Molecular Representations And Nomenclature Molecular representations
From the beginning of chemistry, scientists have devised means of representing the materials they are discussing and have gradually developed a comprehensive range of shorthand notations.
These cover the elements themselves, the bonding between atoms, the arrangement of atoms in molecules, and, of course, a systematic way of naming compounds that are accepted and understood throughout the scientific world.
The study of carbon compounds provides us with the subdivision ‘organic chemistry’, and a few simple organic compounds can exemplify this shorthand approach to molecular representations.
The primary alcohol propanol (systematically propane-1-ol or 1- propanol, formerly n-propanol, n signifying normal or unbranched) can be represented by a structure showing all atoms, bonds, and lone pair or nonbonding electrons.
Lines are used to show what we call single bonds, indicating the sharing of one pair of electrons.
In writing structures, we have to remember the number of bonds that can be made to a particular atom, i.e. the valency of the atom.
In most structures, carbon is tetravalent, nitrogen trivalent, oxygen divalent, and hydrogen and halogens are univalent. These valencies arise from the number of electrons available for bonding.
More often, we trim this type of representation to one that shows the layout of the carbon skeleton with attached hydrogens or other atoms.
This can be a formula-like structure without bonds, or it can be one showing just the principal bonds, those of the carbon chain.
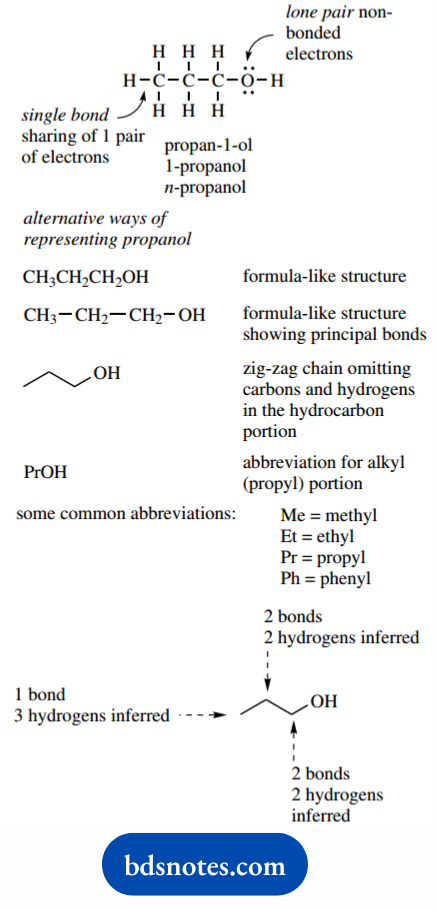
However, for many complex structures, even these approaches become too tedious, and we usually resort to a shorthand version that omits most, if not all, of the carbon and hydrogen atoms.
Propanol is now shown as a zig-zag chain with an OH group at one end. The other end of the chain, where it stops, is understood to represent a methyl group; three attached hydrogens have to be inferred.
At a point on the chain, two hydrogens are assumed, because two bonds to carbons are already shown. In a structure where three bonds joined, a single additional hydrogen would be assumed (see vinyl chloride, below).

Double bonds, representing the sharing of two pairs of electrons, are inferred by writing a double line. Vinyl chloride (systematically chloroethene) is shown as two different representations according to the conventions we have just seen for propanol.
Note that it is customary always to show the reactive double bond so that CH2CHCl would not be encountered as an abbreviation for vinyl chloride.
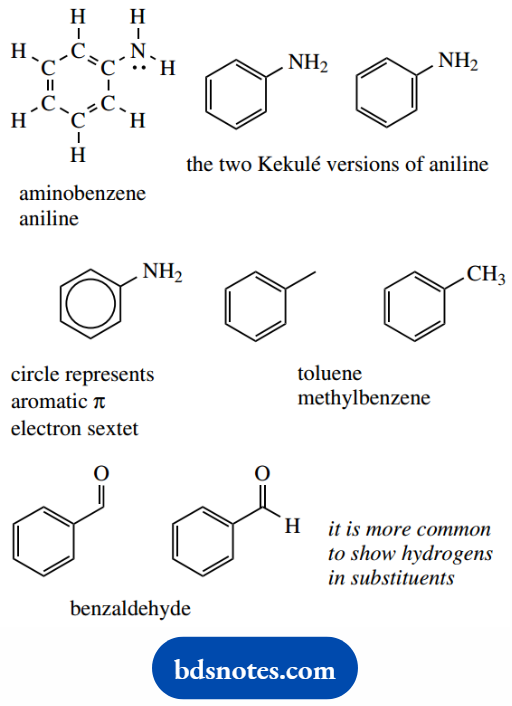
The six-membered cyclic system in aromatic rings is usually drawn with alternating double and single bonds, i.e. the Kekule form´, and it is usually immaterial which of the two possible versions is used.
Aniline (systematically aminobenzene or benzenamine) is shown with and without carbons and hydrogens. It is quite rare to put in any of the ring hydrogens on an aromatic ring.
However, it is sometimes convenient to put some in on the substituent, For Example. on a methyl, as in toluene (methylbenzene), or an aldehyde group, as in benzaldehyde.
Benzene strictly does not have alternating double and single bonds, but the aromatic sextet of electrons is localized in a π orbital system and bond lengths are somewhere in between double and single bonds.
The zig-zag arrangement is convenient so that we see where carbons are located (a long straight line would not tell us how many carbons there are).
But it also mimics the low-energy arrangement (conformation) for such a compound. Note that it is usual to write out the hydroxyl, or some alternative group, in full.
This group, the so-called functional group, tends to be the reactive part of the molecule that we shall be considering in reactions.
When we want an even more concise method of writing the molecule, abbreviations for an alkyl (or aryl) group may be used, in which case propanol becomes PrOH. Some more common abbreviations are given later.
To represent this, a circle may be drawn within the hexagon. Unfortunately, this version of benzene becomes quite useless when we start to draw reaction mechanisms.
And most people continue to draw benzene rings in the Kekule form. In some cases, such as fused rings, it is actually incorrect to show the circles.
