Physiology Of Body Fluids
“What is the physiology of body fluids?”
One of the major functions of the kidneys is to maintain the volume and composition of the body’s fluids constant despite wide variations in the daily intake of water and solutes.
Physiology Of Body Fluids chapter, the volume, and composition of the body’s fluids are discussed to provide a background for the study of the kidneys as regulatory organs.
Some of the basic principles, terminology, and concepts related to the properties of solutes in solution also are reviewed.
Physicochemical Properties Of Electrolyte Solutions
“Importance of studying the physiology of body fluids”
Molarity and Equivalence
The amount of a substance dissolved in a solution (i.e., its concentration) is expressed in terms of either molarity or equivalence. Molarity is the amount of a substance relative to its molecular weight.
For example, glucose has a molecular weight of 180 g/mol. If 1 L of water contains 1 g of glucose, the molarity of this glucose solution would be determined as:
⇒ \(\frac{1 \mathrm{~g} / \mathrm{L}}{180 \mathrm{~g} / \mathrm{mol}}=0.0056 \mathrm{~mol} / \mathrm{L} \text { or } 5.6 \mathrm{mmol} / \mathrm{L}\) → (1)
For uncharged molecules, such as glucose and urea, concentrations in the body fluids are usually expressed in terms of molarity.
Because many of the substances of biological interest are present at very low concentrations, units are more frequently expressed in the millimolar range (mmol/L).
“Common components of body fluids explained”
The concentration of solutes, which normally dissociate into more than one particle when dissolved in solution (For example., sodium chloride [NaCl]), is usually expressed in terms of equivalence.
Equivalence refers to the stoichiometry of the interaction between cation and anion and is determined by the valence of these ions.
For example, consider a 1 L solution containing 9 g of NaCl (molecular weight = 58.4 g/mol). The molarity of this solution is 154 mmol/L.
Because NaCl dissociates into Na+ and Cl– ions, and assuming complete dissociation, this solution contains 154 mmol/L of Na-+ and 154 mmol/L of Cl–.
Because the valence of these ions is 1, these concentrations also can be expressed as milliequivalents (mEq) of the ion per liter (i.e., 154 mEq/L for Na+ and Cl–, respectively).
For univalent ions such as Na+ and Cl–, concentrations expressed in terms of molarity and equivalence are identical. However, this is not true for ions having valences greater than 1.
Accordingly, the concentration of Ca++ (molecular weight = 40.1 g/mol and valence = 2) in a 1 L solution containing 0.1 g of this ion could be expressed as:
⇒ \(\frac{0.1 \mathrm{~g} / \mathrm{L}}{40.1 \mathrm{~g} / \mathrm{mol}}=2.5 \mathrm{mmol} / \mathrm{L}\) → (2)
= 2.5 mmol/L × 2 Eq/mol = 5 mEq
Although some exceptions exist, it is customary to express concentrations of ions in milliequivalents per liter (mEq/L).
“Role of electrolytes in body fluid physiology”
Osmosis And Osmotic Pressure
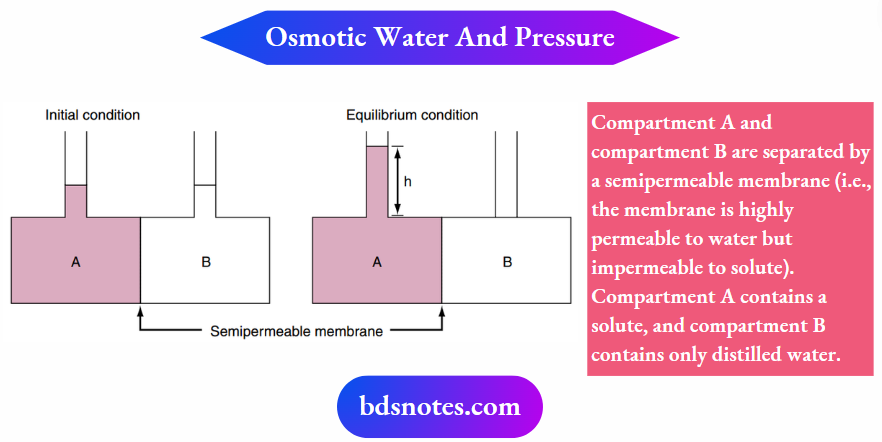
The movement of water across cell membranes occurs by the process of osmosis. The driving force for this movement is the osmotic pressure difference across the cell membrane.
