Free Radicals And Cell Injury
Question 1. Define Necrosis. Name Different Types Of Necrosis. What Are Nuclear Changes In Necrosis?
Answer:
Necrosis is defined as focal death along with the degradation of tissue by hydrolytic enzymes liberated by cells.
Types Of Necrosis
There are five types of necrosis:
- Coagulative necrosis
- Liquefaction necrosis
- Caseous necrosis
- Fat necrosis
- Fibrinoid necrosis.
Free radicals in cell injury
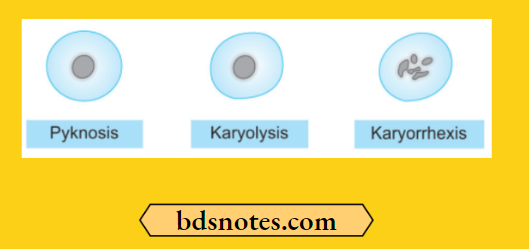
Necrosis Nuclear Changes
The nuclear changes are:
- Pyknosis: Condensation of nuclear chromatin.
- Karyolysis: This may either undergo dissolution.
- Karyorrhexis: Fragmentation into many granular clumps.
Question 2: Define Cell Injury. Write In Brief About Its Pathogenesis.
Answer:
If the limits of adaptive response to a stimulus are exceeded, or in certain instances when adaptation is not possible, a sequence of events follows, loosely termed cell injury.
Cell injury is reversible up to a certain point, but if the stimulus persists or is severe enough from the beginning, the cell reaches the “point of no return” and suffers irreversible cell injury and cell death.
Mechanism of cell injury by free radicals
1. Pathogenesis Of Cell Injury
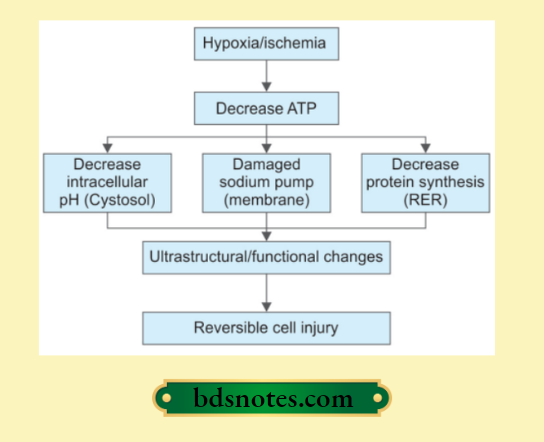
Pathogenesis Of Ischemic And Hypoxic Injury
- Reversible cell injury.
- Irreversible cell injury.
Reversible cell injury
Irreversible Cell Injury
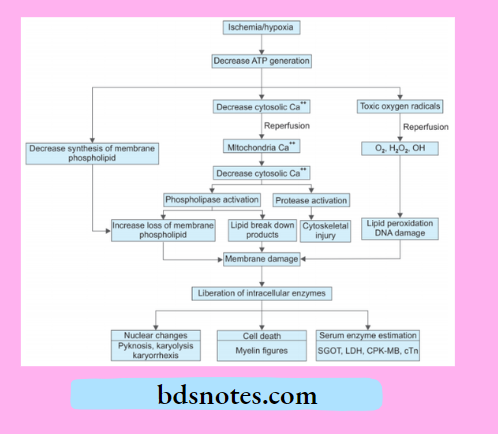
2. Free Radical Mediated Cell Injury
Following is the pathogenesis of free radical-mediated cell injury
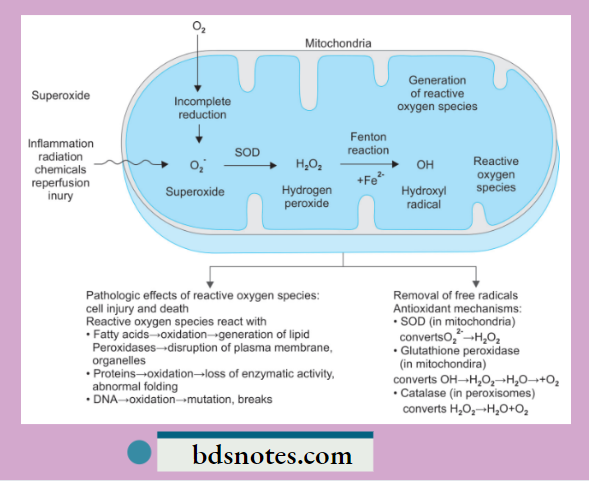
- Generation of oxygen free radicals begins within the mitochondrial inner membrane when cytochrome oxidase catalyzes the four-electron reduction of oxygen (O2) to water (H2O).