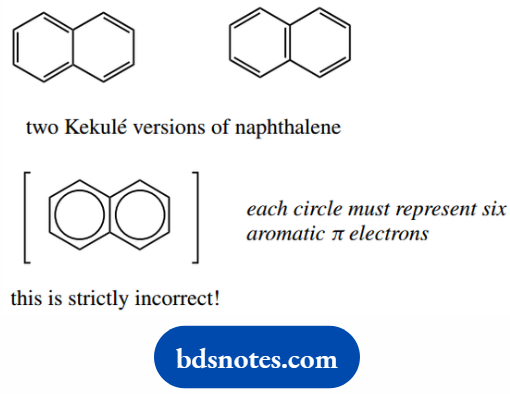
Thus, naphthalene has only 10 π electrons, one from each carbon, whereas the incorrect two-circle version suggests it has 12 π electrons.
We find that, in the early stages, students are usually happier to put in all the atoms when drawing structures, following earlier practices.
However, you are urged to adopt the shorthand representations as soon as possible. This saves time and cleans up the structures of larger molecules.
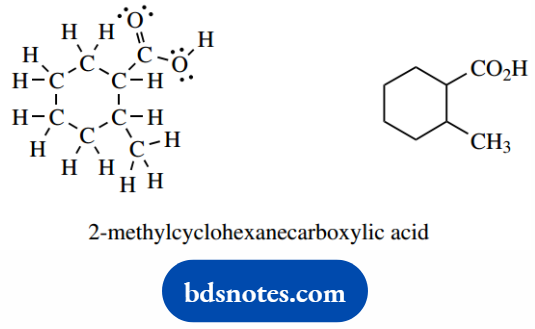
Even a relatively simple molecule such as 2-methylcyclohexane carboxylic acid, a cyclohexane ring carrying two substituents, looks like a mess when all the atoms are put in.
By contrast, the line drawing looks neat and tidy and takes much less time to draw.
Do appreciate that there is no strict convention for how you orientate the structure on paper. In fact, we will turn structures around, as appropriate, to suit our needs.
For example, the amino acid tyrosine has three functional groups, i.e. a carboxylic acid, a primary amine, and a phenol.
How we draw tyrosine will depend upon what modifications we might be considering, and which functional group is being altered.
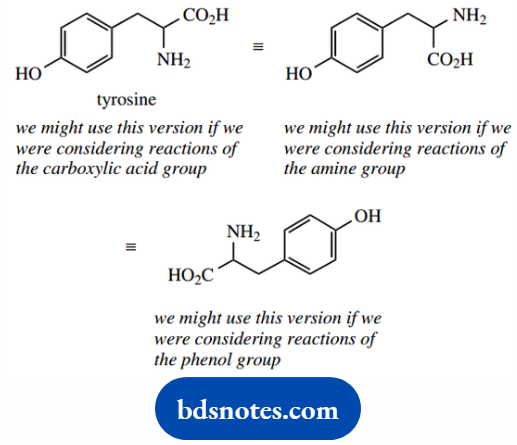
You will need to be able to reorientate structures without making mistakes, and also to be able to recognize different versions of the same thing.
A simple example is with esters, where students have learned that ethyl acetate (ethyl ethanoate) can be abbreviated to CH3CO2C2H5.
When written backwards, i.e. C2H5OCOCH3, the ester functionality often seems less recognizable.
Partial Structures
We have just seen that we can save a lot of time and effort by drawing structures without showing all of the atoms. When we come to draw reaction sequences.
We shall find that we have to repeat large chunks of the structure each time, even though no chemical changes are occurring in that part of the molecule.
This is unproductive, so we often end up writing down just that part of the structure that is of interest, i.e. a partial structure.
This will not cause problems when you do it, but it might when you see one and wish to interpret it.
In the representations overleaf, you can see the line drawing and the version with methyls that stresses the bond ends.
Both are satisfactory. When we wish to consider the reactivity of the double bond, and perhaps want to show that reaction occurs irrespective of the alkyl groups attached to the double bond.
We put in the abbreviation R (see below), or usually just omit them. When we omit the attached groups.
It helps to show what we mean by using wavy lines across the bonds, but in our urge to proceed, we tend to omit even these indicators.
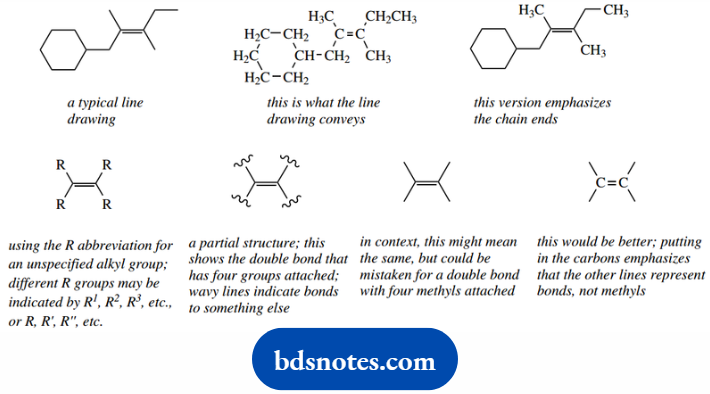
This may cause confusion in that we now have what looks like a double bond with four methyls attached, not at all what we intended.
A convenient ploy is to differentiate this from a line drawing by putting in the alkene carbons.
Functional groups
The reactivity of a molecule derives from its functional group or groups.
In most instances, the hydrocarbon part of the molecule is likely to be unreactive, and the reactivity of the functional group is largely independent of the nature of the hydrocarbon part.
In general terms, then, we can regard a molecule as R–Y or Ar–Y, a combination of a functional group Y with an alkyl group R or aryl group Ar that is not participating in the reaction under consideration.
This allows us to discuss reactivity in terms of functional groups, rather than the reactivity of individual compounds.
Of course, most of the molecules of interest to us will have more than one functional group; it is this combination of functionalities that provides the reactions of chemical and biochemical importance.
Most of the functional groups we shall encounter are included, which also contain details for their nomenclature.
It is particularly important that when we look at the structure of a complex molecule we should visualize it in terms of the functional groups it contains.
The properties and reactivity of the molecule can generally be interpreted in terms of these functional groups.
It may sometimes be impossible to consider the reactions of each functional group in complete isolation, but it is valuable to disregard the complexity and perceive the simplicity of the structure.
With a little practice, it should be possible to dissect the functional groups in complex structures such as morphine and amoxicillin.