Illustrates the concept of osmosis and the measurement of the osmotic pressure of a solution.
Osmotic pressure is determined solely by the number of solute particles in the solution. It is not dependent on factors such as the size of the solute particles, their mass, or their chemical nature (For Example., valence).
Osmotic pressure (π), measured in atmospheres (atm), is calculated by van’t Hoff’s law as follows:
π = nCRT → (3)
where n is the number of dissociable particles per molecule, C is total solute concentration, R is gas constant, and T is temperature in degrees Kelvin (°K).
For a molecule that does not dissociate in water, such as glucose or urea, a solution containing mmol/L of these solutes at 37° C can exert an osmotic pressure of 2.54 x 10-2 atm as calculated by equation 1-3 using the following values: n is 1, C is 0.001 mol/L, R is 0.082 atm L/mol, and T is 310° K.
“Functions of intracellular vs extracellular fluids”
Because 1 atm equals 760 mm Hg at sea level, π for this solution also can be expressed as 19.3 mm Hg.
Alternatively, osmotic pressure is expressed in terms of osmolarity (see the following discussion). Thus a solution containing 1 mmol/L of solute particles exerts an osmotic pressure of 1 milliosmole/L (1 mOsm/L).
For substances that dissociate in a solution, n of equation 3 has a value other than 1.
For example, a 150 mmol/L solution of NaCl has an osmolarity of 300 mOsm/L because each molecule of NaCl dissociates into a Na+ and a Cl– ion (i.e., n = 2).
If the dissociation of a substance into its component ions is not complete, n is not an integer. Accordingly, osmolarity for any solution can be calculated as:
Osmolarity= Concentration × Number of dissociable particles → (2)
mOsm/L = mmol/L × number of particles/mol
“Importance of sodium and potassium in body fluids”
“How does water distribution affect body fluid balance?”
Osmolarity And Osmolality
Osmolarity and osmolality are frequently confused and incorrectly interchanged. Osmolarity refers to the number of solute particles per 1 L of solvent, whereas osmolality is the number of solute particles in 1 kg of solvent.
For dilute solutions, the difference between osmolarity and osmolality is insignificant. Measurements of osmolarity are temperature dependent because the volume of solvent varies with temperature (i.e., the volume is larger at higher temperatures).
In contrast, osmolality, which is based on the mass of the solvent, is temperature-independent. For this reason, osmolality is the preferred term for biological systems and is used throughout this and subsequent chapters.
Osmolality has the units of Osm/kg H2O. Because of the dilute nature of physiologic solutions and because water is the solvent, osmolalities are expressed as milliosmoles per kilogram of water (mOsm/kg H2O).
Shows the relationships among molecular weight, equivalence, and osmoles for several physiologically significant solutes.
“Role of plasma in maintaining fluid homeostasis”

“Mechanisms of fluid balance in the body”
Tonicity
The tonicity of a solution is related to its effect on the volume of a cell. Solutions that do not change the volume of a cell are said to be isotonic.
A hypotonic solution causes a cell to swell, whereas a hypertonic solution causes a cell to shrink.
Although it is related to osmolality, tonicity also takes into consideration the ability of the solute to cross the cell membrane.
Consider two solutions: a 300 mmol/L solution of sucrose and a 300 mmol/L solution of urea. Both solutions have an osmolality of 300 mOsm/kg H2O and therefore are said to be isosmotic (i.e., they have the same osmolality).
When red blood cells (which, for the purpose of this illustration, also have an intracellular fluid osmolality of 300 mOsm/kg H2O) are placed in the two solutions.
Those in the sucrose solution maintain their normal volume, but those placed in urea swell and eventually burst. Thus the sucrose solution is isotonic and the urea solution is hypotonic.
The differential effect of these solutions on red cell volume is related to the permeability of the plasma membrane to sucrose and urea.
The red blood cell membrane contains uniporters for urea. Thus urea easily crosses the cell membrane (i.e., the membrane is permeable to urea), driven by the concentration gradient (i.e., extracellular [urea] > intracellular [urea]).
