Physiology Of Diuretic Action
“What is the physiology of diuretic action?”
Diuretics, as the name implies, are drugs that cause an increase in urine output. It is important, however, to distinguish this diuresis from that which occurs after the ingestion of large volumes of water. In the latter case, the urine is primarily made up of water, and solute excretion is not increased.
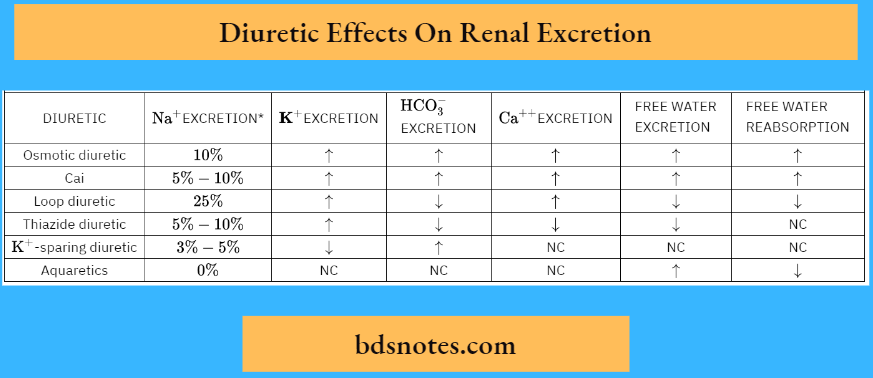
- In contrast, diuretics result in the enhanced excretion of both solute and water. All diuretics (except aquaretics, which will be discussed) have as their common mode of action the primary inhibition of Na+ reabsorption by the nephron.
- Consequently, they cause an increase in the excretion of Na+, termed natriuresis. However, the effects of diuretics are not limited to Na+ handling.
- The renal handling of many other solutes also is influenced, usually as a consequence of alterations in Na+ transport. Recently, drugs have been developed that block the action of arginine vasopressin (AVP) on the distal tubule and collecting duct.
- These drugs, called aquaretics, cause water diuresis. This chapter reviews the cellular mechanisms of action of various diuretics and the nephron sites at which these diuretics act.
In addition to their effects on Na+ handling by the nephron, their effects on the renal handling of other solutes
“Understanding how diuretics work in the body”
Example:
K+, Ca++, inorganic phosphate [Pi], and bicarbonate [HCO3–]) and of water are considered. The effects of aquaretics on water excretion also are discussed.
General Principles Of Diuretic Action
“How do diuretics affect kidney function?”
The primary action of diuretics is to increase the excretion of Na+.
- Alterations in Na+ excretion by the kidneys result in alterations in the volume of the extracellular fluid (ECF) compartment. Consequently, diuretics decrease the volume ofthe ECF.
- Indeed, diuretics commonly are given in clinical situations when the ECF compartment is expanded, with the intent of reducing its volume.
- Because the ECF volume also determines blood volume and pressure, diuretics commonly are used in the therapy of hypertension.
- Although generally predictable for a particular class of diuretics, the effects of diuretic administration can be quite variable.
Several factors are important in determining the overall effect of a particular diuretic:
- The nephron segment where the diuretic acts
- The response of nephron segments not directly affected by the diuretic
- The delivery of sufficient quantities of the diuretic to its site of action
- The volume of the ECF
“Importance of studying diuretic action in medicine”
Sites of Action of Diuretics
Depicts the nephron sites at which the different classes of diuretics act. The osmotic diuretics act along the proximal tubule and portions of the thin descending limb of Henle’s loop (i.e., those portions of the nephron that have a high water permeability).
- The early portion of the distal tubule is the site of action of the thiazide diuretics, and the K+– sparing diuretics act primarily on the late portion of the distal tubule and theoretical portion of the collecting duct where they not only inhibit Na+ reabsorption but also K+ secretion.
- This same class of diuretics also can inhibit Na+ reabsorption in portions of the collecting duct that do not secrete K+.
- The site of action of a diuretic in turn determines the magnitude of the associated natriuresis.
- For example, diuretics acting on the thick ascending limb of Henle’s loop cause a larger diuresis than diuretics acting on the early portion of the distal tubule, because a larger portion of the filtered Na+ is absorbed by the thick ascending limb.
