Peripheral Smear
Aim: To count different types of leucocytes and find out blood pictures by thin blood smear.
Apparatus: Irishman’s stain (0.5 g of Irishman’s powder in 100 ml of acetone-free methyl alcohol).
Procedure:
Preparation of blood smear
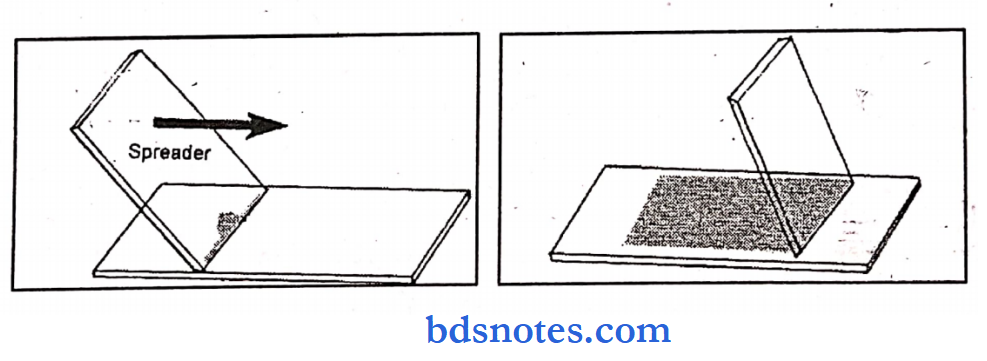
- Place a drop of blood in the center of a clean glass slide 1 to 2 cm from one end.
- Place another slide (spreader) with a smooth edge at an angle of 30-45° near the drop of blood.
- Move the spreader backward so that it makes contact with a drop of blood.
- Then move the spread forward rapidly over the slide.
- A thin peripheral blood film is thus prepared.
- Dry it and stain it.
Pathology practicals for MBBS
Procedure for staining:
- Pour Irishman’s stain dropwise (counting the drops) on the slide and wait for two minutes.
- This allows the fixation of the PBF in methyl alcohol.
- Add double the quantity of buffered water dropwise over the slide (i.e double the number of drops)
- Mix in water for 1 to 2 minutes
- Dry in the air and examine under oil immersion.
Hematology practicals BDS
Observation:
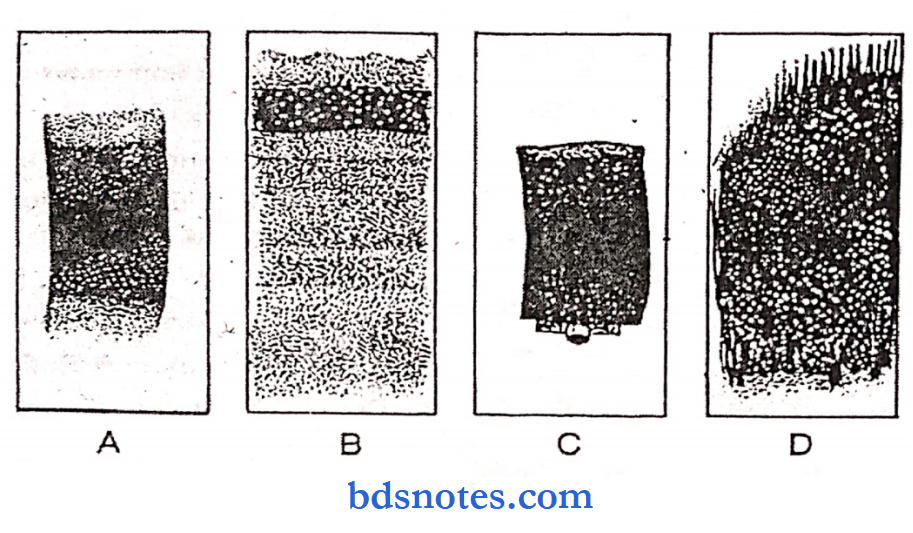
- Under low -power:
- Head: any cells (too thick of count)
- Body: for lymphocytes (small cells) predominant cells
- Tail: For Neutrophils (larger cells) predominant cells.
Quality of film:
RBC and WBC:
- Number
- Distribution
- Staining
- Degree of rouleaux formation
Parasites: microfilaria, hemoflagellates, plasmodia.
- Under oil immersion:
RBC:
- Size:
- Shape:
- Inclusions:
- Haemoparasites: Malaria
- Immature red cells:
Courses:
- Preparation and Staining:
- Well-stained blood film looks like this when stained with Romanwosky’s method of staining.
Macroscopically:
The slide will show a pinkish tint at the area of the blood film, the other part of the slide will show no stain.
Microscopically:
- RBCs will have a pinkish-orange color.
- The nuclei of leucocytes will be purplish-blue.
- Neutrophilic granules will have violet pinkish/iliac color.
- Eosinophilic granules will show red color and Basophilic granules will have dark blue color overlapping the nuclei.