In contrast, the red blood cell membrane does not contain sucrose transporters, and sucrose cannot enter the cell (i.e., the membrane is impermeable to sucrose).
A solute must not permeate the red blood cell membrane to exert an osmotic pressure across a membrane because it is impermeable to sucrose when exerting an osmotic pressure.
It exerts an osmotic pressure equal to and opposite to the osmotic pressure generated by the contents of the red blood cell (in this case, 300 mOsm/kg H2O).
In contrast, urea is readily able to cross the red blood cell membrane, and it cannot exert an osmotic pressure to balance that generated by the intracellular solutes of the red blood cell.
“Role of kidneys in regulating body fluids”
Consequently, sucrose is termed an effective osmole and urea is termed an ineffective osmole. To take into account the effect of a solute’s membrane permeability on osmotic pressure, it is necessary to rewrite equation 1-3 as:
π = σ (nCRT) → (5)
where σ is the reflection coefficient or osmotic coefficient and is a measure of the relative ability of the solute to cross a cell membrane.
For a solute that can freely cross the cell membrane (such as urea in this example), σ = 0, and no effective osmotic pressure is exerted. Thus urea is an ineffective osmole for red blood cells.
In contrast, σ = 1 for a solute that cannot cross the cell membrane (i.e., sucrose). Such a substance is said to be an effective osmole.
Many solutes are neither completely able nor completely unable to cross cell membranes (i.e., 0 < σ < 1) and generate an osmotic pressure that is only a fraction of what is expected from the total solute concentration.
“How does ADH (antidiuretic hormone) affect fluid balance?”
Oncotic Pressure
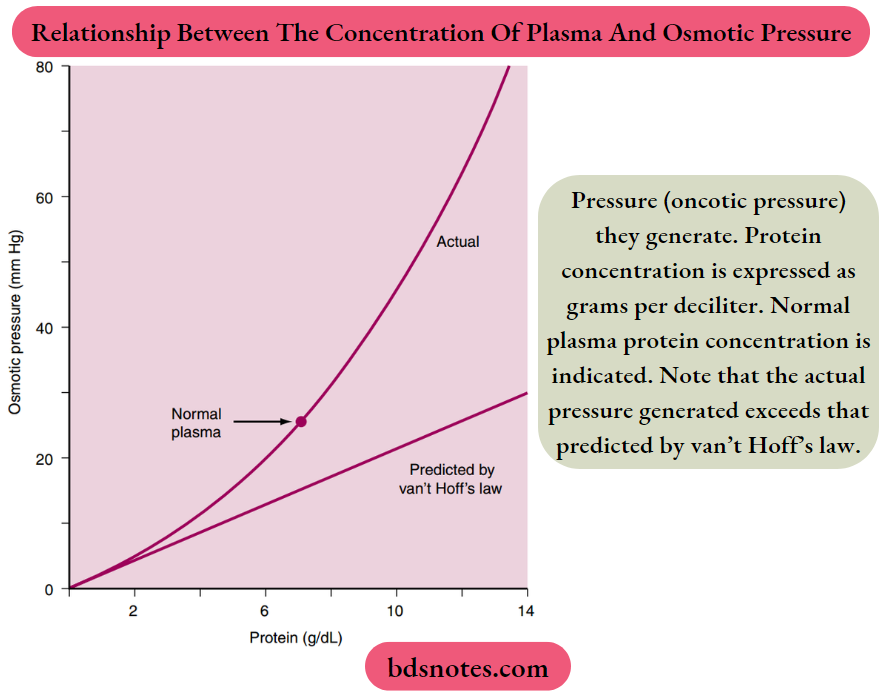
The oncotic pressure is the osmotic pressure generated by large molecules (especially proteins) in solution. As illustrated in Figure 1-2, the magnitude of the osmotic pressure generated by a solution of protein does not conform to van’t Hoff’s law.
The cause of this anomalous relationship between protein concentration and osmotic pressure is not completely understood but appears to be related to the size and shape of the protein molecule.
For example, the correlation to can’t Hoff’s law is more precise with small, globular proteins than with larger protein molecules.
The oncotic pressure exerted by proteins in human plasma has a normal value of approximately 26 to 28 mm Hg.