The effect diuretics have on the handling of solutes other than Na+ also depends on the site of action. Examples illustrating this point are given in subsequent sections.
“Common types of diuretics explained”

“Role of loop diuretics in renal physiology”
Response of Other Nephron Segments
When a diuretic inhibits Na+ reabsorption at one nephron site, it causes increased delivery of Na+ and water to more distal segments. The function of these more distal segments and their ability or inability to handle this increased load ultimately determine the overall effect of the diuretic on urinary solute and water excretion.
Examples of this phenomenon are considered in detail with a discussion of each of the various diuretics. In addition, diuretic-induced changes in ECF volume (discussed later in this chapter) may modulate Na+ transport in segments of the nephron not directly affected by the diuretic and thereby influence the degree of natriuresis.
“How thiazide diuretics reduce blood pressure”
Adequate Delivery of Diuretics to Their Site of Action
The effect of a diuretic on Na+ excretion also depends on the delivery of adequate quantities of the drug to its site of action. Except for the aldosterone antagonists, which act intracellularly, diuretics act from the lumen of the nephron (carbonic anhydrase inhibitors have both luminal and intracellular sites of action).
Diuretics gain access to the lumen by glomerular filtration and through secretion by the organic anion and organic cation secretory systems located in the proximal tubule
Because some diuretics are bound to plasma proteins
Example: Loop diuretics
Loop diuretics secretion by the proximal tubule is the primary mechanism for the delivery of the diuretic to its site of action in the lumen of the nephron. Thus the effect of a diuretic can be blunted if, for example, it is administered with another drug that competes for the same organic anion and organic cation secretory mechanism.
The volume of the Extracellular Fluid
The effect of a diuretic also depends on the volume of the ECF.
- When the volume of the ECF is decreased, the glomerular filtration rate (GFR) is reduced, thereby reducing the amount of filtered Na+.
- In addition, Na+ reabsorption by the nephron is enhanced.
- Thus the effect of a diuretic that acts on the distal tubule would be blunted if administered in the setting of a reduced ECF volume.
Under this condition, the decreased GFR (i.e., decreased filtered Na+), together with enhanced Na+reabsorption by the proximal tubule, would result in the delivery of a smaller quantity of Na+ to the distal tubule. Thus even if the diuretic completely inhibited Na+ reabsorption in the distal tubule, the associated natriuresis would be less than would occur if the ECF volume were normal.
“Mechanism of potassium-sparing diuretics explained”
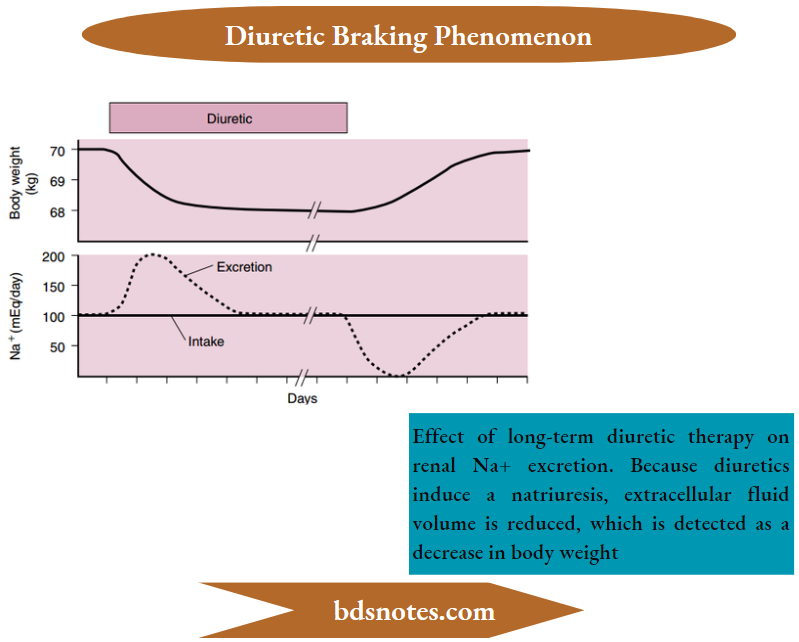
Diuretic Braking Phenomenon
As illustrated, administration of a diuretic to a person with fied Na+ intake results in a short-lived natriuresis.
