Drugs Acting On Autonomic Nervous System Introduction
Adrenergic drugs mimic or interfere with the functioning of the sympathetic nervous system by affecting the release or action of norepinephrine and epinephrine.
- These hormones, which are also known as noradrenaline and adrenaline, are secreted by the adrenal gland, hence their association with the term adrenergic.
- The primary actions of norepinephrine and epinephrine are to mediate the “fight-or-flight response.” Thus, they constrict blood vessels (vasoconstriction), which increases blood pressure, and accelerates the rate and force of contractions of the heart.
- Adrenergic drugs that produce or inhibit these effects are known as sympathomimetic agents and sympatholytic agents, respectively.
Therapeutically, these drugs are used to combat life-threatening disorders, which include acute attacks of bronchial asthma, shock, cardiac arrest, and allergic reactions. In addition, these drugs are used as nasal decongestants and appetite suppressants.
Adrenergic Neurotransmitters
The catecholamines are found in the body as norepinephrine, epinephrine, and dopamine are formed by hydroxylation and decarboxylation of the amino acids phenylalanine and tyrosine.
- All three of these catecholamines act as neurotransmitters in the central nervous system (CNS). Norepinephrine also functions as a neurotransmitter in the sympathetic nervous system.
- Although there are dopamine receptors outside the CNS, the role of dopamine as a hormone or neurotransmitter peripherally is not fully described.
Epinephrine is the circulating hormone secreted by the adrenal medulla and influences processes throughout the body.
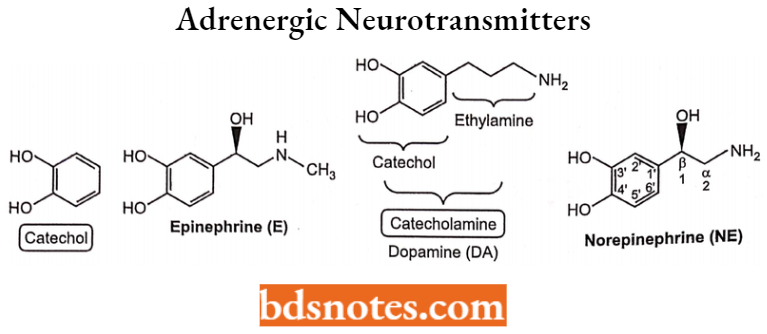
The catecholamines are 3, 4-dihydroxyphenolic amines. The benzene ring structure numbering is counter-clockwise with carbon 1 being bonded to the aliphatic side chain.
- The catechol group consists of two adjacent hydroxyl groups: one at position 4, or para concerning position 1, and the second at position 3, or ortho concerning the hydroxyl at position 4.
- The aliphatic side-chain carbon atoms are labeled (3 and a. As illustrated, the substitutions on these carbons define each of the three most prominent catecholamines.
Biosynthesis And Catabolism Of Catecholamines
Biosynthesis of Catecholamines
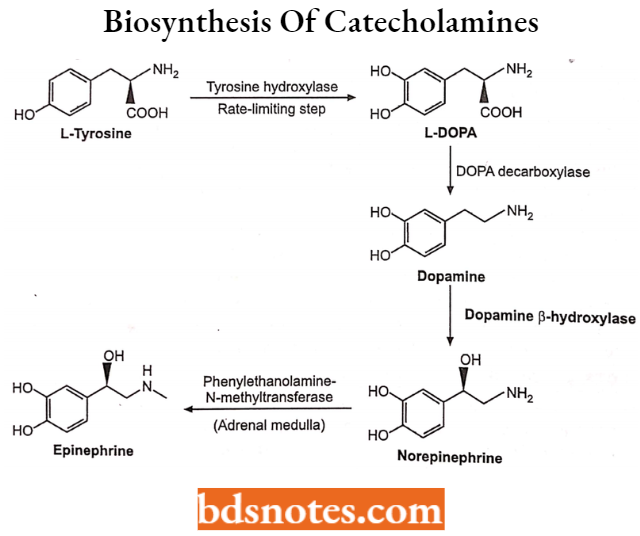
Biosynthesis begins with tyrosine, which consists of a benzene ring hydroxylated in the 4 (para) position to the two-carbon side chain at the 1 position. The β-carbon, closest to the ring, is saturated with hydrogen and is single-bonded to the β-carbon.
- The a-carbon is bonded to the amino and carboxylic acid groups that define the amino acids. The rate-limiting enzyme, tyrosine hydroxylase, 3-hydroxylates (ortho concerning the 4-position hydroxyl) tyrosine to dihydroxyphenylalanine (DOPA).
- The a-carbon is decarboxylated by aromatic L-amino acid decarboxylase to form the first catecholamine, dopamine (L-dihydroxy phenylethylamine).
- Hydroxylation of the β-carbon of dopamine (by dopamine β-hydroxylase) results in the formation of norepinephrine. Dopamine β-hydroxylase requires vitamin C (ascorbic acid) as a cofactor.
Norepinephrine is converted into epinephrine by methylation of the amino group on the a-carbon by phenylethanolamine-N-methyltransferase (PNMT). Tyrosine hydroxylase activity is the rate-limiting step in catecholamine synthesis.
- The noradrenaline formed in the adrenergic nerve endings remains stored in vesicles as its adenosine triphosphate complex. The adrenal medulla also synthesizes and stores noradrenaline and adrenaline.
- The neurotransmitters are released by increasing the permeability of the nerve terminal membrane to Ca++. The inflow of Ca++ triggers the fusion of the vesicle with the cell membrane, resulting in exocytosis.
Catecholamine Catabolism
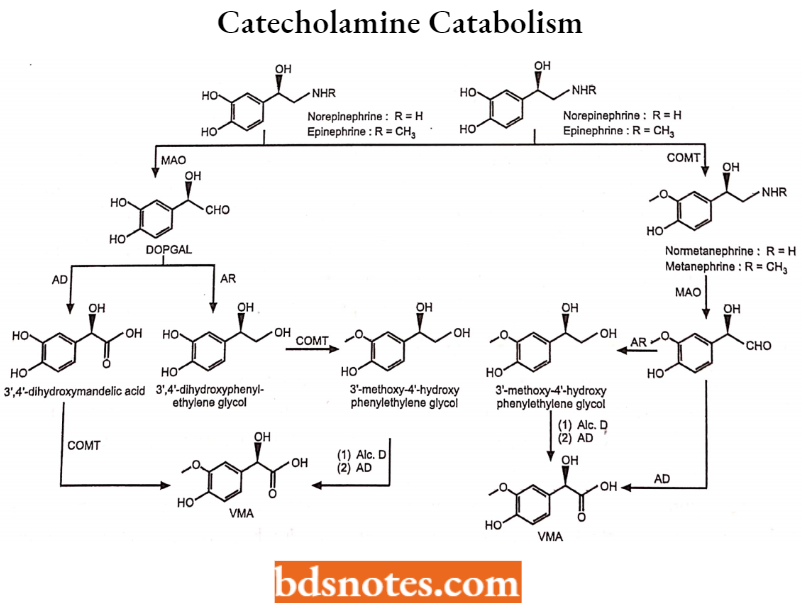
There are two primary pathways for the degradation of catecholamines; one is near the site where catecholamines are synthesized and stored (chromaffin cells and sympathetic neurons), and the second deactivates primarily circulating catecholamines.