However, this pressure appears to be small when considered in terms of osmotic pressure (28 mm Hg ≈ 1.4 mOsm/kg H2O).
It is an important force involved in fluid movement across capillaries (details of this topic are presented in the following section on fluid exchange between body fluid compartments).
Specific Gravity
The total solute concentration in a solution also can be measured as specific gravity. Specific gravity is defined as the weight of a volume of solution divided by the weight of an equal volume of distilled water.
Thus the specific gravity of distilled water is 1. Because biological fluids contain a number of different substances, their specific gravities are greater than 1. For example, normal human plasma has a specific gravity in the range of 1.008 to 1.010.
Volumes Of Body Fluid Compartments
“Impact of aldosterone on body fluid regulation”
Water makes up approximately 60% of the body’s weight, with variability among individuals being a function of the amount of adipose tissue that is present.
Because the water content of adipose tissue is lower than that of other tissue, increased amounts of adipose tissue reduce the fraction of total body weight attributed to water.
The specific gravity of urine is sometimes measured in clinical settings and used to assess the concentrating ability of the kidney. The specific gravity of urine varies in proportion to its osmolality.
However, because specific gravity depends on both the number of solute particles and their weight, the relationship between specific gravity and osmolality is not always predictable.
For example, patients who have been injected with radiocontrast dye (molecular weight >500 g/mol) for radiographic studies can have high values of urine-specific gravity (1.040 to 1.050) even though the urine osmolality is similar to that of plasma (For Example., 300 mOsm/kg H2O).
The percentage of body weight attributed to water also varies with age. In newborns, it is approximately 75%. This percentage decreases to the adult value of 60% by 1 year of age.
As illustrated, total body water is distributed between two major compartments, which are divided by the cell membrane.
The intracellular fluid (ICF) compartment is the larger compartment and contains approximately two-thirds of the total body water.
The remaining one-third of the body water is contained in the extracellular fluid (ECF) compartment. Expressed as percentages of body weight, the volumes of total body water, ICF, and ECF are:
Total body water = 0.6 × (body weight)
ICF = 0.4 × (body weight)
ECF = 0.2 × (body weight) → (6)
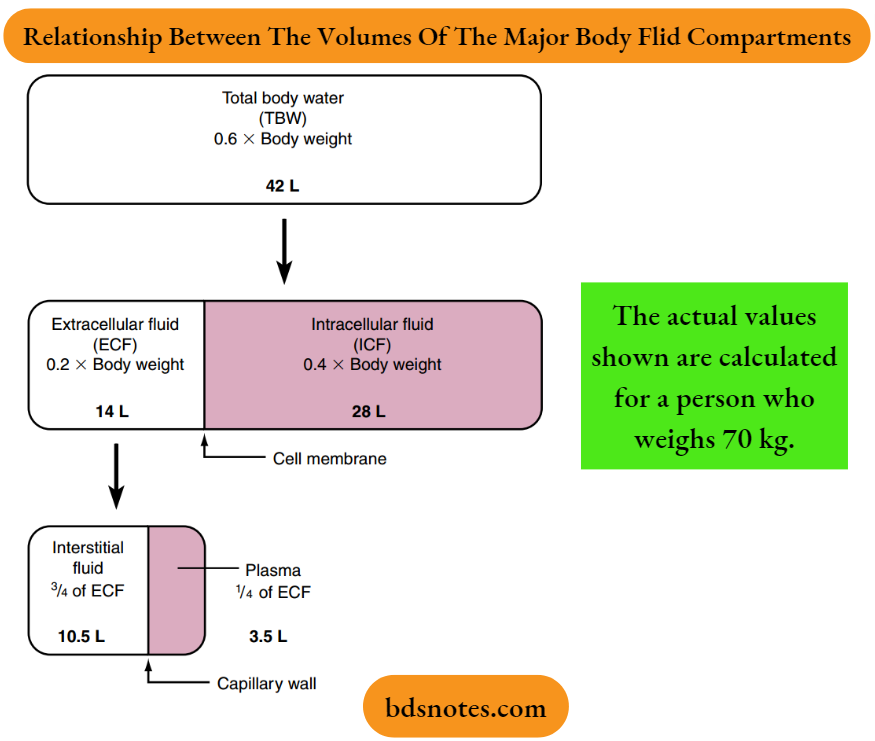
The ECF compartment is further subdivided into interstitial fluid and plasma, which are separated by the capillary wall. The interstitial fluid surrounds the cells in the various tissues of the body and constitutes three-fourths of the ECF volume.