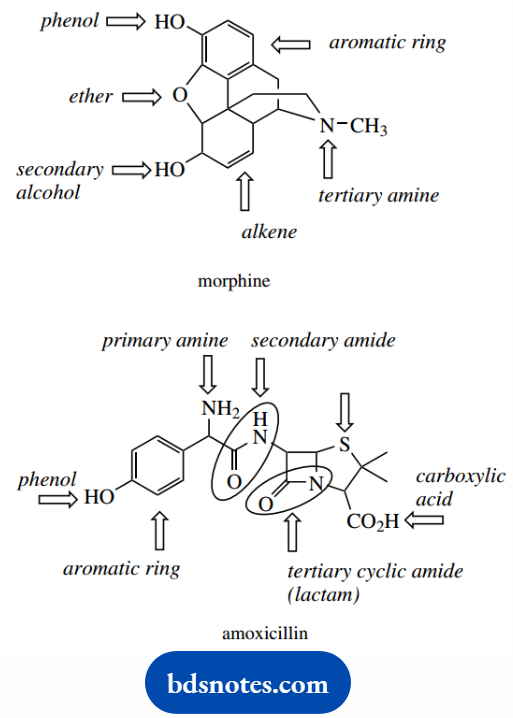
Systematic nomenclature
Organic compounds are named according to the internationally accepted conventions of the International Union of Pure and Applied Chemistry (IUPAC).
Since these conventions must cover all eventualities, the documentation required spans a book of similar size to this volume. A very much-abbreviated version suitable for our requirements is given here:
- The functional group provides the suffix name;
- With two or more functional groups, the one with the highest priority provides the suffix name;
- The longest carbon chain containing the functional group provides the stem name;
- The carbon chain is numbered, keeping minimum values for the suffix group;
- Side-chain substituents are added as prefixes with appropriate numbering, listing them alphabetically.
The stem names are derived from the names of hydrocarbons. Acyclic and cyclic saturated hydrocarbons (alkanes) in the range C1 –C12 are listed.
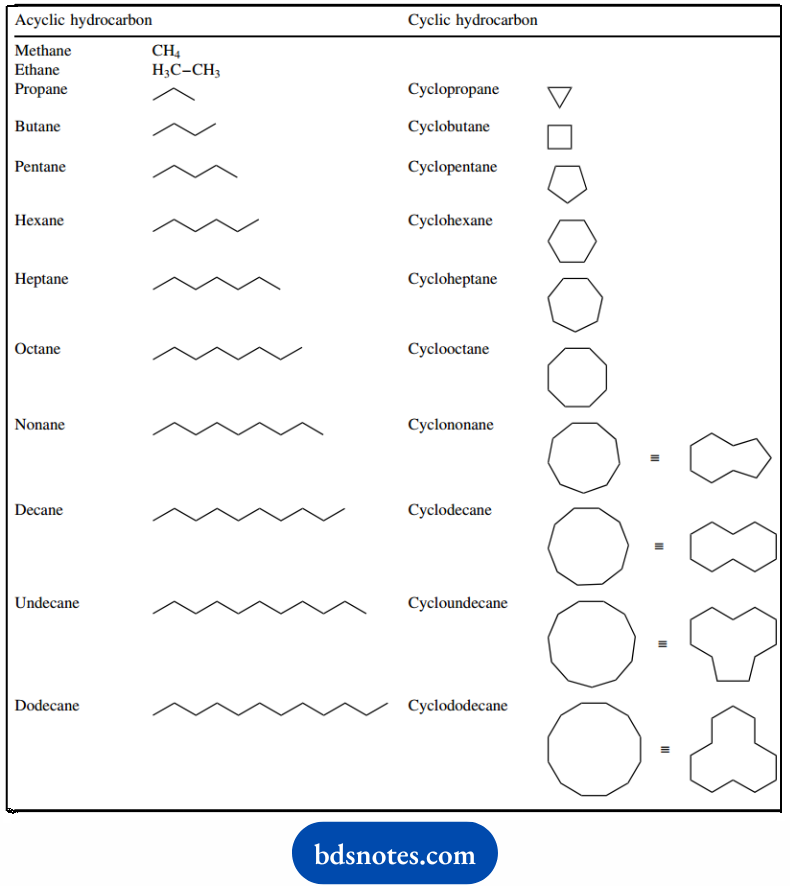
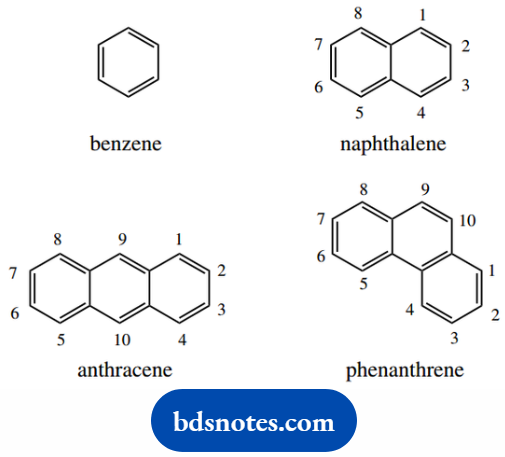
Aromatic systems are named in a similar way, but additional stem names need to be used. Parent aromatic compounds of importance are benzene, naphthalene, anthracene, and phenanthrene.
The last three contain fused rings, and they have a fixed numbering system that includes only those positions at which substitution can take place.
It is anticipated that readers will already be familiar with many of the general principles of nomenclature and will be able to name a range of simple compounds.
It is not the object of this section to provide an exhaustive series of instructions for naming every class of compound.
Instead, the examples chosen here have been selected to illustrate some of the perhaps less familiar aspects that will be commonly encountered and to foster a general understanding of the approach to nomenclature.
Alternative names are shown in some cases; this should emphasize that there is often no unique ‘correct’ name.
Sometimes, it can be advantageous to bend the rules a little so as to provide a neat name rather than a fully systematic one.
Typically, this might mean adopting a lower-priority functional group as the suffix name.
It is important to view nomenclature as a means of conveying an acceptable unambiguous structure rather than a rather meaningless scholastic exercise.
Other examples and specialized aspects will occur in subsequent chapters. For example, heterocyclic nomenclature will be treated in more detail at the appropriate time.
Stereochemical descriptors are omitted here but will be discussed under stereochemistry.