- The transient response, called the diuretic braking phenomenon, reflects several changes in renal function that are both a direct effect of the diuretic and secondary to changes in the volume of the ECF.
- One component of this response is that diuretic-induced inhibition of Na+ reabsorption in the targeted nephron segment increases Na+ delivery to more distal nephron segments and simulates Na+ reabsorption at these sites.
- A second important component of this response is that the loss of sodium chloride (NaCl) and water from the body as a result of diuretic action decreases the volume of the ECF. This decrease in turn is sensed by the body’s vascular baroreceptors and effector mechanisms, prompting them to increase NaCl and water conservation by the kidneys
“Impact of carbonic anhydrase inhibitors on diuresis”
Studies in experimental animals have shown that loop and thiazide diuretics increase the expression of the transporters they inhibit.
For example, loop diuretics, which inhibit the Na+-K+-2Cl– cotransporter (NKCC2) in the apical membrane of the cells of the thick ascending limb of Henle’s loop, also increase the expression of the transporter in this segment.
- Similarly, thiazide diuretics, which inhibit the Na+-Cl– − symporter (NCC) in the apical membrane of the early portion of the distal tubule, also increase the expression of these transporters in this segment.
- In addition, both loop and thiazide diuretics increase the expression of the Na+ channel (epithelial sodium channel [ENaC]) in the late portion of the distal tubule and collecting duct.
- Thiazide diuretics also increase the expression of aquaporin-2 in the principal cells of the late portion of the distal tubule and the collecting duct. Whether this effect is direct or indirect (i.e., related to changes in ECF volume) is uncertain.
- Regardless of the mechanism, the increased expression of aquaporin-2 is expected to increase water reabsorption by the arginine vasopressin (AVP)- sensitive nephron segments.
- With the diuretic-induced decrease in the ECF volume, the renin-angiotensin in-aldosterone system is activated, renal sympathetic nerve activity is increased, and
- AVP secretion is stimulated, which in combination acts to reduce urinary sodium chloride (NaCl) and water excretion.
- At the cellular level, angiotensin II increases the expression of the Na+-Cl– symporter in the early portion of the distal tubule, and aldosterone increases the expression of ENaC in the late distal tubule and collecting duct.
- AVP also increases the expression of key transporters. Specifically, AVP increases the expression of NKCC2 in the thick ascending limb of Henle’s loop, NCC in the early portion of the distal tubule, and ENaC in the late distal tubule and collecting duct.
Finally, angiotensin II increases the abundance of the Na+-H+ antiporter (NHE3) in the proximal tubule. Upregulation of these transporters by diuretics reduces their efficacy, and when the diuretic administration is terminated, results in a rapid increase in NaCl reabsorption.
“Osmotic diuretics: Uses and mechanism of action”
- As a result of the braking phenomenon, a new steady state is reached where even with continued administration of the diuretic, urinary Na+ excretion once again equals intake.
- However, this steady state occurs at a reduced ECF volume, which is detected as a decrease in body weight. When diuretic therapy is discontinued, renal Na+ excretion is reduced.
- After a period of positive Na+ balance, during which the ECF returns to normal (i.e., return of body weight to its original value), a new steady state is again achieved.
- The concept of a steady state deserves special emphasis. Normally, persons are in a steady-state balance between solute (Example: Na+) and water, with intake equaling excretion.
- Administration of a diuretic temporarily disrupts this balance by increasing solute and water excretion, and a negative balance exists. However, solute and water excretion cannot exceed intake indefinitely, and a new steady state eventually is achieved.
- In this new steady state, intake, and excretion are again balanced, but the ECF volume is reduced as a result of the diuretic-induced excretion of NaCl and water.
- In general, when a person has been taking a diuretic for several days or longer, a new steady state is achieved.
- If Na+ intake is not increased, the ECF volume is decreased in proportion to the degree of negative Na+ balance.
Mechanisms of action of diuretics
1. Osmotic Diuretics
Osmotic diuretics, as the name implies, are agents that inhibit the reabsorption of solute and water by altering osmotic driving forces along the nephron.
- Unlike the other classes of diuretics, osmotic diuretics do not inhibit a specific membrane transport protein; they simply affect water transport across the cells of the nephron through the generation of an osmotic pressure gradient.
- The best example of an exogenous osmotic diuretic is the sugar mannitol.