The ECF includes water contained within the bone and dense connective tissue, as well as the cerebrospinal fluid. Plasma represents the remaining one-fourth of the ECF.
Under some pathologic conditions, additional fluid may accumulate in what is referred to as a “third space.”
Third space collections of fluid are part of the ECF and include, for example, the accumulation of fluid in the peritoneal cavity (ascites) of persons with liver disease.
Composition Of Body Fluid Compartments
Sodium is the major cation of the ECF and Cl– and bicarbonate (HCO–3 ) are the major anions.
The ionic composition of the plasma and interstitial fluid compartments of the ECF is similar because they are separated only by the capillary endothelium, a barrier that is freely permeable to small ions.
The major difference between the interstitial fluid and plasma is that the latter contains significantly more protein.
This differential concentration of protein can affect the distribution of cations and anions between these two compartments (i.e., the Donnan effect).
Because plasma proteins have a net negative charge that tends to increase the cation concentrations and reduce the anion concentrations in the plasma compartment.
However, this effect is small, and the ionic compositions of the interstitial fluid and plasma can be considered identical.
Because of its abundance, Na+ (and its attendant anions, primarily Cl– and HCO–3 ) is the major determinant of ECF osmolality.
Accordingly, a rough estimate of the ECF osmolality can be obtained by simply doubling the sodium concentration [Na–].
For example, if the plasma [Na+] is 145 mEq/L, the osmolality of plasma and ECF can be estimated as:
Plasma osmolality= 2(plasma [Na+]) → (7)
= 290 mOsm/kg H2O
Because water is in osmotic equilibrium across the capillary endothelium and the plasma membrane of cells, measurement of the plasma osmolality also provides a measure of the osmolality of the ECF and ICF.
In clinical situations, a more accurate estimate of the plasma osmolality is obtained by also considering the contribution of glucose and urea to the plasma osmolality.
Accordingly, plasma osmolality can be estimated as:
⇒ \(\begin{aligned}
& \text { Plasma osmolality } = 2\left(\text { plasma }\left[\mathrm{Na}^{+}\right]\right)+\frac{[\text { glucose }]}{18}+\frac{[\text { urea }]}{2.8}
\end{aligned}\) → (8)
The glucose and urea concentrations are expressed in units of mg/dL (dividing by 18 for glucose and 2.8 for urea allows conversion from the units of mg/dL to mmol/L and thus to mOsm/kg H2O).
This estimation of plasma osmolality is especially useful when dealing with patients who have an elevated plasma [glucose] level as a result of diabetes mellitus and patients with chronic renal failure whose plasma [urea] level is elevated.
In contrast to the ECF, where the [Na+] is approximately 145 mEq/L, the [Na+] of the ICF is only 10 to 15 mEq/L. K+ is the predominant cation of the ICF, and its concentration is approximately 150 mEq/L.
This asymmetric distribution of Na+ and K+ across the plasma membrane is maintained by the activity of the ubiquitous sodium-potassium–adenosine triphosphatase (Na+-K+-ATPase) mechanism.
By its action, Na+ is extruded from the cell in exchange for K+. The anion composition of the ICF differs from that of the ECF.
For example, Cl– and HCO–3 are the predominant anions of the ECF, and organic molecules and the negatively charged groups on proteins are the major anions of the ICF.
“Fluid intake and output: Physiology explained”
Fluid Exchange Between Body Fluid Compartments
Water moves freely and rapidly between the various body fluid compartments. Two forces determine this movement: hydrostatic pressure and osmotic pressure.
Hydrostatic pressure from the pumping of the heart (and the effect of gravity on the column of blood in the vessel) and osmotic pressure exerted by plasma proteins (oncotic pressure) are important determinants of fluid movement across the capillary wall.
By contrast, because hydrostatic pressure gradients are not present across the cell membrane, only osmotic pressure differences between ICF and ECF cause fluid movement into and out of cells.