- Peripheral blood smear study on leucocytes, platelets, and parasites

Qualities of a good blood film:
- It should not cover the entire surface of the slide and should have a smooth and even appearance.
- It should be free from waves and holes; it should not have an irregular tail.
Types of peripheral blood film:
- Thin blood film
- Thick blood film – prepared for detecting blood parasites such as malaria and microfilaria.
Different methods of preparing thin blood film:
- Slide method.
- Cover glass method.
- Spin method.
Effect of pH on staining with RS:
- A pH of 6.8 is recommended for general staining of the granules of leucocytes.
- A pH of 7 is best for staining the Schuffner’s dots.
- An alkaline pH accentuates the azure components at the expense of eosin and an acidic pH accentuates the eosin Y component at the expense of Azure B.
- An acidic pH disturbs the coloration of the granules of eosinophils.
Parts of thin blood film:
- Head, body, and tail.
Stained blood film shows an indistinct nucleus with scattered granules:
- This occurs due to chlorinated tap water instead of buffer or fresh distilled water.
Stain commonly used for staining the blood film in malaria surveys:
- Field stain is a rapid stain and stains the parasitic nucleus very clearly. Hence it is used in the survey work.
Utility of peripheral blood smear examination:
- Innumerable features are obtained from a PBS for diagnostic purposes and should, therefore, be examined for the following
- The alteration of size (macrocytosis, microcytosis, anisocytosis) of RBCs.
- The alteration in the shape (spherocytosis, elliptocytosis, ovalocytosis, acanthocytosis, burr cells, target cells) of RBCs.
- The appearance and color reaction (normochromia, hypochromia polychromasia) of RBCs.
- Presence of immature red cells – normoblasts, erythroblasts.
- Rouleaux formation – The arrangement of RBCs in Rouleaux is characteristic of multiple myeloma and macroglobulinemia.
- An idea of total leucocyte count (leucocytosis, leucopenia) to check the leucocyte count.
- Differential count to obtain information about neutrophilia, eosinophilia, basophilia, lymphocytosis, monocytosis, etc.
- Detection of immature leucocytes like blasts or partly mature leucocytes.
- A rough estimate of the number of platelets – thrombocytopenia thrombocytosis.
- The morphology of platelets, e.g. giant platelets in thrombasthenia.
- Presence of parasites in dead blood, e.g. malaria, microfilaria, trypanosomes.
- Occasionally LD bodies in monocytes, borrelia of relapsing fever, leptospira of Weil’s disease.
Urine analysis practical
Stains used for staining PBS and mention their utility:
- The stains used are:
Leishman’s stain – This is routinely used for staining the blood smear.
- This is a very easy stain to prepare and takes only a few minutes for staining.
- The granules of WBCs, the RBC, and platelets take this stain very well showing their characteristics.
- Giemsa stain This stain is also widely used as this gives the proper staining of all the cells of PBS, the intracellular parasites, and the viral inclusions.
- Field’ stain – This is a very quick method of staining.
- It takes only 24 hours to prepare the stain and only 4 o 5 seconds for staining the slides.
- This is helpful for field studies of malaria.
- Survey.
- Mac-Grunwald’s stain – This stain and Giemsa stain, if used together, give better results.
- Blood, bone marrow and other aspiration smears are well stained with this technique.
- The slides with proper mounting can be preserved for a long period.
Possible errors if the stain shows excessive blue color:
The possible errors are:
- Over alkalinity of either tire stain or tire buffer.
- Insufficient washing with the buffer and.
- Overtly hick smear.
Possible errors if the stained slide shows red stain:
The errors may be in:
- Too short staining time,
- Excessive washing,
- Acidic pH of the buffer and
- Acidic pH of tire diluting water if distilled water is used instead of buffer.
RBC abnormalities found in the blood after splenectomy:
- After splenectomy, target cells, schistocytes, Pappenheimer bodies, and Howell- Jolly bodies are commonly found in the blood smear.
Composition of Leishman’s Stain and its Role:
- Leishman’s stain contains 0.2 g of Leishman’s powder in 100 ml of acetone-free methyl alcohol.
- Leishman’s powder is eosin methylene blue (acid dye), which stains the nucleus pink and the cytoplasm blue.
- The functions of acetone, free methyl alcohol are – to act as a fixative, maintain neutral pH, prevent hemolysis, and increase the longevity of stain.
Method of staining of thick smear:
- The slide is dehaemoglobinised by immersion into distilled water for 10 minutes.
- LI.The slide is immersed in Stevenel’s blue for 6 seconds and then washed in tap water.
- The slide is then immersed in eosin solution for 12 seconds and then washed in tap water and allowed to dry.
Observation:
Conclusion of central pallor of RBC and its grades:
- Normal red cells have a central area of pallor.
- When this is increased, called hypochromic and suggests decreased hemoglobin concentration.