- When present in abnormally high concentrations, freely filtered endogenous substances such as glucose (i.e., in patients with diabetes mellitus) and urea (i.e., in patients with renal disease whose plasma urea levels are elevated) also can act as osmotic diuretics.
- Osmotic diuretics (For example:, mannitol) gain access to the proximal tubular fluid by glomerular filtration.
- Because they are not reabsorbed or are only poorly reabsorbed, they remain within the tubular lumen, where they can exert an osmotic pressure that inhibits tubular fluid reabsorption.
- Osmotic diuretics affect fluid reabsorption in the segments that have high permeability to water (i.e., the proximal tubule and portions of the thin descending limb of Henle’s loop).
- Because of the large volumes of filtrate reabsorbed in the proximal tubule (60% to 70% of the filtrate), this nephron site is most important when considering the action of osmotic diuretics.
“How do diuretics influence the nephron?”
The reabsorption of the tubular flid by the proximal tubule is essentially an isosmotic process (i.e., the osmolality of the reabsorbed fluid is only slightly hyperosmotic compared with that of the tubular flid).
- Solute (primarily NaCl) is actively reabsorbed by the proximal tubule cells.
- This reabsorption sets up a small osmotic pressure difference across the tubule, with the tubular fluid being 3 to 5 mOsm/kg H2O hypoosmotic concerning the interstitial fluid.
- Given the fact that water is readily able to cross the proximal tubule, this small osmotic pressure gradient is sufficient to cause water reabsorption.
- Also, as water flows from the lumen to the interstitium, it brings additional solute with it by solvent drag.
- When an osmotic diuretic is present in the tubular flid, its concentration increases progressively as a result of NaCl and water reabsorption by the nephron. With this increase in concentration, an osmotic gradient develops opposite to the normal gradient generated by NaCl reabsorption.
- As a result, both NaCl (solvent drag component) and water reabsorption are reduced. With osmotic diuresis, an increase in blood flow to the medulla of the kidney also occurs.
- This increase in blood flw dissipates the standing interstitial osmotic gradient and thus also impairs water reabsorption by the descending limb of Henle’s loop and the medullary collecting duct.
- Some of the Na+ that is not reabsorbed by the proximal tubule is reabsorbed downstream by the thick ascending limb, distal tubule, and collecting duct.
- Thus the degree of natriuresis seen with osmotic diuretics is less than expected based on the magnitude of proximal tubule reabsorption.
Although Na+ excretion rates are as high as 60% of the filtered Na+ have been seen in experimental situations, the usual natriuresis seen in persons treated with osmotic diuretics is only about 10% of the filtered Na+.
“Role of the loop of Henle in diuretic action”
2. Carbonic Anhydrase Inhibitors
Carbonic anhydrase inhibitors
Example: Acetazolamide) reduce Na+ reabsorption by their effect on carbonic anhydrase.
This enzyme is abundant in the proximal tubule and therefore represents the major site of action of these diuretics. Carbonic anhydrase also is present in other cells along the nephron
Example:
Thick ascending limb of Henle’s loop and intercalated cells of the collecting duct), and administration of carbonic anhydrase inhibitors affect the activity of the enzyme at these sites as well.
- However, the effects of these diuretics are almost entirely attributed to their inhibition of the enzyme in the proximal tubule.
- This phenomenon reflects the fact that approximately one-third of proximal tubule Na+ reabsorption occurs in exchange for H+ (through the Na+-H+ antiporter) and thus depends on the activity of carbonic anhydrase
- Even though one-third of proximal tubule Na+ reabsorption is coupled to the secretion of H+, inhibition of this process by the carbonic anhydrase inhibitors does not result in a large natriuresis for several reasons.
- First, even with complete inhibition of carbonic anhydrase, some Na+ reabsorption (linked to bicarbonate reabsorption) still occurs. Second, downstream nephron segments increase their reabsorption of Na+
“Impact of diuretics on glomerular filtration rate (GFR)”
Example:
The thick ascending limb, distal tubule, and collecting duct), and third, increased delivery of Na+ to the macula densa leads to a reduction in the GFR by the tubuloglomerular feedback mechanism.
- Finally, with long-term administration, metabolic acidosis develops, which further decreases the effect of carbonic anhydrase inhibitors by reducing the filtration of HCO3– (i.e., the percentage of Na+reabsorbed with HCO3– in the proximal tubule is reduced).