Intermediate between the reaction of O2 to H2O, three partially reduced species of oxygen are generated depending upon the number of electrons transferred.
These are:- Superoxide oxygen (O2–): One electron
- Hydrogen peroxide (H2O2): Two electrons
- Hydroxyl radical (OH–): Three electrons.
- A few other oxygen radicals that may be generated in reactions other than those during O2 to H2O are hypochlorous acid (HOCl), peroxynitrite ion (ONOO), nitric oxide (NO) generated by various body cells (endothelial cells, neurons, macrophages, etc.), and release of superoxide free radical in Fenton reaction.
Oxidative stress and cell injury
Free Radical Generation
- The three partially reduced intermediate species between O2 to H2O are derived from enzymatic and non-enzymatic reactions as follows:
- Superoxide (O2–): Superoxide ion O2– may be generated by direct auto-oxidation of O2 during mitochondrial electron transport reaction. Alternatively, O2– is produced enzymatically by xanthine oxidase and cytochrome P450 in the mitochondria or cytosol. O2– so formed is catabolized to produce H2O2 by superoxide dismutase (SOD).
- Hydrogen peroxide (H2O2): H2O2 is reduced to water enzymatically by catalase (in the peroxisomes) and glutathione peroxidase GSH (both in the cytosol and mitochondria).
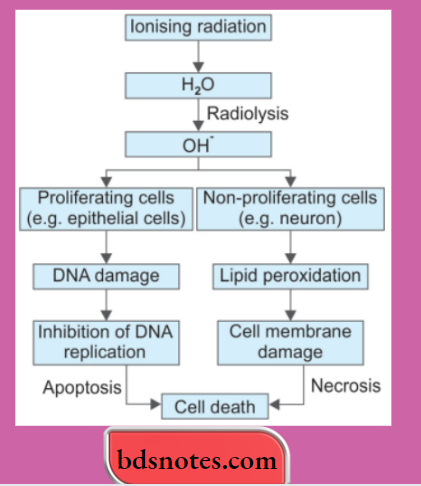
- Hydroxyl radical (OH–): OH– radical is formed in two ways in biological processes:s by radiolysis of water and by reaction of H2O2 with ferrous (Fe++) ions; the latter process is termed the Fenton reaction.
Free Radical Reactions
The hydroxyl radical is the most reactive species. Reactive oxygen species (ROS) produce membrane damage by the following mechanisms:
- ROS reacts with fatty acids and leads to oxidation, which leads to the generation of lipid peroxidases. This leads to disruption of the plasma membrane and organelles.
- ROS reacts with proteins, leading to the oxidation of proteins, and there is a loss of enzymatic activity and abnormal folding of proteins.
- ROS reacts with DNA and leads to the oxidation of DNA, and there is mutation and breakage in single strands of mitochondrial and nuclear DNA.
Cell injury pathology short note
3. Cell Injury By Chemicals
Chemicals induce cell injury by one of the following two mechanisms:
- Direct cytotoxic effects: Some chemicals combine with components of the cell and produce direct cytotoxicity without requiring metabolic activation. The cytotoxic damage is usually greatest to cells that are involved in the metabolism of such chemicals. For Example. in mercuric chloride poisoning, the greatest damage occurs to cells of the alimentary tract and kidney, cyanide kills the cell by poisoning mitochondrial cytochrome oxidase, thus blocking oxidative phosphorylation.
- Conversion to reactive, toxic metabolites: This mechanism involves metabolic activation to yield the ultimate toxin that interacts with the target cells. The target cells in this group of chemicals may not be the samcellsll that metabolized the toxin. An example of cell injury by the conversion of reactive metabolites is toxic liver necrosis caused by carbon tetrachloride.
4. Cell Injury by Physical Agents/Ionizing Radiation
Leave a Reply