- Rarely do cells appear without central pallor (hyperchromic).
- This does not indicate increased hemoglobin concentration but suggests an altered shape of the cell or altered thickness of the membrane.
Predominant cells in body and tail:
In the body lymphocytes are predominant, in the tail neutrophils are predominant cells, v How do you count WBC:
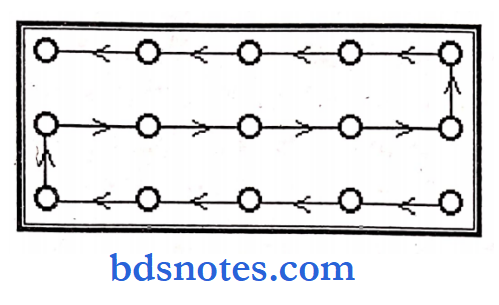
Count the cells as they come till a count of 100 cells is obtained going from one edge of the smear to the other end.
Counted at an area near the junction of the body with the tail of the smear where there is slight overlapping of RBC i.e. neither roulex formation, which occurs in head and body, nor totally scattered RBCs as occurs at the tail.
By moving the slide in a horizontal direction under oil immersion start counting the types of WBCs and go on entering P, L, M, E, and B in a box having 100 cubes.
Alternatively, 100 leucocytes can be counted by pressing the keys of the automated DLC counter. Zigzag counting is discouraged.
Clinical Conditions:
Be thorough with normal appearances size, shape, number of RBC WBC and platelets and their variations:
Agranulocytosis:
It is characterized by rapid fall in total solid leucocyte count, and the disappearance of mature granulocytes, which may not constitute more than 5 to 15% of the differential count.
There is usually no anemia.
Causes; Usually followed by drug allergies to various drugs such as aminopyrine, analgesics (APC, Analgin), hypnotics, diuretics, antihistamines, and antibiotics.
It is a very serious condition, which requires prompt medical attention (isolation and prophylactic antibiotic therapy).
Abnormal findings of peripheral blood smear:
Acute leukemia:
- Normochromic severe progressive anemia. Thrombocytopenia. WBC is more than 1 lakh per ml. Blast cells are present. Neutropenia.
AML:
- Anemia, and thrombocytopenia, show immature forms and blasts – myeloblasts, promyelocytes, myelocytes, metamyelocytes, and band/stab forms.
- “Auer bodies” (eosinophilic rod-like inclusion) are seen in myeloblasts and promyelocytes.
- Myeloid series of cells stain positive with sudden black B (Myeloperoxidase).
ALL:
- Lymphoblasts, Li, L2, L3 Lymphoid series are PAS +ve.
CML:
- Normocytic, normochromic type moderate degree anemia and marked leukocytosis (more than 2 lacks per ml).
- In the chronic phase – the excessive proliferation of myeloid cells of intermediate grade (myelocytes and metamyelocytes) and mature segmented neutrophils.
- An increase in the proportion of basophils up to 10% is a characteristic feature of CML.
CLL:
- Characterized by the proliferation and accumulation of relatively mature appearing lymphocytes in the blood, BM, spleen, liver, and other organs.
- Mild to moderate and normocytic, normochromic anemia. WBC count moderately increased (50,000 to 2,00, 000 per ml), 90% of leukocytes are mature small lymphocytes; platelets are normal/reduced.
Aleukemic/Subleukemic leukemia:
- The total count may be normal or below normal with very few blast cells.
Iron deficiency anemia:
- Red cells are hypochromic and microcytic and there is anisocytosis and poikilocytosis.
- Target cells, elliptical forms, and polychromatic cells are often present.
- Where iron deficiency is associated with Vitamin B12 deficiency there will be a dimorphic picture.
Megaloblastic anemia:
- Red cells are macrocytic. Total white blood cell count may be reduced.
Thalassemia:
- Microcytic, hypochromic red cell morphology marked anisopokilocytosis, basophilic stippling, and the presence of many target cells, teen drop cells, and normoblasts.
Revised FAB classification of acute leukemia:

Storage of blood:
- It should be stored in a chemically clean container in order to avoid bacterial contamination.
- Anticoagulants may be used.
A blood smear is used for:
RBC Count
RBCs are non-nucleated, circular, disc-shaped, biconcave cells being highly elastic because of spectrin (Protein).
- Mean diameter -7.5um
- Area – 120-135u2
- Volume-80u3
- Peripheral thickness- 2um
- Central thickness-1um
- Life spam -120 days
Apparatus & reagents:
- RBC Pipette
- Hemocytometer
- Microscope (observe with 10 x &45 x lenses)
- Sterile blood lancet.