- Typically, administration of carbonic anhydrase inhibitors results in Na+ excretion rates that are 5% to 10% of the filtered Na+.
3. Loop Diuretics
Loop diuretics
Example:
Furosemide, bumetanide, torsemide, and ethacrynic acid) are organic anions that enter the tubular lumen primarily through secretion by the organic anion secretory system of the proximal tubule.
- They directly inhibit Na+ reabsorption by the thick ascending limb of Henle’s loop by blocking the Na+-K+-2Cl– symporter located in the apical membrane of these cells.
- By this action, they not only inhibit Na+ reabsorption but also disrupt the ability of the kidneys to dilute and concentrate the urine. Dilution is impaired because the solute
- (NaCl) reabsorption by the water-impermeable thick ascending limb of Henle’s loop is inhibited.
- NaCl reabsorption by the medullary portion of the thick ascending limb also is critical for the generation and maintenance of an elevated medullary interstitial fluid osmolality.
- Therefore inhibition of NaCl transport by loop diuretics results in a decrease in the osmolality of the medullary interstitial fluid.
- With a decrease in medullary interstitial fluid osmolality, water reabsorption from the collecting duct is impaired, and the concentrating ability of the kidneys is reduced. Water reabsorption from some portions of the thin descending limb of
- Henle’s loop also is impaired by loop diuretics, again because of the decrease in medullary interstitial fluid osmolality.
- This decrease in thin descending limb water reabsorption accounts in part for the increase in water excretion seen with loop diuretics.
“How does diuretic action affect tubular reabsorption?”
Loop diuretics are the most potent diuretics available, increasing the excretion of Na+ to as much as 25% of the amount filtered. This large natriuresis reflcts the fact that the thick ascending limb normally reabsorbs approximately 20% to 25% of the filtered Na+ and that downstream segments of the nephron have a limited ability to reabsorb the excess Na+ delivered as a consequence of loop diuretic action
4. Thiazide Diuretics
Like the loop diuretics, thiazide diuretics (Example: Hydrochlorothiazide, chlorthalidone, and metolazone)* are organic anions.
- Because they largely are bound to plasma proteins, they gain access to the tubular lumen primarily by secretion in the proximal tubule.
- They act to inhibit Na+ reabsorption in the early portion of the distal tubule by blocking the Na+-Cl– symporter in the apical membrane of these cells.
- Because water cannot cross this portion of the nephron, it is a site where the urine is diluted. Therefore thiazides reduce the ability to dilute the urine maximally by inhibiting NaCl reabsorption.
- Because thiazide diuretics act in the cortex and not the medulla, they do not affect the ability of the kidneys to concentrate the urine maximally. Natriuresis with thiazide diuretics is 5% to 10% of the fitered Na+
5. K+-Sparing Diuretics
K+-sparing diuretics act on the region of the nephron where K+ secretion occurs (i.e., the late portion of the distal tubule and cortical collecting duct). They produce a small natriuresis (3% to 5% of the filtered Na+), reflecting the amount of Na+ reabsorbed by this region of the nephron. As the name implies, their utility lies in their ability to inhibit K+ secretion by this region of the nephron.
“Diuretics and their role in regulating urine production”
There are two classes of K+ – sparing diuretics:
- One acts by antagonizing the action of aldosterone on the principal cell
- Example: Spironolactone and eplerenone), whereas the other class blocks the entry of
- Na+ into the same cells through the Na+– selective channels (epithelial sodium channels [ENaC]) in the apical membrane
Example:
Amiloride and triamterene). Amiloride and triamterene are organic cations that enter the tubular lumen primarily by secretion by the organic cation secretory system of the proximal tubule.
- Aldosterone stimulates both Na+ reabsorption and K+ secretion by the principal cells of the late distal tubule and collecting duct.
- Thus in the presence of an aldosterone antagonist, these effects are inhibited and both Na+ reabsorption and K+ secretion are reduced.
- The ability of the Na+ channel blockers amiloride and triamterene to inhibit Na+ reabsorption and K+ secretion is similar to that of spironolactone, but the cellular mechanism is different.
Amiloride and triamterene block the entry of Na+ into the principal cell by directly inhibiting the Na+ channel (ENaC) in the apical membrane.