Systematic nomenclature: some example
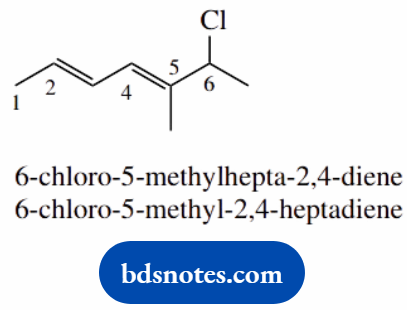
- alkenes have higher priority than halides; the suffix is -ene
- longest carbon chain is seven carbons: heptane
- numbering is chosen to give the lowest numbers for the double bonds; 2-ene denotes 2,3-double bond, 4-ene denotes 4,5-double bond
- the European system hepta-2,4-diene is less prone to errors than the US system 2,4-heptane
- an additional syllable -a- is used but is not obligatory; heptadiene is easier to say than heptdiene
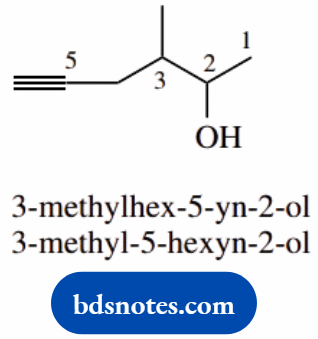
- Alcohols have higher priority than alkynes; the suffix is -ol
- The longest carbon chain is six carbons: hexane
- Numbering is chosen to give the lowest number for alcohol
- The European system hex-5-yn-2-ol keeps numbers and functionalities together
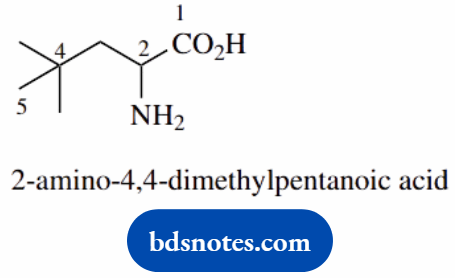
- Acids have higher priority than amines; remember ‘amino acids’
- The suffix is -oic acid
- One of the methyls is part of the five-carbon chain, the others are substituents
- Note the use of 4,4-, which shows both methyls are attached to the same carbon; 4-dimethyl would not be as precise
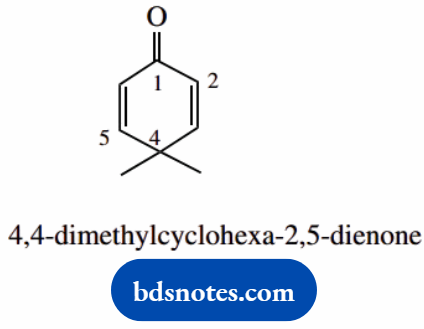
- The highest priority group is a ketone; suffix -one
- The longest carbon system is the ring cyclohexane numbering is around the ring starting from ketone as position 1
- 2,5-diene conveys 2,3- and 5,6-double bonds
- Note 2,5-dienone means two double bonds and one ketone; in contrast, endzone which would be one double bond and two ketones
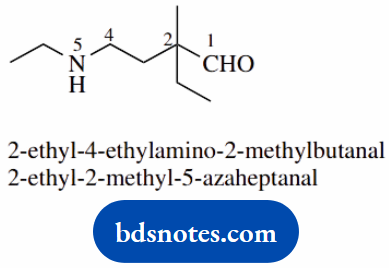
- The highest priority group is aldehyde; suffix -al
- The amino group at 4 is also substituted; together they become ethylamine
- The alternative name invokes a seven-carbon chain with one carbon (C-5) replaced by nitrogen; this is indicated by using the extra syllable -aza-, so the chain becomes 5-azaheptane
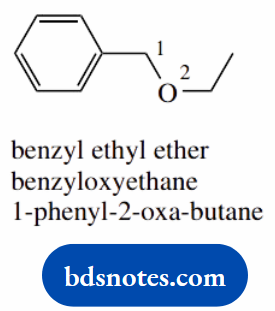
- Simple ethers are best named as an alkyl alkyl ether
- The phenylmethyl group is commonly called benzyl
- An acceptable alternative is as an alkoxy alkane: the alternative ethoxy toluene would require an indication of the point of attachment
- The second alternative invokes a three-carbon chain with one carbon replaced by oxygen; this is indicated by using the extra syllable -oxa-, so the chain becomes 2-oxabutane
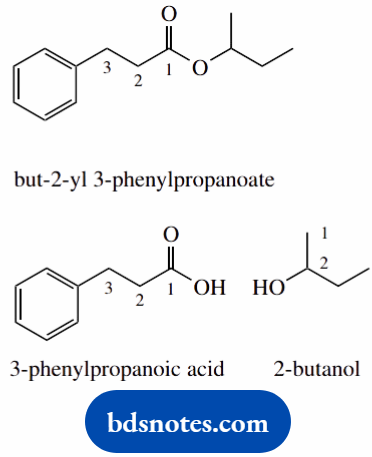
- Esters are named alkyl alkanoate – two separate words with no hyphen or comma
- Alkyl signifies the alcohol part from which the ester is constructed, whilst alkanoate refers to the carboxylic acid part
- but-2-yl means the ester is constructed from the alcohol butan-2-ol; 3-phenylpropanoate means the acid part is 3-phenylpropanoic acid
- Note the numbers 2 and 3 are in separate words and do not refer to the same part of the molecule
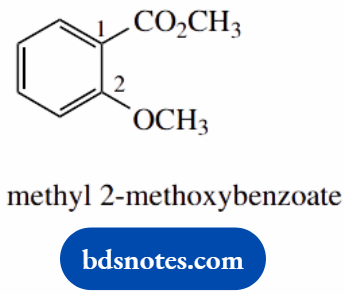
- This is a methyl ester of a substituted benzoic acid; the ring is numbered from the point of attachment of the carboxyl
- The acid portion for the ester is 2-substituted
- The ether group is most easily treated as a methoxy substituent on the benzene ring
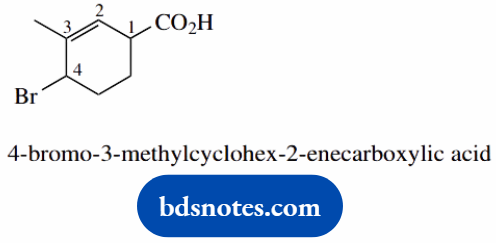
- The carboxylic acid takes priority; suffix usually-oic acid
- The carboxylic acid is here treated as a substituent on the cyclohexane ring; the combination is called cyclohexanecarboxylic acid
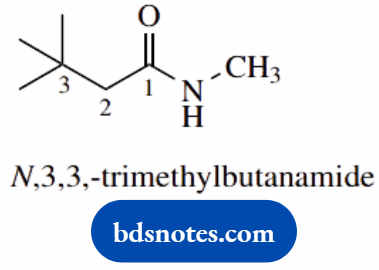
- This is a secondary amide of butanoic acid; thus the root name is butanamide
- Two methyl substituents are on position 3, and one on the nitrogen, hence N,3,3-trimethyl; the N is given in italics
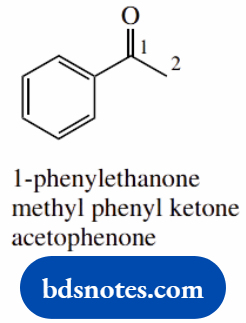
- A ketone in which the longest chain is two carbons; thus the root name is methanone
- The phenyl substituent is on the carbonyl, therefore at position 1
- Without the 1-substituent, ethanone is actually an aldehyde, and would be ethanal!