- Monoamine oxidase (MAO) catalyzes the first and catechol-O-methyltransferase (COMT). The second. MAO, a mitochondrial enzyme, cleaves off the terminal aliphatic amine and oxidizes the a-carbon to carboxylic acid.
- The product, 3,4-dihydroxymandelic acid, is the same for epinephrine and norepinephrine because the N-methyl group that distinguishes the two is removed.
COMT using the methyl-group donor S-adenosylmethionine methylates the 3-hydroxy group, producing metanephrine and normetanephrine, respectively, from epinephrine and norepinephrine.
- O-Methylation of 3,4-dihydroxymandelic acid by COMT or oxidative deamination of the metanephrines by MAO produces vanillylmandelic acid (VMA).
- Most of the substrates and products of these reactions may be conjugated to sulfate (primarily) or glucuronide, which reduces further metabolism and enhances excretion.
Adrenergic Receptors And Their Distribution
The two major categories of adrenergic receptors are designated alpha and beta which mediate the vasoconstrictor actions of catecholamines. Alpha receptors have been further subdivided into:
- Receptor α1 involves a Gq protein that elevates Ca++, resulting in smooth muscle contraction. Receptor α2 involves a Gi protein that decreases cAMP and C++ influx which inhibits neurotransmission.
- Receptors β1, β2, and β3 involve a Gs protein and cAMP elevation that increases Ca++ for smooth muscle contraction (β2 can be linked to a Gi protein that decreases cAMP resulting in smooth muscle relaxation).
β1 adrenoceptors occur in the heart and are involved in heart stimulation processes and its discharge. β2 adrenoceptors appear in skeletal muscles and they stimulate vasodilatations, Thus the effect of NE can be either excitatory or inhibitory, based on the receptor distribution within visceral organs.
Sympathomimetic Agents (SM)
Sympathomimetic drugs or adrenergic drugs are stimulant compounds that mimic the effects of endogenous catecholamines (adrenergic neurotransmitters) agonists of the sympathetic nervous system (i.e., epinephrine [adrenaline], norepinephrine [noradrenaline], and dopamine).
- These agents include drugs that act on adrenergic receptors (adrenoceptors) directly or indirectly, such as by blocking the breakdown of neuronal uptake of catecholamines.
- Because sympathomimetic drugs raise blood pressure and increase heart rate, they are useful in treating systemic trauma, including bronchial asthma, shock, and cardiac arrest.
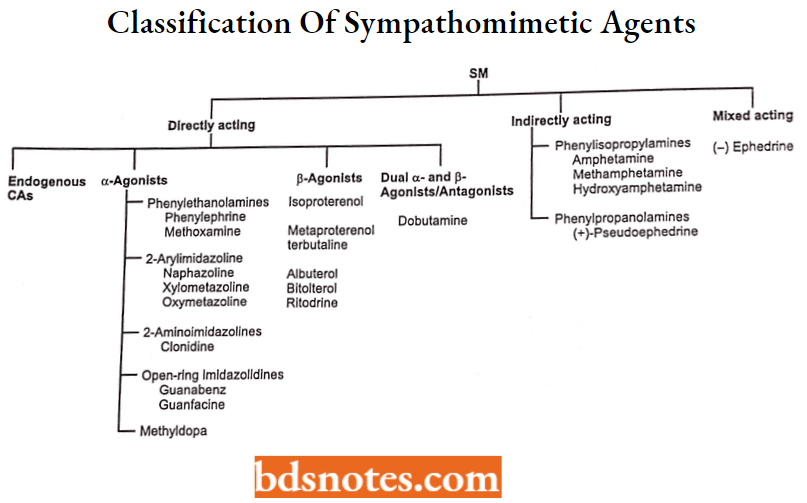
Classification Of Sympathomimetic Agents
Sympathomimetic agents are classified as follows:
Direct acting: Nor-epinephrine, Epinephrine, Phenylepinephrine, Dopamine, Methyldopa, Clonidine, Dobutamine, Isoproterenol, Terbutaline, Salbutamol, Bitolterol, Naphazoline, Oxymetazoline and Xylometazoline.
Indirect acting: Hydroxyamphetamine, Pseudoephedrine, Propylhexedrine. Mixed acting: Ephedrine, Metaraminol.
Sar Of Sympathomimetic Agents
The parent structure of many adrenergic drugs is (3-phenylethylamine.
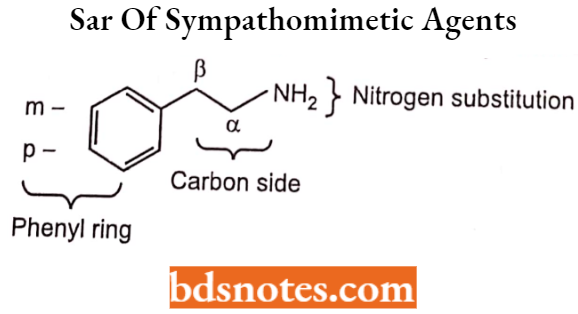
The modifications of p-phenylethylamine influence not only the mechanism of action the receptor selectivity, but also their absorption, oral activity, metabolism, and thus the duration of action (DOA).
- Naturally occurring catecholamine is active at both α and β-preceptor but has poor oral activity as it is rapidly metabolized by COMT. Thus the change in position of the hydroxyl group from meta and para podition to meta and other positions gives a drug with good metabolic activity.
- For the direct-acting sympathomimetic amines, maximal activity is seen in β-phenylethylamine derivatives containing:
- Catechol moiety and
- β-OH group on the ethylamine portion of the molecule.
- The greatest adrenergic activity occurs when two carbon atoms separate the aromatic ring from the amino group.
- For catecholamine, the more potent enantiomer has the (1R) configuration. This enantiomer is typically several 100 fold more potent than the enantiomer with the (1S) configuration.
- Primary and secondary amines have good adrenergic activity, whereas tertiary amines and quaternary ammonium salts do not.
- The nature of the amino substituent also affects the receptor selectivity of the compound. As the size of the nitrogen substituent increases, a-receptor agonist activity generally decreases and p-receptor agonist activity increases.