Capillary Fluid Exchange
The movement of fluid across a capillary wall is determined by the algebraic sum of the hydrostatic and oncotic pressures (the so-called Starling forces) as expressed by the following equation:
⇒ \(\text { Filtration rate }=K_f\left[\left(\mathrm{P}_c-\mathrm{P}_{\mathrm{i}}\right)-\sigma\left(\pi_c-\pi_{\mathrm{i}}\right)\right]\) → (9)
where the filtration rate is the volume of fluid moving across the capillary wall (expressed in units of either volume or capillary surface area or volume/time).
Where Kf is the filtration coefficient of the capillary wall, Pc is hydrostatic pressure within the capillary lumen, πc is the oncotic pressure of the plasma.
Pi is the hydrostatic pressure of the interstitial fluid, πi is the oncotic pressure of the interstitial fluid, and σ is the reflection coefficient for proteins across the capillary wall.
The Starling forces for capillary fluid exchange vary between tissues and organs. They also can change in a given capillary bed under physiologic conditions (For Example., exercising muscle) and pathophysiologic conditions (For Example., congestive heart failure).
Illustrates these forces for a capillary bed located in skeletal muscle at rest.
The capillary filtration coefficient (Kf) reflects the intrinsic permeability of the capillary wall to the movement of fluid, as well as the surface area available for filtration.

“Pathophysiology of fluid imbalances explained”
The Kf varies among different capillary beds. For example, the Kf of glomerular capillaries in the kidneys is approximately 100 times greater in magnitude than that of skeletal muscle capillaries.
This difference in Kf accounts for the large volume of fluid filtered across glomerular capillaries compared with the amount filtered across skeletal muscle capillaries.
The hydrostatic pressure within the lumen of a capillary (Pc) is a force promoting the movement of fluid from the lumen into the interstitium.
Its magnitude depends on arterial pressure, venous pressure, and precapillary (arteriolar) and postcapillary (venular and small vein) resistances.
An increase in arterial or venous pressures results in an increase in Pc, whereas a decrease in these pressures has the opposite effect.
Pc increases with either a decrease in precapillary resistance or an increase in postcapillary resistance. Likewise, an increase in precapillary resistance or a decrease in postcapillary resistance decreases Pc.
For virtually all capillary beds, precapillary resistance is greater than postcapillary resistance, and thus the precapillary resistance plays a greater role in determining Pc.
An important exception is the glomerular capillaries, where both precapillary and postcapillary resistances modulate Pc.
The magnitude of Pc varies not only among tissues but also among capillary beds within a given tissue; it also is dependent on the physiologic state of the tissue.
Precapillary sphincters control not only the hydrostatic pressure within an individual capillary but also the number of perfused capillaries in the tissue.
For example, in skeletal muscle at rest, not all capillaries are perfused. During exercise, the relaxation of precapillary sphincters allows perfusion of more capillaries.
The increased number of perfused capillaries reduces the diffusion distance between the cells and capillaries and thereby facilitates the exchange of O2 and cellular metabolites (For Example., carbon dioxide [CO2] and lactic acid).
The hydrostatic pressure within the interstitium (Pi) is difficult to measure, but in the absence of edema (i.e., abnormal accumulation of fluid in the interstitium), its value is near zero or slightly negative.
“Emerging research on body fluid regulation”
Thus under normal conditions, it causes fluid to move out of the capillary. However, when edema is present, Pi is positive and it opposes the movement of fluid out of the capillary.
The oncotic pressure of plasma proteins ( πc) retards the movement of fluid out of the capillary lumen. At a normal plasma protein concentration, πc has a value of approximately 26 to 28 mm Hg.
The degree to which oncotic pressure influences capillary fluid movement depends on the permeability of the capillary wall to the protein molecules.
If the capillary wall is highly permeable to protein, σ is near zero and the oncotic pressure generated by plasma proteins plays little or no role in capillary fluid exchange.
This situation is seen in the capillaries of the liver (i.e., hepatic sinusoids), which are highly permeable to proteins. As a result, the protein concentration of the interstitial fluid is essentially the same as that of plasma.
In the capillaries of skeletal muscle, σ is approximately 0.9, whereas in the glomeruli of the kidneys, the value is essentially 1.