- With decreased Na+ entry, reduced Na+ extrusion occurs across the basolateral membrane through Na+– K+ –adenosine triphosphatase (ATPase).
- This effect in turn reduces cellular K+ uptake and ultimately its secretion into the tubular fluid. Inhibition of apical membrane Na+ channels also alters the electrical profile across the luminal membrane, with the voltage across this membrane increasing in magnitude.
- Because of this voltage change, the electrochemical gradient for K+ movement out of the cell is reduced
This membrane voltage effect also contributes to the inhibition of K+ secretion. Trimethoprim is an antibiotic used to treat Pneumocystis jerovecii infections. P. jerovecii infections are commonly seen in persons whose immune systems are compromised
Example: Persons with acquired immunodeficiency syndrome).
Hyperkalemia may occur in persons treated with trimethoprim as a result of reduced renal K+ excretion. K+ excretion is reduced because trimethoprim inhibits K+ secretion by the principal cells of the late distal tubule and cortical collecting duct. The mechanism for this inhibition of K+ secretion is similar to that of amiloride and triamterene (i.e., direct inhibition of the Na+ channel [ENaC] in the apical membrane of the cell)
6. Aquaretics
In recent years, drugs that are antagonists of the AVP receptor (V2) have been developed
Example:
Tolvaptan and lixivaptan. These drugs act on the late portion of the distal tubule and the collecting duct to block the action of AVP.
- As a result of their action, the urine cannot be concentrated and dilute urine is excreted, reflecting the fact that tubular fluid reaching these AVP-sensitive segments of the nephron is hypoosmotic to the ECF
- These drugs are particularly helpful in treating patients whose ECF is hypoosmotic as a result of the failure of the kidneys to excrete solute-free water because AVP levels are elevated by nonosmotic and nonhemodynamic mechanisms
“Pathophysiology of diuretic-induced electrolyte imbalances”
Example: Syndrome of inappropriate AVP secretion [SIADH] *
Effect Of Diuretics On The Excretion Of Water And Solutes
Through their effects on Na+ handling along the nephron, diuretics also influence the handling of water and solutes. The various diuretics on the handling of some of these solutes and the ability of the kidneys to excrete (CH2O) and reabsorb (TCH2O) solute-free water.
1. Solute-Free Water
The ability of the kidneys to excrete or reabsorb solute-free water depends on several factors.
About the action of diuretic agents, the factors of concern are as follows:
- The normal function of the nephron segments (particularly the thick ascending limb)
- The delivery of adequate solute to Henle’s loop
- The maintenance of a hyperosmotic medullary interstitium (selective reabsorption of solute-free water)
The thick ascending limb of Henle’s loop is the most important site for the separation of solute and water.
- As noted, this separation not only dilutes the tubular fluid but also, by establishing a hyperosmotic medullary interstitium, allows water reabsorption from the collecting duct and thus the concentration of the urine.
- Inhibition of thick ascending limb Na+ reabsorption by loop diuretics therefore results in inhibition of both solute-free water excretion (CH2O) and solute-free water reabsorption (TCH2O).
- The early portion of the distal tubule is also a site of solute and water separation and thus tubular fluid dilution. Accordingly, inhibition of Na+ reabsorption by the thiazide diuretics impairs dilution of the urine.
- However, thiazide diuretics impair urine dilution to a lesser degree than do loop diuretics, reflecting the difference in the NaCl reabsorptive capacity between the distal tubule (5% of the filtered Na+) and the thick ascending limb (25% of the filtered Na+).
- In contrast to loop diuretics, thiazide diuretics do not significantly impair the ability of the kidneys to concentrate the urine.
- As already noted, the concentration of the urine requires a hyperosmotic medullary interstitium so that water can be reabsorbed from the collecting duct in the presence of AVP.
- Because thiazide diuretics act on distal tubules that are located in the cortex, their action at this site does not appreciably alter the medullary interstitial osmotic gradient.
- Consequently, urine-concentrating ability is unaffected by thiazide diuretics.
- The action of diuretics in the proximal tubule (osmotic diuretics and carbonic anhydrase inhibitors) increases the delivery of NaCl and water to Henle’s loop.