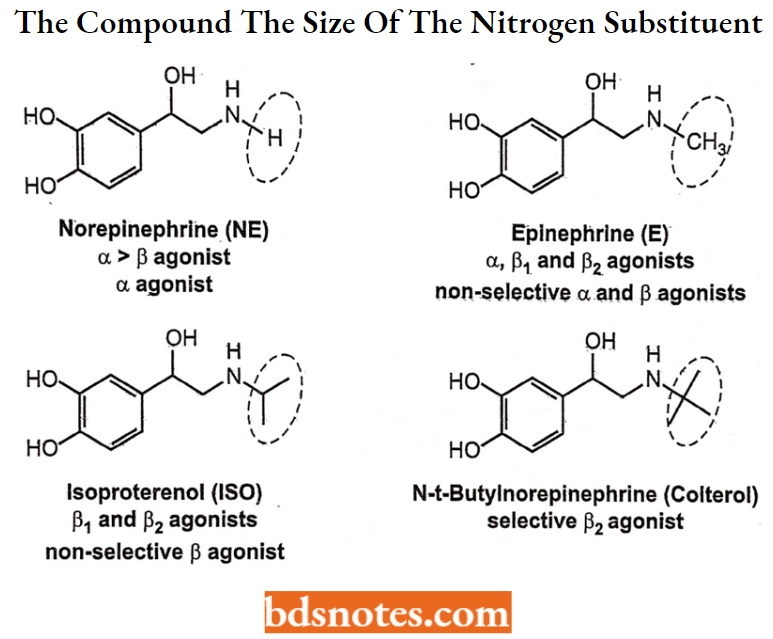
Thus, NE has more α-activity than β-activity and E is a potent agonist at α-, β1-, and β2– receptors. N-tert-butyl group enhances β2-selectivity.
- Substitution of the a-carbon by a small alkyl group (For Example., CH3– or C2H5-) slows metabolism by MAO. This is very important for non-catechol compounds where the addition of a small alkyl group increases.
- The resistance to metabolism and lipophilicity, so such compounds often exhibit enhanced oral effectiveness and greater CNS activity than other compounds that do not contain an oc-alkyl group.
- OH, substitution on the β-carbon greatly enhances agonist activity at both α- and β-receptors and largely decreases CNS activity because it lowers lipid solubility.
- Maximal α and β-activity depend on the presence of meta and para OH groups. Compounds without one or both phenolic OH substituents are not metabolized by COMT, and they are orally active and have a longer duration of action.
For example, phenylephrine (removal of OH group, which lacks β action but has less 1 – agonist property).
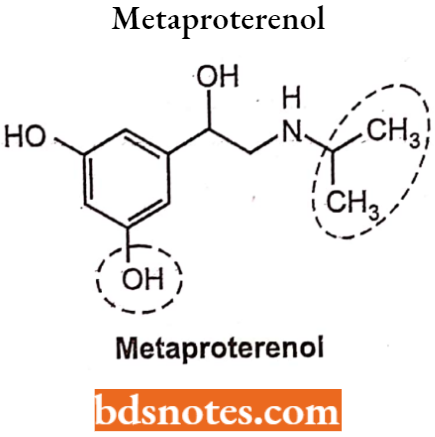
Although the catechol moiety is important, maximal agonist activity at adrenoceptors can be replaced with other substituted phenyl moieties to provide selective adrenergic agonists.
Replacement of the catechol moiety of isoproterenol with the resorcinol structure gives a selective β2-agonist which shows a longer duration of action because they are resistant towards COMT.
Example: Metaproterenol
Catecholamine without OH group on phenyl group (catechol moiety) loss of direct sympathomimetic activity becomes indirectly sympathomimetic and not metabolized by COMT and they are orally active and have a longer duration of action.
Example: Amphetamine
A second chemical class of α-antagonists is imidazolines. These imidazolines are non-selective or can be selective for either α1 – or α2 -receptors.
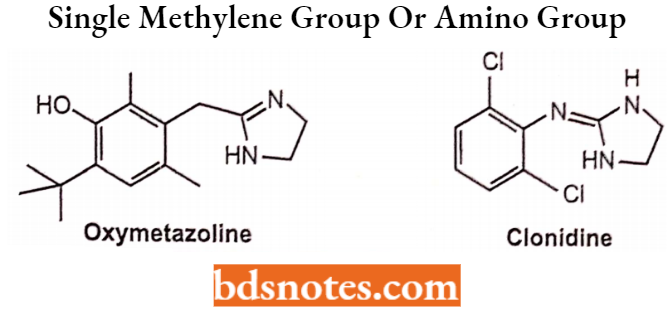
Structurally, most imidazolines have their heterocyclic imidazoline nucleus linked to a substituted aromatic moiety via some type of bridging unit. The optimum bridging unit is usually a single methylene group or amino group.
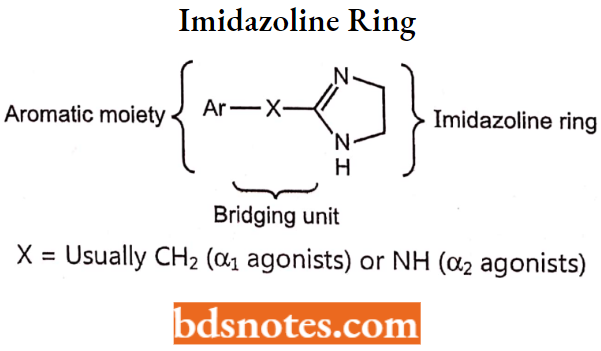
The optimum bridging unit is usually a single methylene group or amino group.
Direct Acting Sympathomimetic Agents
Direct-acting drugs produce effects directly by interacting with adrenergic receptors and produce sympathomimetic effects.
Structurally direct-acting adrenergic agonists contain catechol moiety (i.e. OH group at both meto and paro position) along with OH group at {3-position and lacks β-position substitution in the side chain. These compounds are easily metabolized by both MAO and COMT.
- Direct-acting sympathomimetics are classified based on their selectivity for adrenoceptors, of which there are several types, including α1, α2, β1, and β2 substances that activate β2 – adrenoceptors (known as β2 – adrenoceptors agonists) are some of the most widely used direct-acting sympathomimetics.
- Especially in the treatment of asthma because of their ability to relax smooth muscle tissue in the airways of the lungs. However, none of the available drugs are completely selective for the β2-adrenoceptor.
- They tend to produce unwanted effects on the heart, such as increased heart rate and disturbances of cardiac rhythm, through their action on cardiac β1-adrenoceptors. To reduce these side effects.
- The β2 agonists are usually given by inhalation, which increases pulmonary exposure to the drug while lowering systemic exposure and therefore activation of cardiac receptors.
Examples of β2 agonists include terbutaline, albuterol, and metaproterenol. Some direct-acting agents are nonselective; for example, isoproterenol produces effects at all β receptors, and the (+) and (-) isoforms of dobutamine produce varying effects at α and β receptors.