- Cotton swabs
- Hayem’s fluid (Diluting fluid for RBC counting)
Composition of Hayem’s Fulid:
- Nacl-0.5g( Maintains isotonicity)
- Na2SO4-2.5g (anticog)
Procedure:
- Under aseptic conditions, give a sharp finger prick (discard first 2-3 drops of blood)
- Draw blood up to 0.5 mark and Hayem’s fluid up to 101 mark into the RBC pipette respectively and mix them for about 5 min.
- Discard the first 2 drops from the pipette and charge the hemocytometer with the diluted blood and wait for 3-4 min for the cells to settle.
- Use LP cells to focus the grid lines & HP lens to count the cells.
Counting and calculations:
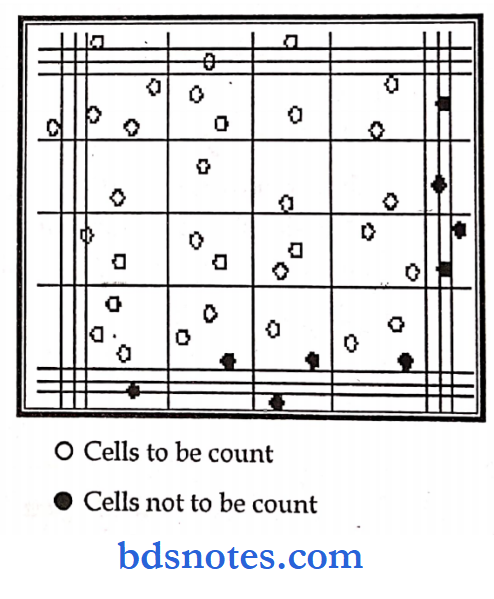
RBCs are counted in the four corners & one central square (each consisting of 16 small Squares, a total of 80 small squares), and the cells lying on their left and lower lines are included while those lying on the right and upper lines are excluded to avoid double counting.
No. of total squares = 80 Vol. of each square = (L x B x H) = 1/ 20×1/ 20×1/ 20 = 1/ 4000 mm3 Vol. of 80 squares = 1/4000 x 80 = 1/50 mm3 Dilution factor = 200 (Since volume of bulb = 100 Paiis of which 0.5 part is blood & 99.5 parts is diluent).
Let the no. of cells in 80 squares be X X cells present in fyso mm3 No. of RBC in 1mm3. = X x 50.
Total no. of RBC in 1mm3 = X x 50 x 200
Degree of error: 10 – 15 %.
Normal cell counts: Adult male – 5 to 5.5 million per mm3
Adult female – 4.5 to 5 million per mm3 Newborn – 7 to 8 million per mm3
Hemoglobin Estimation
- Hemoglobin is a complex substance present in the RBC and gives a red color to the blood.
- It is an iron-containing protein made up of a large protein molecule, globin, and four heme molecules.
- The function of hemoglobin is to carry oxygen to the tissues and CO2 away from it.
- Estimation of Hemoglobin: Is indicated as a part of the complete blood picture, in all types of anemia, leukemias, and chronic diseases. This can be done by:
Colorimetric method:
Tal quist method: This is very quick and easy but is inaccurate.
Sahli’s method or Acid Haematin Method.
Alkaline haematin method: Here Hb is converted to alkaline haematin by the addition of N/10 NaOH.
Haldane method: Here Hb is exposed to CO so carboxy Hb is formed.
Dane method.
Photo Electric method: Cyanmethemoglobin method: the most perfect &accurate method.
Specific gravity methods: This is mostly used for screening blood donors.
Chemical method: Where Fe content is the criteria. It is a complex, difficult, and time-consuming method but is accurate.
Gasometric method: Based on the O2 combining capacity of the blood, it measures only functional Hb.
- It is difficult and time-consuming.
Sahlis method
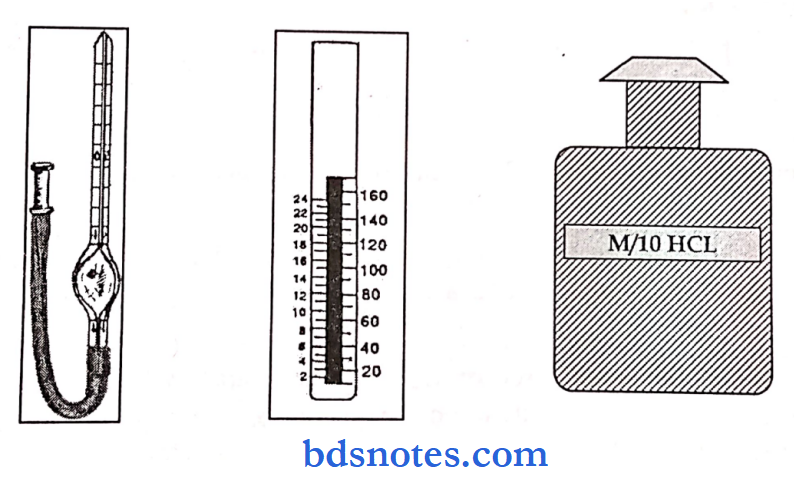
Aim: To estimate Hb in blood by acid haematin method.
