Gypsum Products
What are gypsum products?
Or
Write about manufacturing and theories of setting of gypsum products.
Answer:
Most commonly used materials that produce positive replication of oral structures are known as gypsum products. Gypsum products are derived from mineral gypsum which is chemically known as calcium sulfate dihydrate. When powdered gypsum get heated by calcinations to remove the part of its moisture content, the gypsum product a fie powder of calcium sulphate hemihydrate is formed. So dehydration of gypsum leads to the production of gypsum products which ranges from hydrous to anhydrous form of calcium sulfate.
Manufacturing of Gypsum Products:
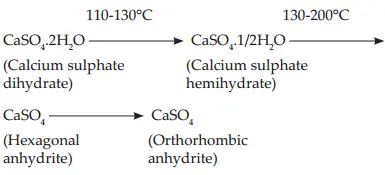
The process of heating gypsum for the manufacture of plaster is known as calcinations. Mined gypsum is ground and heated. When heated, gypsum (calcium sulphate dihydrate) looses part of its water of crystallization and changes to calcium sulphate hemihydrate. On further heating the remaining water of crystallization is lost. First, hexagonal anhydrite (soluble anhydrite) is formed. Later, orthorhombic anhydrite (insoluble anhydrite) is formed.
Manufacture of Dental Plaster:
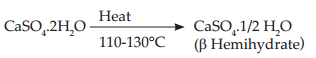
Gypsum is ground and heated in an open letter or kiln at a temperature of 110 to 130° C. The process is called dry calcination. β Type of crystals are formed. Microscopically, firous aggregate of fie crystals with capillary pores. They are then ground to breakup the needle-like crystals. This improves packing.
Manufacture of Dental Stone:
Gypsum is calcined under steam pressure in an autoclave at 120 to 130°C at 17 lbs/sq inch for 5 to 7 hours. Thus, the product obtained is much stronger and harder than b hemihydrate.
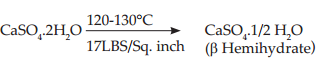
Manufacture of High Strength (α modified) Stone:
The gypsum is calcined by boiling it in 30% calcium chloride solution. The chlorides are then washed away or autoclaved in presence of sodium succinate 0.5%. These particles are the densest of all three types. After controlled grinding these powders have an even higher apparent density and yield a stronger set. Microscopically it is cuboidal in shape.
Theories of Setting:
Three theories have been put forward, i.e.
- Colloidal theory
- Hydration theory
- Dissolution—precipitation theory.
- Colloidal Theory: This theory was given by Mahaelis in 1893. The theory proposes that when mixed with water, plaster enters into a colloidal state through a sol-gel mechanism. In the sol state, hemihydrate combines with water to form dihydrate. As the water is consumed the mass turns to a ‘solid gel’.
- Hydration Theory: The hydration theory suggests that rehydrated plaster particles join together through hydrogen bonding to the sulfate groups to form the set solid mass.
- Dissolution-Precipitation Theory (Crystalline Theory):
This theory is widely accepted.
- According to the theory, the plasterdissolves and reacts to form gypsum crystals which interlock to form the set solid.
- The settling reaction is explained on the basis of difference in solubility of hemihydrates and dihydrate.
- Hemihydrate is four times more soluble than dihydrate. When hemihydrate is mixed in water it forms a flid workable suspension.
- Hemihydrate dissolves until it forms a saturated solution. Some dihydrate is formed due to the reaction.
- The solubility of dihydrate is much less than hemihydrate, the saturated hemihydrate is supersaturated with respect to the dihydrate.
- All supersaturated solutions are unstable. So the dihydrate crystals precipitate out.
- As the dihydrate precipitates out, the solution is no longer saturated with hemihydrate and so it continues to dissolve.
- The process continues until further dihydrate precipitates out of the solution.
- Initially, there is little reaction and thus little or no rise in temperature. This time is referred to as the induction period.
- As the reaction proceeds gypsum is formed in the form of needle-like clusters called spherulites.
- Continued growth and intermeshing of crystals of gypsum leads to thickening and hardening of the mass into a strong solid structure.
Leave a Reply