“Emerging research on diuretic mechanisms”
Because of the ability of the thick ascending limb to increase its transport rate in response to an increased delivered load of NaCl, the separation of solute and water increases. As a result, these diuretic agents increase the ability of the kidneys to excrete solute-free water and reabsorb solute-free water.
- Thus diuretics that act on the proximal tubule enhance the ability to concentrate and dilute the urine.
- Although the late portion of the distal tubule and the collecting duct can dilute the luminal fluid in the absence of AVP, Na+ transport in these segments is not of sufficient magnitude to contribute significantly to the excretion of solute-free water. Consequently, the K+-sparing diuretics do not appreciably alter free-water excretion.
- Like thiazide diuretics, K+-sparing diuretics do not alter solute-free water reabsorption because the nephron sites of action are located in the cortex.
- Aquaretics, because they act directly on the medullary portion of the collecting duct, increase solute-free water excretion and impair solute-free water reabsorption.
- K+ Excretion One of the major consequences of diuretic use (excluding the K+-sparing diuretics) is increased excretion of K+, which can be of sufficient magnitude to result in hypokalemia. The basis for this diuretic-induced increase in renal K+ excretion lies in the fact that when a diuretic inhibits Na+ and water reabsorption in segments upstream from the late portion of the distal tubule and cortical collecting duct (K+secretory site of the nephron), tubular fluid flow rate increases.
- The increased tubular fluid flow rate stimulates K+ secretion at this site addition, by their action on Na+ balance, diuretics decrease the ECF volume. This mechanism, in turn, leads to increased secretion of aldosterone by the adrenal cortex, which acts at this site to stimulate K+ secretion.
- The decrease in ECF volume also stimulates AVP secretion. As described, AVP stimulates K+ secretion by the principal cells of the late distal tubule and collecting duct.
- This stimulatory effect normally is offset by the inhibitory effect of the reduced tubular flow rate, which also is induced by AVP.
- As a result, AVP does not normally alter renal K+ excretion. However, in the presence of a diuretic that is acting upstream to the late portion of the distal tubule and the collecting duct, AVP does increase renal K+ excretion because in this setting tubular fluid flow is elevated by the action of the diuretics.
- Because K+-sparing diuretics prevent the increase in K+ excretion caused by the other diuretics, they usually are given in combination with these other diuretics to prevent or at least minimize the development of hypokalemia.
“Case studies on diuretic therapy outcomes”
2. HCO3– Excretion
By inhibiting H+ secretion in the proximal tubule and thereby increasing HCO3– excretion, carbonic anhydrase inhibitors can result in the development of metabolic acidosis.
- Although only carbonic anhydrase inhibitors directly alter H+ secretion by the nephron, all diuretics can affect systemic acid-base balance secondarily.
- Both loop and thiazide diuretics can induce metabolic alkalosis, which is a consequence of the decrease in ECF volume that accompanies their use. With a decrease in the ECF volume, Na+ is more avidly reabsorbed by the nephron.
- In the proximal tubule, this enhanced Na+ reabsorption results in enhanced H+ secretion through the Na+-H+ antiporter.
- Thus a greater fraction of the fitered HCO3– is reabsorbed. In addition, the reduction in ECF volume stimulates aldosterone secretion by the adrenal cortex. Aldosterone stimulates H+ secretion by intercalating cells of the distal tubule and collecting duct.
- Because, as noted, proximal tubule HCO3– reabsorption is increased, virtually none of the filtered HCO3– reaches the distal tubule.
Therefore the increased H+ secretion that occurs in the distal tubule and collecting duct results in the production of new HCO3– as the H+ is excreted with non HCO3– urinary buffers (i.e., titratable acid).
- The increased secretion of H+ in the distal tubule and collecting duct also enhances the excretion of NH4+, which results in the addition of new HCO3– to the ECF. As a result, net acid excretion by the kidneys is increased and metabolic alkalosis develops.
- By inhibiting Na+ reabsorption in the late portion of the distal tubule and cortical collecting duct, K+ – sparing diuretics secondarily inhibit H+ secretion and thus can lead to the development of metabolic acidosis.
- H+secretion by these nephron segments is facilitated by the lumen-negative transepithelial voltage.
- Normally, Na+ reabsorption in these nephron segments results in the generation of such a voltage. By inhibiting Na+ reabsorption and thus the negative luminal voltage, K+ -sparing diuretics reduce H+secretion.