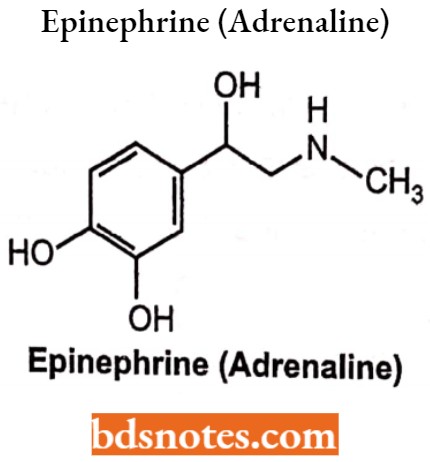
Epinephrine (Adrenaline)
Epinephrine is a naturally occurring catecholamine, a direct-acting sympathomimetic agent exerting its effect on alpha and beta-adrenoreceptors. It is a powerful cardiac stimulant.
- α stimulation causes peripheral, renal, splanchnic, and pulmonary vasoconstriction.
- β1 stimulation causes an increase in heart rate, contractility, and excitability.
- β2 stimulation causes an increase in bronchodilation and vasodilation in skeletal muscles.
- It is administered in cardiac arrest to cause peripheral vasoconstriction via its alpha-adrenergic action (increases available cardiac output to the myocardium and brain).
- It may facilitate defibrillation by improving myocardial blood flow during CPR.
Epinephrine is the chemical, (R)l-(3,4-hydroxyphenyl)-2-methylamino ethanol. (R)-epinephrine is 12 times more potent than (S)-form.
Major effects are increased systolic blood pressure, reduced diastolic pressure, tachycardia, hyperglycemia, and hypokalaemia. It has vasopressor properties, an antihistaminic action, and is a bronchodilator
Adrenaline is rapidly distributed to the heart, spleen, several glandular tissues, and adrenergic nerves. It crosses the placenta and is excreted in breast milk.
- It is approximately 50% bound to plasma proteins. The onset of action is rapid and after intravenous infusion, the half-life is approximately 5-10 minutes. Adrenaline is rapidly metabolized in the liver and tissues.
- Up to 90% of the 4 doses are excreted as metabolites in the urine due to rapid oxidation and conjugation by both COMT and MAO.
Uses:
- It is used in sympathomimetics, bronchiolitis, and antiasthmatic. It prevents internal bleeding during surgery. Used to treat heart blocks and open-angle glaucoma.
- Also used in emergencies to treat serious allergic reactions to insect stings/bites, foods, drugs, or other substances. It improves breathing, stimulates the heart, raises dropping blood pressure, and reduces swelling of the face, lips, and throat.
Adverse Effects:
- Tachycardia, anxiety, restlessness, tremors, weakness, dizziness, headache, nausea, vomiting, flushing and redness of face and skin, hypertension, and increased after load.
- Exacerbation of myocardial ischemia, and renal vasoconstriction may reduce renal blood flow and glomerular filtration rate, hypokalaemia, and hyperglycemia.
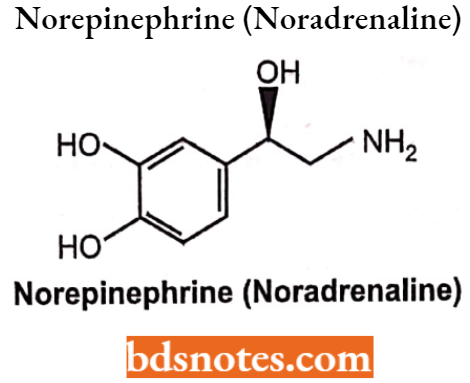
Nor-epinephrine (Nor-adrenaline)
Nor-epinephrine is a natural catecholamine and a phenethylamine derivative. It differs from adrenaline only by lacking the methyl substitution on the aminoethanol and, as for adrenaline.
- Chemically it is 1- β (3,4-dihydroxyphenyl]-α-aminoethanol. The L-isomer is a pharmacologically active and potent α1 receptor agonist but has relatively less β2-receptor activity.
- It functions as a peripheral vasoconstrictor (alpha-adrenergic action) and inotropic stimulator of the heart and dilator of coronary arteries (beta-adrenergic action).
- Along with epinephrine, norepinephrine also underlies the fight-or-flight response, directly increasing heart rate, triggering the release of glucose from energy stores, and increasing blood flow to skeletal muscle.
- Norepinephrine is synthesized from dopamine by dopamine β-hydroxylase. It is released from the adrenal medulla into the blood as a hormone and is also a neurotransmitter in the central nervous system and sympathetic nervous system where it is released from noradrenergic neurons.
- The actions of norepinephrine are carried out via the binding to adrenergic receptors. Noradrenaline is predominantly metabolized by catechol-o-methyltransferase (COMT) and monoamine oxidase (MAO).
Noradrenaline has a rapid onset of action (1-2 minutes), and a fast elimination when the infusion is ceased due to the short half-life of 1-2 minutes.
Mechanism of Action:
Norepinephrine is synthesized from tyrosine as a precursor, and packed into synaptic vesicles. It performs its action by being released into the synaptic cleft.
Where it acts on adrenergic receptors, followed by the signal termination, either by degradation of norepinephrine or by uptake by surrounding cells.
Biosynthesis:
Norepinephrine is synthesized by a series of enzymatic steps in the adrenal medulla and postganglionic neurons of the sympathetic nervous system from the amino acid tyrosine.
- The first reaction is the hydroxylation into dihydroxyphenylalanine (L-DOPA) (DOPA = 3,4-DiHydroxy-L-Phenylalanine), catalyzed by tyrosine hydroxylase. This is the rate-limiting step.
- This is followed by decarboxylation into the neurotransmitter dopamine, catalyzed by pyridoxal phosphate and DOPA decarboxylase.
- Last is the final β-oxidation into norepinephrine by dopamine beta-hydroxylase, requiring ascorbate as a cofactor (electron donor).
Uses:
Norepinephrine is similar to adrenaline used for blood pressure control in certain acute hypotensive states (For Example., pheochromocytomectomy, sympathectomy, poliomyelitis, spinal anesthesia, myocardial infarction, septicemia, blood transfusion, and drug reactions).
Adverse Effects:
Ischaemic injury due to potent vasoconstrictor action and tissue hypoxia, bradycardia, anxiety, transient headache, and respiratory difficulty.