Therefore plasma protein oncotic pressure plays an important role in fluid movement across these capillary beds.
The protein that leaks across the capillary wall into the interstitium exerts an oncotic pressure ( πi) and promotes the movement of fluid out of the capillary lumen.
In skeletal muscle capillaries under normal conditions, πi is small and has a value of only 8 mm Hg. As depicted, the balance of Starling forces across muscle capillaries causes fluid to leave the lumen (filtration) along its entire length.
Some of this filtered fluid reenters the vasculature across the postcapillary venule where the Starling forces are reversed (i.e., the net driving force for fluid movement is into the vessel).
The remainder of the filtered fluid is returned to the circulation through the lymphatics. The sinusoids of the liver also filter along their entire length.
In contrast, during the digestion of a meal, the balance of forces across the capillaries of the gastrointestinal tract results in the net uptake of fluid into the capillary.
Normally, 8 to 12 L/day of fluid moves across capillary beds throughout the body and is collected by lymphatic vessels. This lymphatic fluid flows first to lymph nodes, where most of the fluid is returned to circulation.
Fluid not returned to the circulation at the lymph nodes (1 to 4 L/day) reenters the circulation through the thoracic and right lymphatic ducts.
However, under conditions of increased capillary filtration, such as that which occurs in persons with congestive heart failure, thoracic and right lymphatic duct flow can increase 10-fold to 20-fold.
Cellular Fluid Exchange
Osmotic pressure differences between ECF and ICF are responsible for fluid movement between these compartments. Because the plasma membrane of cells contains water channels (aquaporins [AQPs]), water can easily cross the membrane.
Thus a change in the osmolality of either ICF or ECF results in rapid movement (i.e., in minutes) of water between these compartments. Thus, except for transient changes, the ICF and ECF compartments are in osmotic equilibrium.
Water movement across the plasma membrane of cells occurs through a class of integral membrane proteins called aquaporins (AQPs).
Although water can cross the membrane through other transporters (For Example., an Na+– glucose symporter), AQPs are the main route of water movement into and out of the cell.
To date, 13 AQPs have been identified. These AQPs can be divided into two subgroups.
One group, which includes the AQP involved in the regulation of water movement across the apical membrane of renal collecting duct cells by arginine vasopressin (AQP-2), is permeable only to water.
The second group is permeable not only to water but also to low-molecular-weight substances, including gases and metalloids.
Because glycerol can cross the membrane via this group of aquaporins, they are termed aquaglyceroporins.
AQPs exist in the plasma membrane as a homotetramer, with each monomer functioning as a water channel.
In contrast to the movement of water, the movement of ions across cell membranes is more variable from cell to cell and depends on the presence of specific membrane transport proteins.
Consequently, as a first approximation, fluid exchange between the ICF and ECF under pathophysiologic conditions can be analyzed by assuming that appreciable shifts of ions between the compartments do not occur.
A useful approach for understanding the movement of fluids between the ICF and the ECF is outlined. To illustrate this approach, consider what happens when solutions containing various amounts of NaCl are added to the ECF.
Principles For Analysis Of Fluid Shifts Between ICF And ECF
The volumes of the various body fluid compartments can be estimated in a healthy adult as shown.
- All exchanges of water and solutes with the external environment occur through the extracellular fluid (ECF) (For Example., intravenous infusion and intake or loss via the gastrointestinal tract).
- Changes in the intracellular fluid (ICF) are secondary to fluid shifts between the ECF and the ICF. Fluid shifts occur only if the perturbation of the ECF alters its osmolality.
- Except for brief periods of seconds to minutes, the ICF and the ECF are in osmotic equilibrium. A measurement of plasma osmolality provides a measure of both the ECF and the ICF osmolality.
- For the sake of simplification, it can be assumed that equilibration between the ICF and the ECF occurs only by the movement of water and not by the movement of osmotically active solutes.
- Conservation of mass must be maintained, especially when considering either the addition or removal of water and or solutes from the body.
Example 1: Addition of Isotonic NaCl to ECF
The addition of an isotonic NaCl solution (For Example., intravenous infusion of 0.9% NaCl:
osmolality ≈290 mOsm/kg H2O to a patient) to the ECF increases the volume of this compartment by the volume of fluid administered.