Apparatus: Sahli’s haemoglobinometer tube and apparatus, pipette, glass rods, N/10 HC1, blood, distilled water, spirit, sterile cotton wool, and pricking needle.
Principle: is converted into acid hematin by dilute HQ (cause hemolysis and case Hb), which is brown in color.
The intensity of this color depends on, CTC’ C0J°iur solution being matched against the brown tinted glass filter, by direct vision, which gives the reading in gm per 100 ml blood.
Method:
- Take N/ 10 HCL up to 0 marks on the percentage scale of Sahli’s tube.
- Pricknger under sterile conditions and draw blood.
- Then introduce this blood into e graduated tube immediately into the pipette up to the mark 20cmm and empty the pipette till it is completely free of blood.
- Mix it with HQ already present, with a stirrer, and wait for 10 mins.
- Acid haematin is formed and shows grew (clear) color. Add distilled water till the color approaches that of the standards.
- Take the reading on the graduated tube as gm/lOOml of blood. The color matching should be against natural light and reading noted at the lower meniscus.
- The color of the comparator is based on the British Hematological standards called Gibson’s Harrison’s color is obtained with picric acid tartrate. But a disadvantage is that the color fades after a long time.
Precautions:
- Wait for at least 10 min for the formation of acid haematin.
- (Because 95% of it is formed in 10 min and 98% in 20 mins).
- Remove the stirrer while adding distilled water and also while taking the reading, Tap water cannot be used as the pH varies & accurate values may not be obtained, which also causes turbidity.
- Degree of error: 10-15%
- Strong oxidizing agents like HNO3 or PI2SO4 cannot be used in place of HC1.
- Hb cone is lower in females because estrogens have an inhibitory effect on the secretion of EP while the androgens in males have a stimulatory effect.
- Newborns have high Hb cone because they are in a state of relative hypoxia.
- If Hb is present freely in plasma, it would increase the viscosity of blood, (thus increasing BP) and the osmotic pressure.
Normal values:
- Males – 13 to 16 gms%
- Females – 12 to 15 gms%
Course Of Discussion
- Collection of blood for estimation of hemoglobin:
- Blood may be collected from a finger prick or from the vein (venous blood).
- Blood collected in EDTA or double oxalate in appropriate proportion may also be conveniently used for the estimation of hemoglobin.
Possible shortcomings or drawbacks of acid haematin method:
- Acid haematin is unstable.
- The color fades away immediately after reaching the peak concentration.
- The color intensity has to be matched with the Hie brown color of the comparator box visually.
- There is no international standard for this brown-colored comparator.
- There is more chance of personal error as it is a visual method.
- Not all types of hemoglobin are converted to acid haematin, e.g. Sulphaemoglobin, methemoglobin.
Possible sources of errors in the estimation of hemoglobin:
The sources of errors are:
- Errors of sampling including the presence of microclot in EDTA/ oxalated blood due to inadequate mixing.
- There may be excess tissue fluid diluting the capillary blood due to squeezing in the finger prick method.
- While diluting the blood.
- Degree of error: 10-15%
- Strong oxidizing agents like HNO3 or H2SO4 cannot be used in place of HC1.
- Hb cone is lower in females because estrogens have an inhibitory effect on the secretion of EP while the androgens in males have a stimulatory effect.
- Newborns have high Hb cone because they are in a state of relative hypoxia.
- If Hb is present freely in plasma, it would increase the viscosity of blood, (thus increasing BP) and the osmotic pressure.
Normal values:
- Males – 13 to 16 gms%
- Females – 12 to 15 gms%
Courses Of Discussion
Collection of blood for estimation of hemoglobin:
- Blood may be collected from a finger prick or from a vein (venous blood).
- Blood collected in EDTA or double oxalate in appropriate proportion may also be conveniently used for the estimation of hemoglobin.
- Possible shortcomings or drawbacks of acid haematin method:
Acid haematin is unstable. - The color fades away immediately after reaching the peak concentration.
- The color intensity has to be matched with the brown color of the comparator box visually.
- There is no international standard for this brown-colored comparator.
- There is more chance of personal error as it is a visual method.
- Not all types of hemoglobin are converted to acid haematin, e.g. Sulphaemoglobin, methemoglobin.
Possible sources of errors in the estimation of hemoglobin:
The sources of errors are:
- Errors of sampling including the presence of microclot in EDTA/oxalated blood due to inadequate mixing.
- There may be excess tissue fluid diluting the capillary blood due to squeezing in the finger prick method.
- While diluting the blood.
- In the estimation of color intensity.
- Improperly lysed RBC, nucleated red cells, para-proteins, and lipids may give erroneously high results.
- Principle and the procedure of estimation of hemoglobin by Cyanmethemoglobin method.