- The alternative methyl phenyl ketone is a neat and easy way of conveying the structure
- This structure has a common name, acetophenone, which derives from an acetyl (CH3CO) group bonded to a phenyl ring
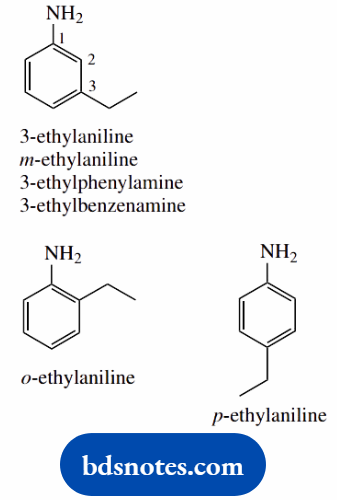
- An amine; suffix usually -amine
- The root name can be phenylamine, as an analog of methylamine, or the systematic benzydamine; in practice, the IUPAC accepted name is aniline
- The ring is numbered from the point of attachment of the amino group
- The prefixes ortho-, meta-, and para- are widely used to denote 1,2-, 1,3-, or 1,4-arrangements respectively on an aromatic ring; these are abbreviated to o-, m-, and p-, all in italics
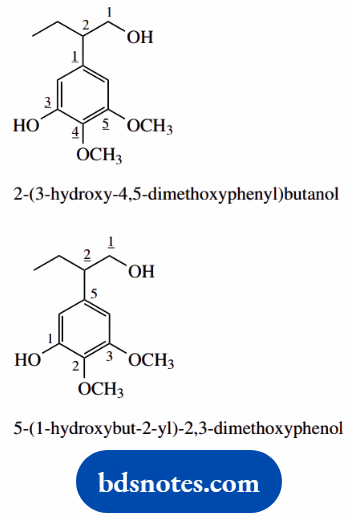
- This could be named as an alcohol, or as a phenol
- As an alcohol (butanol), there is a substituted phenyl ring attached at position 2
- Note the phenyl and its substituents are bracketed to keep them together and to separate their numbering (shown underlined) from that of the alcohol chain
- As a phenol, the substituted butane side-chain is attached through its 2-position so has a root name but-2-yl to show the position of attachment; again, this is in brackets to separate its numbering from that of the phenol
- di-, tri-, tetra-, etc. are not part of the alphabetical sequence for substituents; dimethoxy comes under m, whereas trihydroxy would come under h, etc.
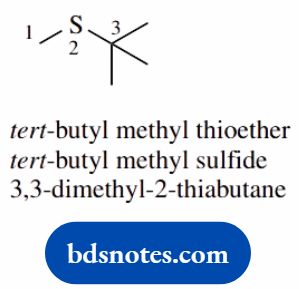
- This is a thioether, which can be named as a thioether or as a sulfide
- An alternative invokes a four-carbon chain with one carbon replaced by sulfur using the extra syllable -thia-; this chain thus becomes 2-thiabutane
- Note how the (trimethyl)methyl group is most frequently referred to by its long-established name of tertiary-butyl, abbreviated to tert-butyl, or t-butyl
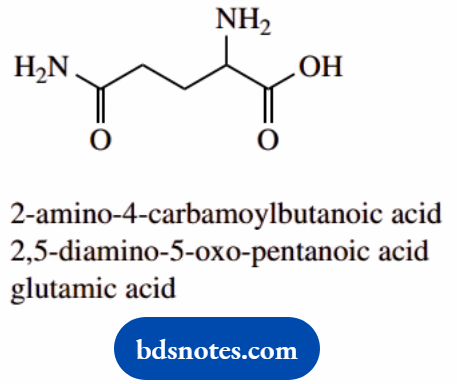
- This contains an amine, an amide, and a carboxylic
- The amide group as a substituent is termed carbamoyl;
- It is rather easier to consider the amide as amino and
- The ketone is indicated by oxo-; do not confuse this
- The common name is glutamic acid; it is an amino acid found in proteins
- There now follow several examples demonstrating how to convert a systematic name into a structure, with appropriate guidance hints.
- For added relevance, these are all selected from routinely used drugs. Again, any stereochemical aspects are not included.
