C}_{\mathrm{OSM}}=\frac{\mathrm{U}_{\mathrm{OSM}} \times \dot{\mathrm{V}}}{\mathrm{P}_{\mathrm{OSM}}}[latex]
where Uosm is the urine osmolality, V is the urine flow rate, and Posm is the osmolality of plasma. Cosm has units of volume/unit time. CH2O is then calculated as follows:
⇒ [latex]\mathrm{C}_{\mathrm{H}_2 \mathrm{O}}=\dot{\mathrm{V}}-\mathrm{C}_{\mathrm{osm}}\)
By rearranging equations 5-3; it should be apparent that
⇒ \(\dot{\mathrm{V}}=\mathrm{C}_{\mathrm{H}_2 \mathrm{O}}+\mathrm{C}_{\mathrm{osm}}\)
In other words, it is possible to partition the total urine output (V) into two hypothetical components. One component contains all the urine solutes and has an osmolality equal to that of plasma (i.e., Uosm = Posm).
- This volume is defined by Cosm and represents a volume from which there has been no separation of solute and water. The second component is a volume of solute-free water (i.e., CH2O).
- When dilute urine is produced, the value of CH2O is positive, indicating that solute-free water is excreted from the body. When concentrated urine is produced, the value of CH2O is negative, indicating that solute-free water is retained in the body.
- By convention, negative CH2O values are expressed as TCH2O (tubular conservation of water).
- Calculating CH2O and TCH2O can provide important information about the function of the portions of the nephron involved in producing dilute and concentrated urine.
Whether the kidneys excrete or reabsorb free water depends on the presence of AVP. When AVP is absent or AVP levels are low, solute-free water is excreted. When AVP levels are high, solute-free water is reabsorbed.
The following factors are necessary for the kidneys to excrete a maximal amount of solute-free water (CH2O):
- AVP must be absent. Without AVP, the collecting duct does not reabsorb a significant amount of water.
- The tubular structures that separate solute from water (i.e., dilute the luminal fluid) must function normally. In the absence of AVP, the following nephron segments can dilute the luminal fluid:
- Thin ascending limb of Henle’s loop
- Thick ascending limb of Henle’s loop
- Distal tubule
- Collecting duct
- Because of its high transport rate, the thick ascending limb is quantitatively the most important of these segments involved in the separation of solute and water.
- An adequate amount of tubular fluid must be delivered to the aforementioned nephron sites for maximal separation of solute and water.
- Factors that reduce delivery (for example., decreased GFR or enhanced proximal tubule reabsorption) impair the kidneys’ ability to excrete solute-free water.
Similar requirements also apply to the conservation of water by the kidneys (TCH2O). For the kidneys to conserve water maximally, the following conditions must exist:
- An adequate amount of tubular fluid must be delivered to the nephron segments in which separation of solute from water occurs. The important segment in the separation of solute and water is the thick ascending limb of Henle’s loop. Delivery of tubular fluid to Henle’s loop depends on GFR and proximal tubule reabsorption.
- Reabsorption of NaCl by the nephron segments must be normal; again, the most important segment is the thick ascending limb of Henle’s loop.
- A hyperosmotic medullary interstitium must be present. The interstitial fluid osmolality is maintained by NaCl reabsorption by Henle’s loop (con-ditions 1 and 2) and by effective accumulation of urea. Urea accumulation in turn depends on ade-quate dietary protein intake.
- Maximum levels of AVP must be present and the collecting duct must respond normally to AVP.
The concept of free-water clearance as just described does not distinguish between effective and ineffective osmoles, either in the plasma or in the urine.
However, urea, which can account for half of the total urine osmoles, is not an effective osmole when the movement of water between intracellular fluid and extracellular fluid is considered.
Accordingly, when one wants to understand how the handling of water by the kidneys contributes to the maintenance of whole-body water balance, it is more appropriate to consider only the solutes that are effective osmoles. For plasma (i.e., extracellular fluid), the effective osmoles are Na+ and its attendant anions. For urine, they are the nonurea solutes.
The importance of using effective osmoles in determining the impact of renal water handling on whole-body water balance (i.e., body fluid osmolality) is illustrated by the following example.
A patient has an elevated plasma [urea], and his plasma [Na+] also is increased to 150 mEq/L. His total plasma osmolality (including urea) is 320 mOsm/kg H2O, but his effective plasma osmolality (calculated as 2 x plasma [Na+]) is only 300 mOsm/kg H2O.
His urine osmolality is 600 mOsm/kg H2O, with 300 mOsm/ kg H2O related to urea and 300 mOsm/kg H2O related to nonurea solutes. His urinary flow rate is 3 L/day.
According to equations 5-2 and 5-3, his total osmolar clearance (Cosm) and free-water clearance (CH2O) are as follows:
⇒ \(\mathrm{C}_{\mathrm{oSM}}=\frac{600 \mathrm{mOsm} / \mathrm{kg} \mathrm{H}_2 \mathrm{O} \times 3 \mathrm{~L} / \text { day }}{320 \mathrm{mOsm} / \mathrm{kg} \mathrm{H}_2 \mathrm{O}}=5.6 \mathrm{~L} / \text { day }\)
⇒ \(\mathrm{C}_{\mathrm{H}_2 \mathrm{O}}=3 \mathrm{~L} / \text { day }-5.6 \mathrm{~L} / \text { day }=-2.6 \mathrm{~L} / \text { day }\left(\mathrm{T}_{\mathrm{H}_2 \mathrm{O}}^{\mathrm{C}}\right)\)
Thus it appears that the kidneys are conserving 2.6 L/day of solute-free water, which would be an appropriate response to correct the elevated plasma osmolality.
However, when Cosm and CH2O are analyzed from the perspective of effective osmoles, the following results are obtained:
⇒ \(\text { Cosm }=\frac{300 \mathrm{mOsm} / \mathrm{kg} \mathrm{H}_2 \mathrm{O} \times 3 \mathrm{~L} / \text { day }}{300 \mathrm{mOsm} / \mathrm{kg} \mathrm{H}_2 \mathrm{O}}=3 \mathrm{~L} / \text { day }\)
⇒ \(\mathrm{C}_{\mathrm{H}_2 \mathrm{O}}=3 \mathrm{~L} / \text { day }-3 \mathrm{~L} / \text { day }=0 \mathrm{~L} / \text { day }\)
When viewed from the more appropriate perspective of effective osmoles, it thus is apparent that the kidneys are not reabsorbing solute-free water and the patient’s kidneys are not correcting the hyperosmolality.
Leave a Reply