Principle:
When blood is mixed with a solution containing potassium ferricyanide and potassium cyanide, all types of hemoglobins, except Sulphaemoglobin, are converted to cyanmethemoglobin, which is measured at 540 nm in a spectrophotometer or green filter in a photoelectric colorimeter, against a standard.
Method:
Instruments required:
- Haemoglobin pipette,
- Small test tubes, stirrer, dropper, etc.
- Spectrophotometer or colorimeter.
Reagents required:
- Drabkin’s solution – Pot ferricyanide 200 mg
- Pot cyanide 50 mg
- Pot dihydrogen phosphate 140 mg
- Non-ionic detergent 1 ml
- Distilled water up to 1 liter.
- Standard solution.
- 20 microlitres, of blood, is added to 4 ml (1: 200) or 5 ml (1: 250) of Drabkin’s solution (12 gm/dl for dilution of 1: 200; 15 gm/ dl for dilution of 1: 250) in colorimeter with the green filter of a spectrophotometer at 540 nm.
- Measurement of hemoglobin measured by acid hematin method when hemoglobin is too low:
- When hemoglobin is too low, less than 20% or 3 gm/dl, 40 microlitres of blood is added o HC1 up to 20 marks in the tube.
- The color is matched and the result is halved.
- Conditions where hemoglobin is falsely raised or normal;
Burns, severe dehydration due to haemoconcentration. - Immediately after acute hemorrhage.
- If the blood is taken during the intravenous infusion of iron-containing drugs.
Total WBC Count
Principle: A sample of blood is diluted with a diluting fluid, which destroys RBC & stains the nuclei of WBC.
These cells are then counted in a hemocytometer and then the number is reported as WBC/mm3.
Procedure:
- First focus on one peripheral square under low power & then to high power to study the WBC. Place the cover slip on the ruled area.
- Under aseptic conditions give a prick to the pulp of the distal phalanx of the index finger.
- Suck blood up to 0.5 marks in the WBC pipette. Suck the Thomas fluid up to 11 marks & mix well. Dilution is 1 in 20.
- Charging and counting are similar to that of RBC, except for counting done in the squares mentioned for WBC.
Thomas fluid:
- Glacial acetic acid -1.5 ml (to lyse RBC)
- Gentian violet -1.5 ml (for staining WBC nuclei)
- D. H2O – 100 ml
Measurements:
- Area of 1 square = 1 mm2
- The volume of 1 square = 0.1mm3
- No. of cells in 4 squares = X
- No. of cells in 1 square = X/4
- No. cells in 1mm of dil. blood = X/4 x 10
- Dilution factor = 1 in 20
- Calculations: Let the cells counted be ‘X
- Total no. of cells = No. of cells counted x dilution factor x depth = X/4 x 20 x 10
- No. of WBC / mm3 = 50 x X /mm3- Normal value = 4,500 -11,000 mm3
Course Of Discussion
Complete blood count:
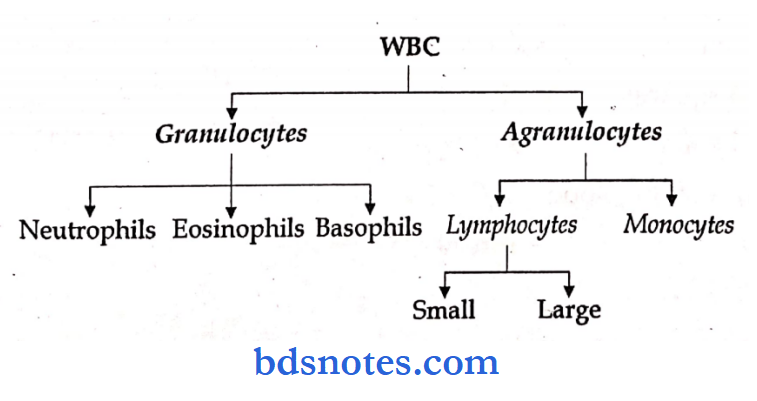
Complete blood count includes:
- Total leucocyte count – TLC
- Differential leucocyte count – DLC
- Red blood cell count
- Hemoglobin percentage
- Hematocrit estimation
- RBC indices
- Platelet count and
- Peripheral blood smear examination.
Lazy leucocyte syndrome:
- This condition can be congenital or acquired affecting the mobilization of neutrophils.
- The blood may show severe neutropenia but the marrow shows the normal number of granulocytes and their precursors.
Differential Count Of WBC
Principle: A stained blood film is examined under oil immersion and different types of leucocytes are identified until 200 cells have been examined.
The percentage distribution of each type of leucocyte is determined. Knowing the total leucocytic count the absolute number of each type then be calculated.
Procedure: A drop of blood is taken on a slide in one corner.
Hold another glass slide at an angle of 45° beyond the blood drop.
After touching the drop, push it quickly toward the other end to make a thin even smear.