Converting systematic names into structures: selected drug molecules
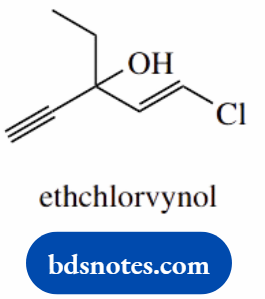
1-chloro-3-ethylpent-1-en-4-yn-3-ol (ethchlorvynol)
Main chain is pentane (C5) number it ![]()
Put in unsaturation 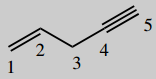
- 1-ene (=1,2-ene)
- 4-yne (=4,5-yne)
Put in substituents 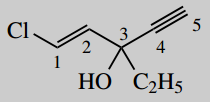
- 1-chloro
- 3-ethyl
- 3-hydroxy (3-ol)
4-aminohex-5-enoic acid (vigabatrin)
The main chain is hexane (C6) number it 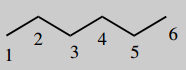
Put in unsaturation 5-ene (=5,6-ene) 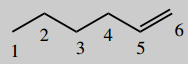
A main functional group is an acid (-oic acid) this will be carbon-1 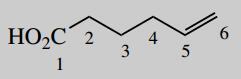
Put in substituent 4-amino 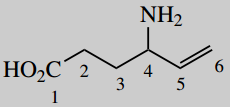
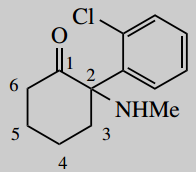
2-(2-chlorophenyl)-2-methylaminocyclohexanone (ketamine)
The main chain is cyclohexane (C6) number it 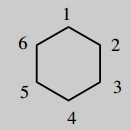
A main functional group is a ketone (-one) which will be carbon-1 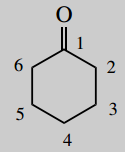
Put in substituents 2-methylamino = 2-amino carrying a methyl (contrast aminomethyl = methyl carrying an amino) 2-(2-chlorophenyl) = 2-chlorophenyl at position 2; the phenyl carries a chloro substituent at its own position 2; note the use of brackets to separate the two types of numbering
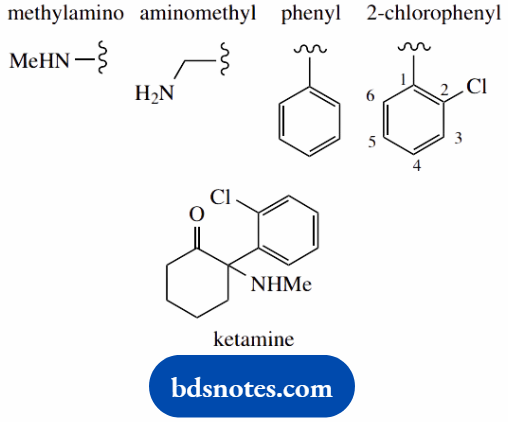
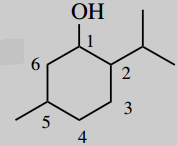
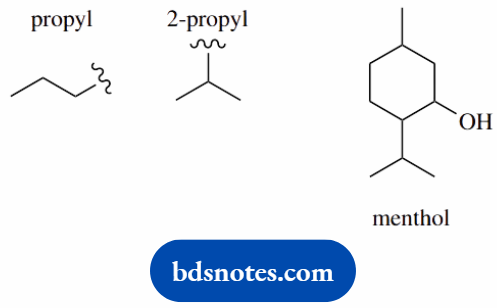
5-methyl-2-(2-propyl)-cyclohexanol (menthol)
Main chain is cyclohexane (c6) number it 
Main functional group is an alcohol (-ol) this will be carbon-1 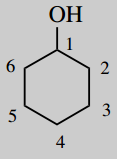
put substituents 5-methyl 2-(2-propyl)=2-propyl position 2;
2-propyl is a peopyl group joined via its 2-position
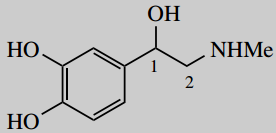
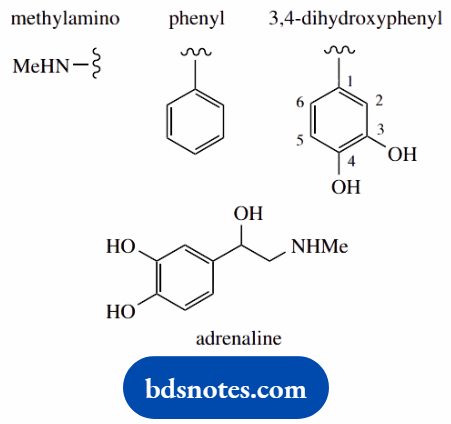
1-(3,4-dihydroxyphenyl)-2-dimethylaminoethanol (adrenaline; epinephrine)
Main chain is ethane (C2) number it 
Main functional group is an alcohol (-ol) this will be carbon-1 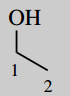
Put in substituents 2-methylamino = 2-amino carrying a methyl 1-(3,4-dihydroxyphenyl) = 3,4-dihydroxyphenyl at position 1; the phenyl carries hydroxy substituents at its own positions 3 and 4; note the use of brackets to separate the two types of numbering
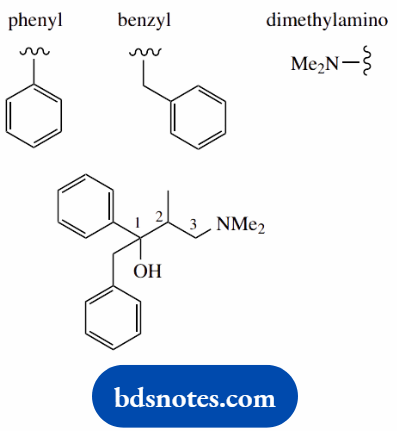
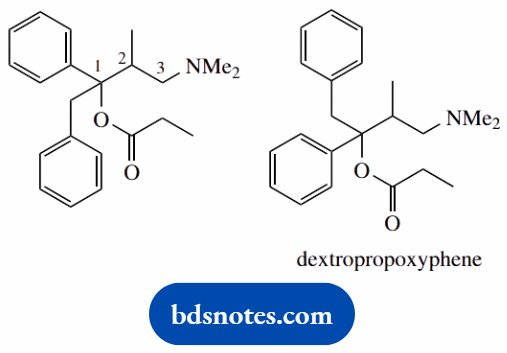