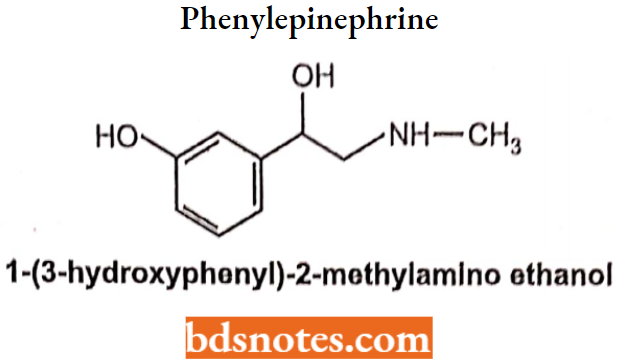
Phenylepinephrine
Phenylephrine hydrochloride is a synthetic sympathomimetic agent. Chemically, phenylephrine hydrochloride is (-)-m-Hydroxy-α-[(methylamino)methyl]benzyl alcohol.
It differs from adrenaline only by the absence of a hydroxyl group on the para position on the aromatic ring.
- It is a potent vasoconstriction and is active when given orally because it is resistant to COMT and active orally.
- It is a selective direct-acting receptor agonist.
- Its duration of action is about twice that of epinephrine.
- Due to a lack of catechol moiety, it is not metabolized by COMT, but metabolized by MAO.
- Relatively non-toxic and produce little CNS stimulation.
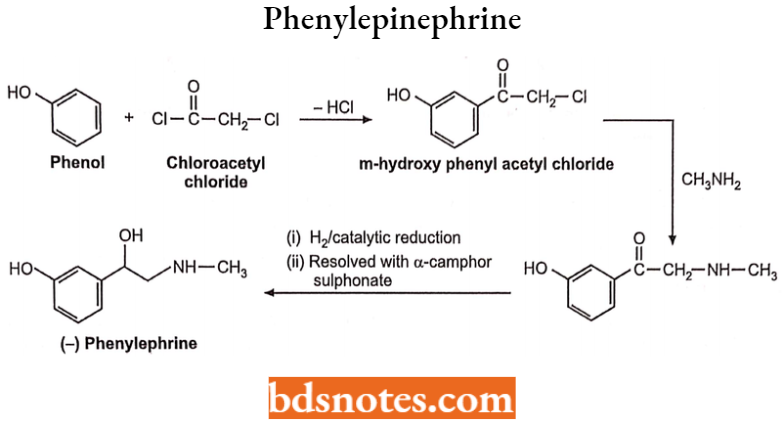
Synthesis: Synthesised by treating phenol with chloroacetyl chloride under acidic conditions. Later treated with methyl amine and then reduced and resolved with sulphonate derivative to yield phenylepinephrine.
Uses: It reduces congestion and swelling by constricting the blood vessels of the membrane. It dilates the pupil and is used to treat open-angle glaucoma.
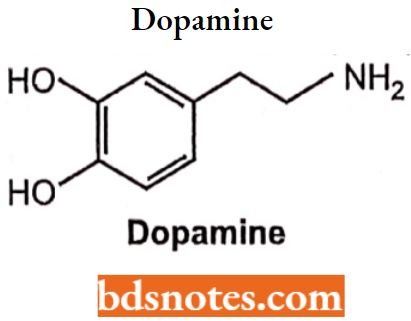
Dopamine
Chemically it is 2-(3,4-dihydroxy phenyl)-l-amino ethane. It is ineffective orally as it acts as a substrate for both MAO and COMT. It acts on the β2-adrenergic receptor causes positive ionotropic effects and also causes the release of nor-epinephrine.
Uses: It increases blood flow to the kidneys. It exerts the CVS effect by interacting with Di-dopaminergic receptors, especially in the mesenteric, renal, and coronary beds. Also in heart attack, trauma, surgery, and other serious medical conditions.
Methyldopa
Chemically it is (2S)-2-amino-3-(3,4-dihydroxyphenyl)-2-methylpropanoic acid. It is used as a prodrug, it is metabolized by enzymes and gives active metabolite a-methyl norepinephrine which is α2 agonist acting in the CNS thereby decreasing the sympathetic outflow leads to lower blood pressure.
It is a drug of choice for treating hypertension during pregnancy.
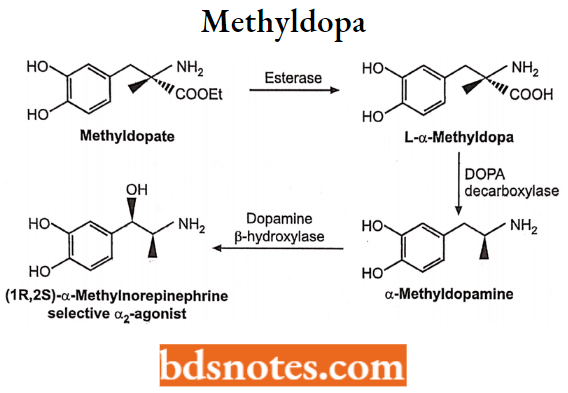
Uses: This medication is used alone or with other medications to treat high blood pressure (hypertension). Lowering high blood pressure helps prevent strokes, heart attacks, and kidney problems. Methyldopa works by relaxing blood vessels so blood can flow more easily.
Clonidine
Clonidine is an example of 2-aminoimidazoline derivatives. Chemically, it is N-(2,6-dichlorophenyl)-4,5-dihydro-lH-imidazol-2-amine that possesses a selective α2adrenergic receptor.
- The inductive and resonance effect of the dichloro phenyl ring decreases the pKa of clonidine. Clonidine enters the CNS and stimulates the α2 receptor located in the brain.
- Thus decreases the peripheral vascular resistance (PVR) and decreases the blood pressure and heart rate.
- It inhibits both dopaminergic and adrenergic neurotransmitters by inhibiting DOPA decarboxylase which converts L-Dopa into dopamine.
- Clonidine should normally be used in those patients in whom treatment with a diuretic or beta-blocker was found ineffective.
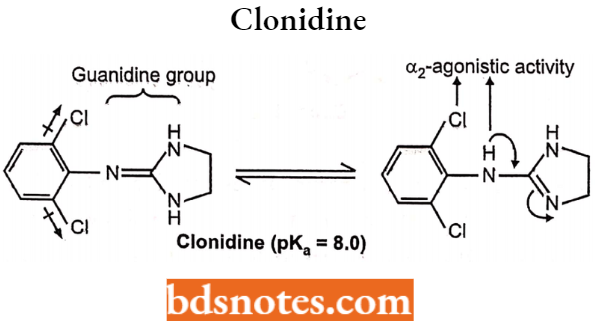
Mechanism Of Action
Clonidine hydrochloride is an adrenergic agonist which also has some adrenergic antagonist effects.
- The antihypertensive effect of clonidine hydrochloride is thought to be due to central α2 adrenergic stimulation, which results in a decreased sympathetic outflow to the heart.
- Kidneys, and peripheral vasculature and thus decreased peripheral vascular resistance, decreased systolic and diastolic blood pressure, and decreased heart rate.