- With reduced H+ secretion, insufficient quantities of net acid are excreted and metabolic acidosis ensues.
“Complications of ignoring diuretic side effects”
3. Ca++ and Pi Excretion
With the Excretion of K+ – sparing diuretics, all the diuretics can significantly alter Ca++ excretion by the kidney.
- With inhibition of proximal tubule solute and water reabsorption (osmotic diuretics and carbonic anhydrase inhibitors), reduced reabsorption of Ca++ and thus increased excretion occurs.
- The amount of Ca++ excreted is less than expected from inhibition of proximal tubule transport. This situation again reflects the ability of the downstream segments (particularly the thick ascending limb of Henle’s loop) to increase reabsorption after an increased delivery of Ca++.
- The mechanism by which these diuretics inhibit proximal tubule Ca++ reabsorption is related to their ability to reduce solvent drag. With the use of carbonic anhydrase inhibitors, increased Ca++ excretion occurs in the setting of alkaline urine (increased urinary (HCO3– ).
Because Ca++ is less soluble in alkaline urine, the potential exists for the formation of renal stones that contain Ca++ Loop diuretics also increase Ca++ excretion, an action explained by the effect of these diuretics on the transepithelial voltage of the thick ascending limb of Henle’s loop.
- Normally, the transepithelial voltage of this segment is oriented lumen positive 4), providing a driving force for the movement of Ca++ from the lumen to blood through the paracellular pathway.
- When the transport of NaCl by Henle’s loop is blocked by loop diuretics, this lumen-positive voltage is abolished, and thus the driving force for Ca++ reabsorption is reduced.
- Normally, Henle’s loop reabsorbs about 15% of the fitered Ca++ Inhibition of Ca++ reabsorption by loop diuretics therefore can have a significant effect on Ca++ excretion.
- For this reason, loop diuretics often are used to treat hypercalcemia.
- Despite this action of loop diuretics, hypercalcemia can occur with their long-term use. The mechanism responsible for this effect is related to the diuretic-induced decrease in the ECF volume.
- When the ECF volume is decreased, proximal tubule reabsorption is enhanced, which increases Ca++ reabsorption at this site and therefore decreases urinary Ca++ excretion.
“Advances in understanding diuretic action”
Thiazide diuretics stimulate Ca++ reabsorption by the cells of the distal tubule and thus reduce Ca++ excretion. The distal tubule normally reabsorbs approximately 10% to 15% of the filtered Ca++
- The reabsorption of Ca++ at this site is an active, transcellular process involving the entry of Ca++ into the cell through channels in the apical membrane and extrusion from the cell across the basolateral membrane by the Ca++ -ATPase and 3Na +– Ca++ antiporter.
- Thiazide diuretics, by inhibiting the entry of NaCl into the cell, cause the membrane potential to hyperpolarize (i.e., the cell interior becomes more electrically negative).
- This hyperpolarization in turn activates the Ca++ channel (TRPV5) in the apical membrane of the cell and increases the electrochemical gradient for Ca++ entry into the cell.
- The increased entry of Ca++ into the cell is matched by increased extrusion across the basolateral membrane by the Ca++-ATPase and 3Na+– Ca++antiporter (extrusion of Ca++ via the 3Na+– Ca++ antiporter is stimulated because the intracellular concentration of Na+ is decreased as a result of the diuretic blocking NaCa+ entry into the cell across the apical membrane).
- The net effect is an increase in Ca++ reabsorption.
“Global prevalence of diuretic use in hypertension”
- With long-term use of thiazide diuretics, the associated decrease in ECF volume stimulates proximal tubule reabsorption, including that of Ca++
- Because thiazides reduce urinary Ca++ excretion, they sometimes are used to lower the incidence of the formation of stones containing Ca++ in persons who normally excrete high levels of Ca++ in their urine. Except for the K+-sparing diuretics, all diuretics acutely increase Pi excretion.
- However, the cellular mechanisms for this effect are not completely understood.
The effect is modifid, however, with long-term diuretic therapy. With the decrease in ECF volume that accompanies long-term diuretic use, proximal tubule Na+ reabsorption is stimulated. Because the proximal tubule reabsorbs the largest portion of the filtered Pi and because this reabsorptive process is coupled with Na+, Pi excretion is reduced in this setting.
Leave a Reply