1-benzyl-3-dimethylamino-2-methyl-1-phenylpropyl propionate (dextropropoxyphene)
This is an ester (two words, -yl -oate) the -oate part refers to the acid component, the -yl part to the esterifying alcohol
Main chain of acid is propane (C3) main chain of alcohol is propane (C3)
these are numbered separately (the ester has two separate words)
No substituents on acid component
Put in substituents on alcohol components 1-phenyl; 1-benzyl; 2-methyl; 3-dimethylamino
Join with acid components via an ester linkage
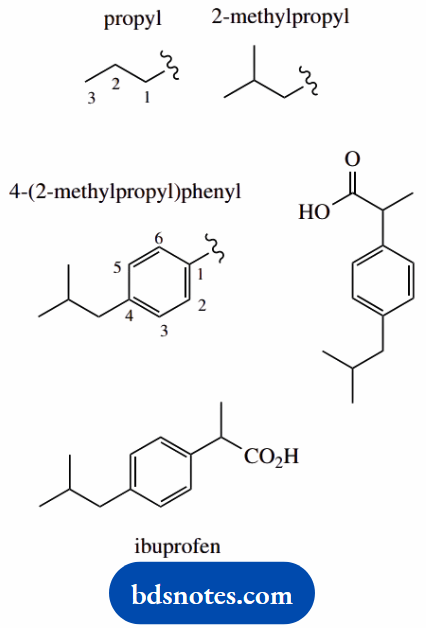
2-[4-(2-methylpropyl)phenyl]propanoic acid (ibuprofen)
Main chain propane (C3) number it 
A main functional group is an acid (-oic acid) this will be carbon-1 
Put in substituents consider brackets; we have square brackets with curved brackets inside initially ignore the contents of the curved brackets and their numbering (4), this reduces to 2-[phenyl] propanoic acid, which indicates phenyl at position 2 on propanoic acid
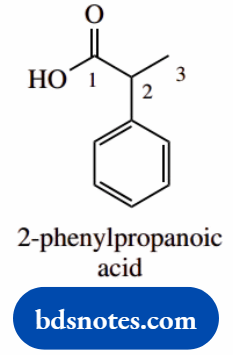
4-(2-methylpropyl)phenyl = 2-methylpropyl at position 4 of the phenyl;
2-methylpropyl = propyl with methyl at position 2 note the brackets separate different substituents and their individual numbering systems
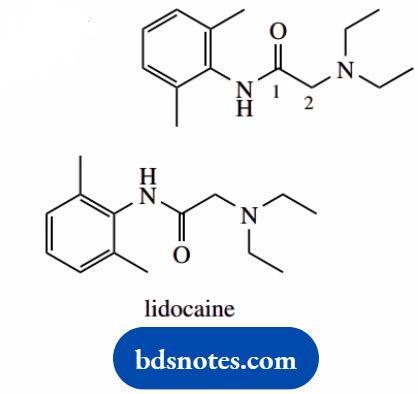
2-(diethylamino)-N-(2,6-dimethylphenyl)acetamide (lidocaine; lignocaine)
This is an amide; acetamide is the amide of acetic acid (C2)
number it; the carbonyl carbon is C-1 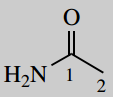
There are two main substituents, on C-2 and the nitrogen, with brackets to keep the appropriate groups together the substituent at C-2 is diethylamino.
An amino which is itself substituted with two ethyl groups the substituent on the nitrogen is 2,6-dimethylphenyl, a phenyl group substituted at positions 2 and 6 on the phenyl.
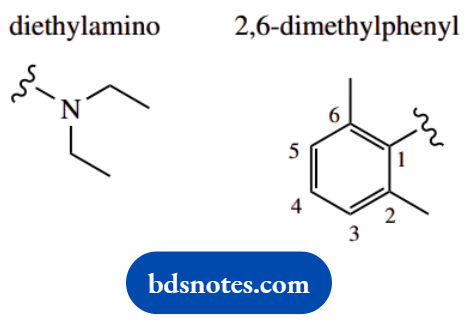
Put in substituents
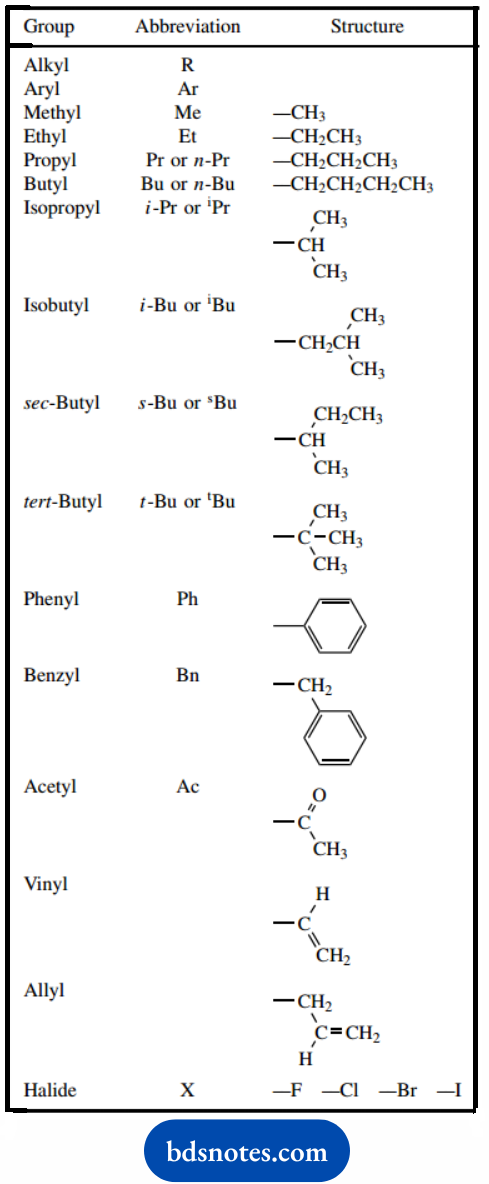
Common Groups And Abbreviations
In drawing structures, we are already using a sophisticated series of abbreviations for atoms and bonding.
Functional groups are also abbreviated further, in that –CO2H or –CHO convey considerably more information to us than the simple formula does.
Other common abbreviations are used to specify particular alkyl or aryl groups in compounds, to speed up our writing of chemistry.
It is highly likely that some of these are already familiar, such as Me for methyl, and Et for ethyl. Others are included.