Because this fluid has the same osmolality as ECF and therefore also has the same osmolality as ICF, no driving force for fluid movement between these compartments exists, and the volume of ICF is unchanged.
Although Na+ can cross cell membranes, it is effectively restricted to the ECF by the activity of Na+-K+-ATPase, which is present in the plasma.
Neurosurgical procedures and cerebrovascular accidents (strokes) often result in the accumulation of interstitial fluid in the brain (i.e., edema) and swelling of the neurons.
Because the brain is enclosed within the skull, edema can raise intracranial pressure and thereby disrupt neuronal function, leading to coma and death.
The blood-brain barrier, which separates the cerebrospinal fluid and brain interstitial fluid from the blood, is freely permeable to water but not to most other substances.
As a result, excess fluid in brain tissue can be removed by imposing an osmotic gradient across the blood-brain barrier. Mannitol can be used for this purpose.
Mannitol is a sugar (molecular weight = 182 g/ mol) that does not readily cross the blood-brain barrier and membranes of cells (neurons as well as other cells in the body).
Therefore mannitol is an effective osmole and intravenous infusion results in the movement of fluid from the brain tissue by osmosis.
“Case studies on body fluid physiology outcomes”
Example 2: Addition of Hypotonic NaCl to ECF
The addition of a hypotonic NaCl solution to the ECF (For Example., intravenous infusion of 0.45% NaCl:
osmolality <145 mOsm/kg H2O to a patient) decreases the osmolality of this fluid compartment, resulting in the movement of water into the ICF.
After osmotic equilibration, the osmolalities of ICF and ECF are equal but lower than before the infusion, and the volume of each compartment is increased. The increase in ECF volume is greater than the increase in ICF volume.
Example 3: Addition of Hypertonic NaCl to ECF
The addition of a hypertonic NaCl solution to the ECF (For Example., intravenous infusion of 3% NaCl:
osmolality ≈1000 mOsm/kg H2O to a patient) increases the osmolality of this compartment, resulting in the movement of water out of cells.
After osmotic equilibration, the osmolalities of ECF and ICF are equal but higher than before the infusion. The volume of the ECF is increased, whereas that of the ICF is decreased.
Fluid and electrolyte disorders often are seen in clinical practice (For Example., in patients with vomiting and/or diarrhea). In most instances, these disorders are self-limited, and correction of the disorder occurs without the need for intervention.
However, more severe or prolonged disorders may require fluid replacement therapy. Such therapy may be administered orally with special electrolyte solutions, or intravenous fluids may be administered.
Intravenous solutions are available in many formulations. The type of fluid administered to a particular patient is dictated by the patient’s need.
For example, if an increase in the patient’s vascular volume is necessary, a solution containing substances that do not readily cross the capillary wall is infused (For Example., 5% albumin solution).
The oncotic pressure generated by the albumin molecules retains fluid in the vascular compartment, expanding its volume.
Expansion of extracellular fluid (ECF) is accomplished most often by using isotonic saline solutions (For Example., 0.9% sodium chloride [NaCl]).
“Global prevalence of fluid imbalance disorders”

“Complications of ignoring fluid balance issues”
As already noted, administration of an isotonic NaCl solution does not result in the development of an osmotic pressure gradient across the plasma membrane of cells.
Therefore the entire volume of the infused solution remains in the ECF. Patients whose body flids are hyperosmotic need hypotonic solutions.
These solutions may be hypotonic NaCl (For Example., 0.45% NaCl or 5% dextrose in water [D5W]). Administration of D5W is equivalent to an infusion of distilled water because the dextrose is metabolized to CO2 and water.
The administration of these fluids increases the volumes of both the intracellular fluid (ICF) and ECF.
Finally, patients whose body fluids are hypotonic need hypertonic solutions, which typically are solutions that contain NaCl (For Example., 3% and 5% NaCl).
These solutions expand the volume of the ECF but decrease the volume of the ICF.
Other constituents, such as electrolytes (For Example., K+) or drugs, can be added to intravenous solutions to tailor the therapy to the patient’s fluid, electrolyte, and metabolic needs.
Leave a Reply