The smear is dried and adds 8 drops of Leishman’s stain & wait for 2 min. Dilute with double the quantity of buffered water & wait for 7-10 min.
After this flood off the stain with distilled water, drain & air-dry it. Then observe under low power.
The stain used is Leishman’s stain which contains 0.15 gms of Leishman powder in 100 ml of acetone-free methyl alcohol.
Leishman powder consists of methylene blue, which stains the nucleus pink and the cytoplasm blue.
The functions of acetone-free methyl alcohol are:
- Acts as fixative
- Maintain neutral pH
- Prevents hemolysis
- Increases longevity of a stain.
- Examination under oil immersion: Place a drop of cedar wood oil and examine under the oil immersion objective.
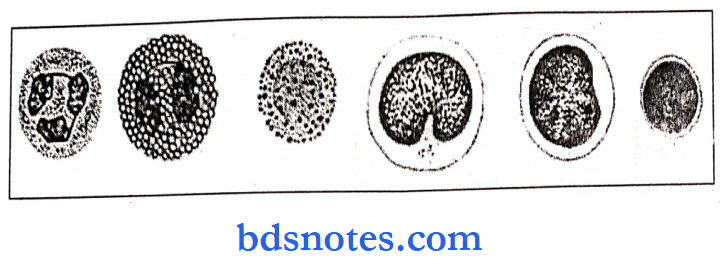
Three types of cells are seen:
- RBC
- WBC
- Platelets
- Counting of WBC is done in a zigzag or strip method.
- A count of 100 WBC is done
Procedure:
- Under aseptic conditions, give a deep finger prick and note the time.
- Then remove the blood drop every 15 sec with a clotting paper (along the edges) at a different spot till the bleeding stops. Count the number of spots.
- Normal bleeding time; 1 to 5 min.
- BT is prolonged in purpura (since thrombocytopenia) while normal in hemophilia.
Bleeding time depends on:
- Platelet count
- Quality of platelets
- The integrity of the capillary wall
Other methods are:
- IVY bleeding time method
- Clotting Time Glass Capillary Tube Method
- Defined as the time taken by the blood to clot after collecting from the body. It ranges between 3 – 6 min.
Apparatus: Dry capillary tube, 1mm diameter & 15 cm long.
Procedure: Under aseptic conditions, get a deep finger prick; discard the first 2- 3 drops of blood (due to the presence of thromboplastin).
Once a drop of blood is formed fill the capillary tube by dipping inclinedly in the drop (fills the tube by capillary action).
Note the time. A small bit of the capillary tube is carefully broken off every fifteen sec.
until a fine thread of clotted blood appears while the die tube is being broken.
Note the number of pieces. (Clotting is by both intrinsic & extrinsic methods).
Clotting time = (Number of pieces – 1) x 15 + initial time. = (Number of pieces -l)5 + 2 min.
Clotting time prolonged in:
- Hemophilia
- All conditions of low platelet count like:
- Splenomegaly
- X-ray irradiation
- Aplastic anemia
- Pernicious anemia
Other methods:
- Drop method
- Lee and white test tube method
- CT & BT are carried out when an individual complains of frequent & persistent bleeding, before minor or major surgeries or positive family history.
- BT is the time from the onset of bleeding to the stoppage of bleeding. The bleeding stops due to the formation of a temporary hemostatic plug.
- CT is the time taken from the onset of bleeding to the formation of the definitive hemostatic plug (clot).
- Temporary hemostatic plug formation occurs earlier than the definitive hemostatic plug.
- Therefore, normally, CT is more than BT.
Blood Groups
- Blood Groups – discovered by “Land Steiner”
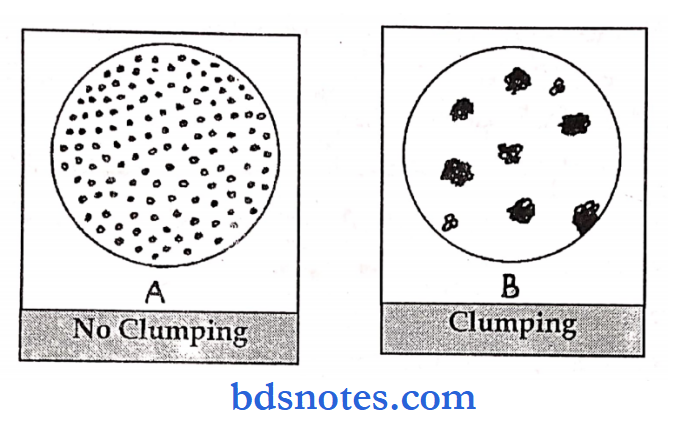
Principle: Blood typing is done on the basis of agglutination.
RBC contains a series of antigens on their cell membranes while plasma contains antibodies.
To determine the blood group, the RBC is made to react with sera containing known agglutinins.