Uses: Treatment of hypertension, nasal decongestant, open eye glaucoma. It is a potent sedative-hypnotic drug and can prevent post-operative shivering.
Dobutamine
Dobutamine is a synthetic catecholamine and acts as dual α and β agonists or antagonists that stimulate beta receptors of the heart to produce mild chronotropic, hypertensive, arrhythmogenic, and vasodilative effects. Chemically it is an analog of dopamine in which, l-methyl-3-(4-hydroxyphenyl)propyl substituent has been replaced on the amino group.
It possesses an asymmetric carbon atom and exhibits a pair of enantiomers.
- The (+) enantiomer possesses a potent full agonist activity at both β1 and β2-receptors but the (+) isomer is a potent α1 antagonist.
- The (-) enantiomer possesses a potent agonist activity at α1-receptors and is 10 times less potent at β1 and β2-receptors.
- It can be metabolized by COMT and conjugation but not by MAO.
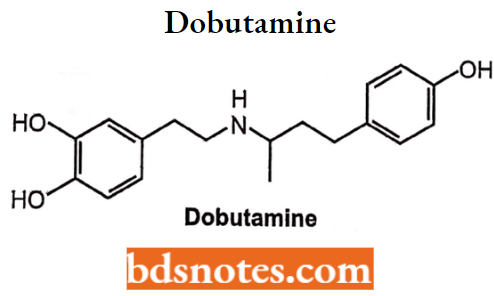
It stimulates the beta receptors in the heart and produces mild chronotropic, hypertensive, arrhythmogenic, and vasodilative effects.
- It stimulates the beta -1 stimulation resulting in an increase in heart rate (mild), myocardial contractility, and excitability.
- Beta-2 stimulation is minimal and may have some peripheral vasodilatation and bronchodilation. The onset of action is within 1-2 minutes, but with an infusion, it may take up to 10 minutes to obtain the peak effect.
Uses: It is used in the ICU for the treatment of congestive heart failure, cardiogenic shock, and pulmonary edema and to increase cardiac output. It is less arrhythmogenic than adrenaline.
Isoproterenol (Isoprenaline)
- Isoproterenol hydrochloride is 3,4-Dihydroxy-α-[(isopropylamine)methyl] benzyl alcohol hydrochloride, a synthetic sympathomimetic amine that is structurally related to epinephrine.
- But acts almost exclusively on beta receptors. Isoproterenol hydrochloride is a racemic compound. It is a potent non-selective and synthetic catecholamine with a β-adrenergic receptor agonist and with very low affinity for alpha-adrenergic receptors.
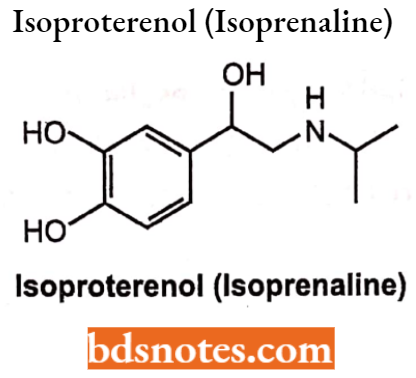
It acts on both β1 and β2 receptors and does not act on a-receptors. It increases the cardiac output by stimulating the β-receptor and bronchodilation by stimulating the β2-receptor located in the respiratory tract.
It is available as an inhalator, injection, and sublingual tablet. It is not taken orally due to less or no oxidation deamination by MAO. It undergoes metabolism by COMT. It is synthesized by the following method.
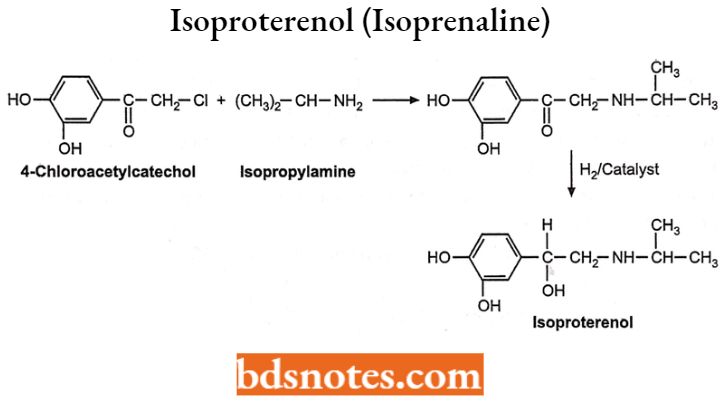
Uses: It is used in the treatment of bronchial asthma, as an antiarrhythmic agent, CNS stimulant, and peripheral vasodilator. Also used in the treatment of shock to increase the heart rate.
Adverse Effects:
CNS: Nervousness, headache, dizziness, nausea, visual blurring. Cardiovascular: Tachycardia, palpitations, angina, Adams-Stokes attacks, pulmonary edema, hypertension, hypotension, ventricular arrhythmias, tachyarrhythmias. Respiratory. Dyspnea.
Terbutaline
Terbutaline is a non-catecholamine and, therefore is resistant to COMT. Chemically terbutaline is N-tert-butyl-N-[2-(3,5-dihydroxyphenyl)-2-hydroxymethyl] amine.
- It is a selective β2-adrenergic receptor agonist resulting in smooth muscle relaxation. It is a fast-acting bronchodilator.
- Terbutaline is prepared by reduction of 2-(start-butylamine)-3′, 5′-dihydroxyacetophenone by catalytic hydrogenation.
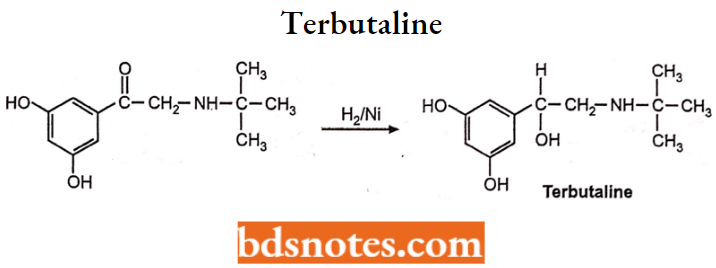
The effect of this medicine can be observed within 30-45 minutes and is effective for up to 4-8 hours upon oral administration.
Uses: Terbutaline is a bronchodilator medicine that is used to relieve symptoms such as wheezing, shortness of breath, chest tightness, breathing difficulties, coughing, etc. associated with asthma. It is used to treat an acute attack of asthma as well as for prevention of further asthma attacks.
Salbutamol (Albuterol)
Salbutamol or albuterol are selective beta-2-adrenergic bronchodilators and structurally are the replacement of the meta OH of the catechol structure with a hydroxymethyl group.
- Salbutamol is sold as a racemic mixture. The (R)-(-)-enantiomer is responsible for the pharmacologic activity; the (S)-(+)-enantiomer blocks metabolic pathways.