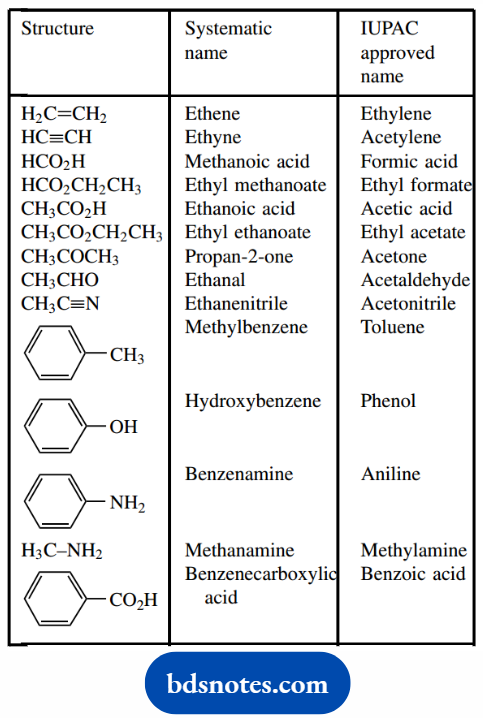
Common Non – Systematic Names
Systematic nomenclature was introduced at a relatively late stage in the history of chemistry, and thus common names had already been coined for a wide range of chemicals.
Because these names were in everyday usage, and familiar to most chemists, a number have been adopted by IUPAC as the approved name, even though they are not systematic.
These are thus names that chemists still use, that are used for labeling reagent bottles, and are those under which the chemical is purchased.
Some of these are given in Table 1.4, and it may come as a shock to realize that the systematic names school chemistry courses have provided will probably have to be ‘relearned’.
The use of the old terminology n- (normal) for unbranched hydrocarbon chains, with i- (iso), s- (secondary), and t- (tertiary) for branched chains is still quite common with small molecules and can be acceptable in IUPAC names.
Trivial Names For Complex Structures
Biochemical and natural product structures are usually quite complex, some exceedingly so, and fully systematic nomenclature becomes impracticable.
Names are thus typically based on so-called trivial nomenclature, in which the discoverer of the natural product exerts his or her right to name the compound.
The organism in which the compound has been found is frequently chosen to supply the root name, For Example. hyoscyamine from Hyoscyamus, atropine from Atropa, or penicillin from Penicillium.
Name suffixes might be -in to indicate a constituent of’, -side to show the compound is a sugar derivative, -genin for the aglycone released by hydrolysis of the sugar derivative -toxin for a poisonous constituent, or they may reflect chemical functionality, such as -one or -ol.
Traditionally, -one is always used for alkaloids (amines). Structurally related compounds are then named as derivatives of the original, using standard prefixes, such as hydroxy-, methoxy-, methyl-, dihydro-, homo-, etc. for added substituents, or deoxy-, dimethyl-, dimethoxy-, dehydro-, nor-, etc.
For removed substituents. Homo- is used to indicate one carbon more, whereas nor- means one carbon less. The position of this change is then indicated by the systematic numbering of the carbon chains or rings.
Some groups of compounds, such as steroids and prostaglandins, are named semi-systematically from an accepted root name for the complex hydrocarbon skeleton.
Drug names chosen by pharmaceutical manufacturers are quite random and have no particular relationship to the chemical structure.
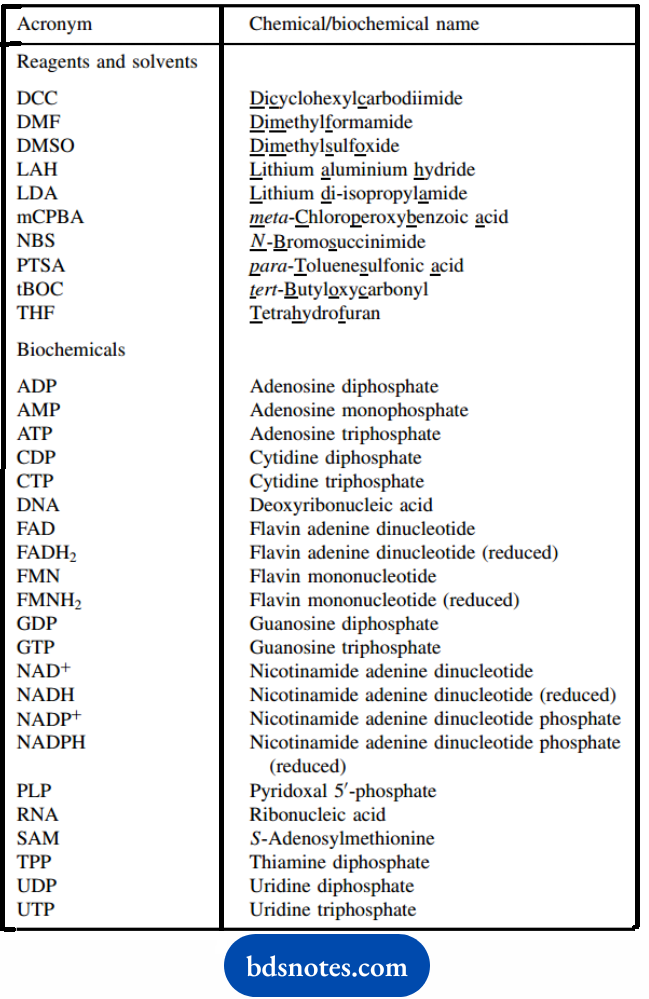
Acronyms
Some of the common reagent chemicals and solvents are usually referred to by acronyms, a sequence of letters derived from either the systematic name or a trivial name.
We shall encounter some of these in due course, and both names and acronyms will be introduced when we first meet them. For reference purposes, those we shall meet are also listed.
Far more examples occur with biochemicals. Those indicated cover many, but the list is not comprehensive.
Pronunciation
As you listen to chemists talking about chemicals, you will soon realize that there is no strict protocol for pronunciation.
Even simple words like ethyl produce a variety of sounds. Many chemists say ‘eethyle’.
But the Atlantic divide gives us ‘ethel’ with short ‘e’s, and continental European chemists often revert to the German pronunciation ‘etool’.
There is little to guide us in the words themselves, since methane is pronounced ‘meethayne’ whilst methanol tends to have short ‘e’, ‘a’, and ‘o’, except for occasional cases.
Mainly European, when it may get a long ‘o’. On the other hand, propanol always seems to have the first ‘o’ long, and the second one short.
Vinyl can be ‘vinil’ or ‘vynyl’ according to preference, and amino might be ‘ameeno’ or ‘amyno’. Need we go on? Your various teachers will probably pronounce some common words quite differently.
Try to use the most commonly accepted pronunciations, and don’t worry when a conversation with someone involves differences in pronunciation.
As long as there is mutual understanding, it’s not really important how we say it. By and large, chemists are a very tolerant group of people.
Leave a Reply