The slide is then examined under the microscope to detect the presence or absence of agglutination & hemolysis of RBC.
Agglutination occurs if an antigen is mixed with its corresponding agglutinin (Iso-agglutinin).
Procedure:
Put a drop of antiserum A on one end & antiserum B on the other end of a glass slide.
Give a prick on the pulp of the left index finger and make a suspension with the blood.
A drop of RBC suspension is mixed with each serum. The slide is slightly moved for 2 min. The presence or absence of agglutination is observed.
Clumping: Thick masses of RBC are seen.
Absence of clumping: The mixture is clear with separate cells.
Results
If agglutination occurs with antisera A – blood group A
If agglutination occurs with antisera B . – blood group B
If agglutination occurs with both antisera A & B – blood group AB.
If agglutination does not occur with both antisera A & B – blood group O.
During Blood transfusion, only compatible blood should be used.
Erythrocyte Sedimentation Rate
ESR: If a sample of blood is mixed with an anticoagulant and allowed to stand in a vertical tube, the RBC settles down due to gravity (denser, specific gravity -1.095) as compared to plasma (Specific gravity 1.032) with a clear supernatant layer of plasma. The rate at which the cells settle down is called ESR.
Precautions: Add proper anticoagulant, and do the test at room temperature and within 12 hrs of drawing blood. Do not disturb the tubes for 1 hour.
Definition: It is the volume of red cells expressed as a percentage of the volume of whole blood in the sample. Hematocrit literally means separation.
Plasma layer Methods of estimation: Buffy coat
- Using Wintrobe’s tube, blood is centrifuged for half an hour at 3000 RPM.
- Using a capillary tube
Observation: Three layers are observed after centrifugation:
Plasma layer: Top most, clay yellow or straw colored.
Buffy cost: middle layer of WBC and platelets About 1mm Thick. Platelets from a thin Layer over WBC.
- column of RBC: Bottom Layer.
- Abnormal Constituents Of Urine
- Urine Analysis: General considerations:
- Normal values
- Color: Straw color
- Turbidity: Clear
- Average pH: 6.0
- Specific Gravity: 1.012 -1.024
- Average volume excreted in 24 hours: 1.5 liters
- Composition of urine in 24 hours
- Solids: 60 gms (40gms. per lit)
- Water: 1200 gms
- Inorganic constituents:
- Sodium chloride 15 gms
- Calcium 0.2
- Phosphates 3.0 gms
- Sulfates 2.5 gms
- Potassium 3.0 gms
- Nitrogenous constituents
- Urea: 20-30gms
- Uric Acid: 0.7-1.0gms
- Ammonia:0.6-0.7gms
- Creatinine: 15-0gms
The Pathological Alternations:
- They are proteins, blood glucose, ketone bodies, Bile Pigments, and bile salts
Principle: All methods depend on the precipitation of proteins by chemical agents or coagulation by heat.
Causes of proteinuria:
Accidental: Due to contamination by pus, vaginal fluid, seminal fluid, or blood.
Functional: Pregnancy, infancy, after exercise, exposure to cold, postural proteinuria.
Pre-Renal: No primary renal disease, like CHF, Cerebral injury, fever, toxemia, ascites, intra-abdominal tumors, drugs, severe anemia, or plasma cell myeloma.
Renal: Nephritis, DM, SLE, TB kidney, Amyloidosis, HTN, Carcinoma kidney, Nephrotic syndrome.
Benzidine reagent: Mixture of equal quantities of Reagent A.
- Saturated solution of benzidine in glacial acetic acid Reagent B.
- Freshly prepared H2O2
Principle: The test is very sensitive.
- Hb is a complex substance and peroxidase activity of Hb decomposes H202 and the liberated nascent 02 oxidizes benzidine to give blue color.
- Blood may be found in urine as RBC – [Hematuria] or as pigments – [Hemoglobinuria]
Causes of Hematuria: Lesions in the kidney, cystitis, AGN, Nephrolithiasis, Urethrirtis, Renal TB, malignant Nephrosclerosis, Neoplasms of the kidney, lesions of the lower urinary tract, stones in the bladder, Bilharziasis.
Advantages: Simple Uses a more Stable reagent, more sensitive and specific
Causes of Glycosuria:
- Diabetes Mellitus
- Renal glycosuria
- Hyperthyroidism
- Intracranial injury
- Alimentary glycosuria
- Pancreatitis
- Pregnancy
- Renal tubular disease
- Severe burns and sepsis
- Glucagon secreting tumors of islets of the pancreas
- Ketone Bodies
Principle: In the presence of (NH4)2S04 and liquor NH4 CH nitroprusside reacts with acetoacetate or acetone and gives permanganate colored ring.
Causes of ketonuria:
- Infants & children with acute febrile conditions
- Toxic states, Cachexia.
- Vomiting of pregnancy,
- Severe exercise,
- Following exposure to cold.
Leave a Reply