- Chemically, it is (RS)-4-[2-(tert-butylamino)-l-hydroxyethyl]-2-(hydroxymethyl) phenol.
- It is resistant to MAO and COMT metabolism due to the presence of hydroxymethyl moiety and bulky N-substitutions.
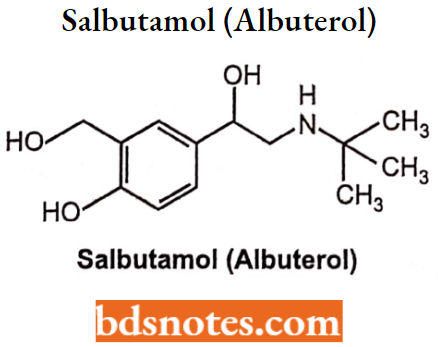
Salbutamol is a bronchodilator, β2 adrenoreceptor stimulant, with some β1 effects at high dosage levels.
- β1 stimulation causes an increase in heart rate, contractility, and excitability.
- β2 stimulation causes bronchodilatation and vasodilatation.
- It causes uterine smooth muscle relaxation and is used to help prevent premature labor.
Mechanism of Action:
The prime action of beta-adrenergic drugs is to stimulate adenyl cyclase, the enzyme that catalyzes the formation of cyclic-3′,5′-adenosine monophosphate (cyclic AMP) from adenosine triphosphate (ATP). The cyclic AMP thus formed mediates the cellular responses.
Salbutamol is prepared from an acetophenone derivative as follows:
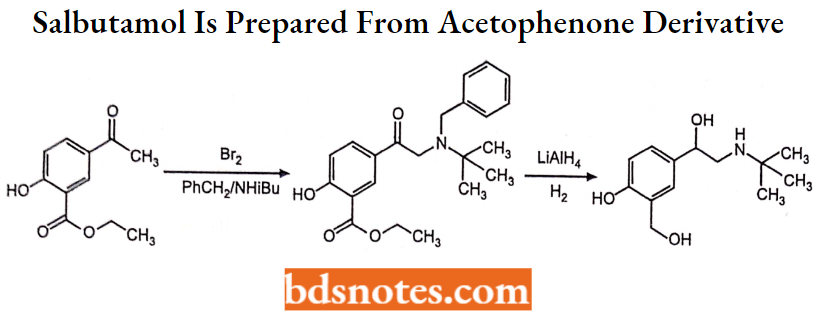
Uses: This drug relaxes the smooth muscle in the lungs and opens airways to improve breathing. It is used to treat asthma, chronic bronchitis, and emphysema.
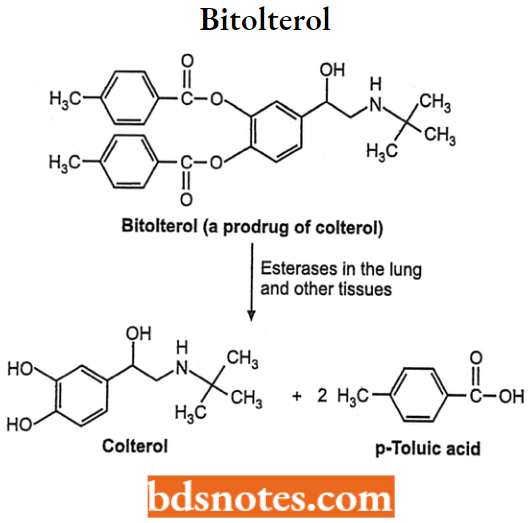
Bitolterol
Bitolterol, (3-4 diester colterol) is a new β2-adrenergic agonist. Since it is itself biologically inactive, bitolterol is considered a pro-drug.
- When administered it is activated within the lung by esterase hydrolysis to the active compound colterol catecholamine A/-t-butyl-arterenol).
- Chemically, it is [4-(l-Hydroxy-2-tert-butylamino-ethyl)-2-(4-methylbenzoyl)oxy-phenyl] 4-methylbenzoate.
- The presence of two p-toluic acids made the drug more lipophilic than the parent drug. It has a longer duration of action than isoproterenol and is metabolized by COMT and conjugation.
Mechanism of Action:
- Bitolterol is an adrenergic beta-2 agonist. Asthma results from a narrowing of the bronchial tubes. This narrowing is caused by muscle spasms and inflammation within the bronchial tubes.
- Agonist of the beta-2 adrenergic receptors by bitolterol leads to the relaxation of smooth muscles surrounding these airway tubes which then increases the diameter and ease of airflow through the tubes.
Uses: In bronchial asthma and reversible bronchospasm.
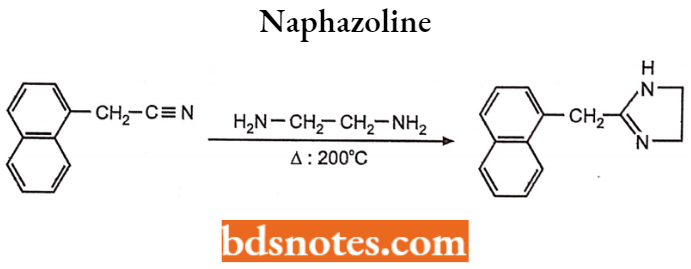
Naphazoline
Naphazoline is a rapid and direct-acting sympathomimetic drug and exists in an ionized form at physiological pH due to the basic nature of the imidazole ring (pH = 9-10).
- Chemically it is 2-(l-naphthylmethyl)-2-imidazoline and an imidazoline derivative. It is partial agonist at both cc1 and a2-adrenergic receptors.
- Lipophilic substituents on the phenyl ring are important for oci-selectivity. Naphazoline is used to induce systemic vasoconstriction, thereby decreasing nasal congestion and inducing constriction around.
- The conjunctiva also constricts the smaller arterioles of nasal passages, producing a decongesting effect. Naphazoline ophthalmic causes constriction of blood vessels in the eyes.
- It also decreases itching and irritation of the eyes. It is prepared by strong heating of 1-naphthaleneacetonitrile with ethylenediamine monochloride at 200°C.
Uses: It is used as a local vasoconstrictor for the relief of nasal congestion due to allergic manifestation. Used for the relief of ocular congestion and blepharospasm.
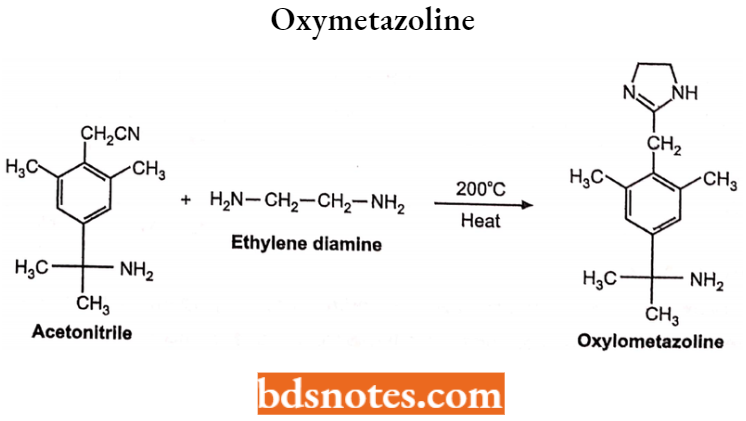
Oxymetazoline
Oxymetazoline is a direct-acting sympathomimetic drug. Chemically it is 3-(4,5-dihydrolH-imidazol-2-ylmethyl)-2,4-dimethyl-6-tert-butyl-phenol and shows selective a2-receptor agonist activity.
Uses: As a topical decongestant in the form of nasal spray (Otrivin).
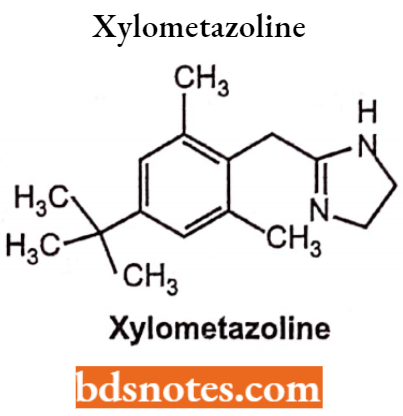
Xylometazoline
Chemically, it is 2-[(4-tert-butyl-2,6-dimethylphenyl)methyl]-4,5-dihydro-lH-imidazole. It is a selective α2-adrenergic receptor and stimulates the blood vessels of the nose.
Uses: As a decongestant during the allergy or infection of the nasal passage.
Indirect Acting Sympathomimetic Agents
Indirect-acting sympathomimetic drugs are those that act indirectly to increase the concentration of the endogenous neurotransmitter by causing the release of endogenous NE.
- These drugs enter the nerve endings by active uptake and displace NE from its storage granules. This class is of non-catecholamines, they are similar to phenylethylamine with some structural modifications.
- These compounds are resistant to COMT and MAO enzymes due to the lack of phenolic hydroxyl groups and the presence of a-methyl groups. These compounds pass more readily through blood blood-brain barrier because of increased lipophilicity.
Hydroxyamphetamine
Hydroxyamphetamine are indirect sympathomimetic agents and derivatives of amphetamines i.e. a metabolic product of amphetamine. It is chemically 4-(2-aminopropyl) phenol. It is an α-receptor and stimulates the sympathetic nervous system.
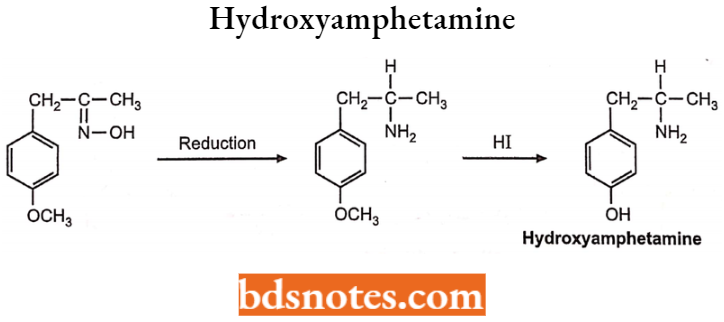
Mechanism of Action:
Hydroxyamphetamine hydrobromide is an indirect-acting sympathomimetic agent which causes the release of norepinephrine from adrenergic nerve terminals.
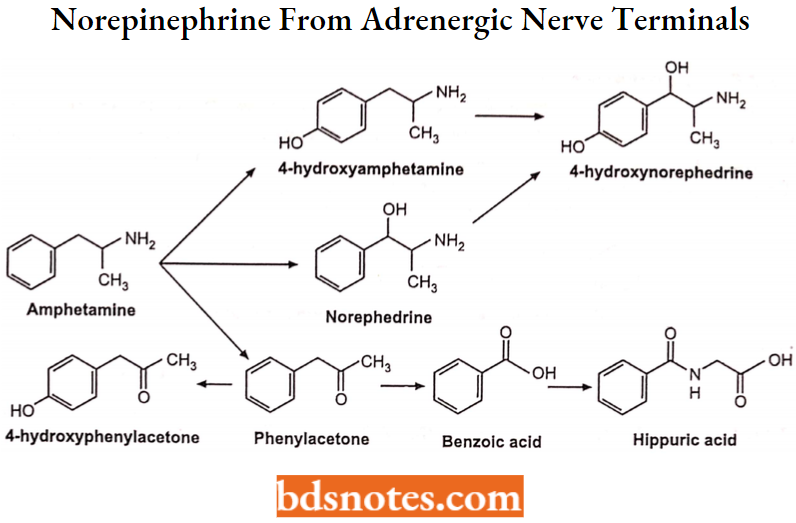
Metabolic Pathways of Amphetamine in Humans:
Uses: It is a sympathomimetic and anticholinergic combination and relaxes the muscles of the eye by dilating the pupil (mydriasis).
In narcolepsy (sudden attack of sleep in completely inappropriate situations). Also used in children with hyperkinetic syndrome and the treatment of obesity.
Pseudoephedrine
Chemically, it is (S, S)-2-methylamino-l-phenylpropan-l-ol. Pseudoephedrine is a diastereomer of ephedrine and gets oxidized to methcathinone or reduced into methamphetamine.
- Whereas ephedrine has a mixed mechanism of action, L-(+)- pseudoephedrine acts mostly by an indirect mechanism and has virtually no direct activity.
- The structural basis for this difference in mechanism is the stereochemistry of the carbon atom possessing the β-OH group.
- It is a sympathomimetic agent and has a direct effect on adrenergic receptors, (a-adrenergic receptors present on the walls of blood vessels).
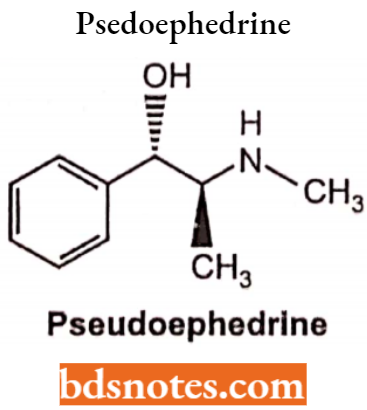
Uses: Used as vasoconstrictor, to treat nasal and sinus congestion, or congestion of the tubes that drain fluid from your inner ears.
Propylhexedrine
Chemically it is (±)-1-cyclohexyl-N-methylpropan-2-amine. It is an analog of amphetamine in which the aromatic ring has been replaced by the cyclohexane ring.
It produces vasoconstriction and a decongestant effect. It inhibits MAO and is metabolized by COMT and thus increases NE to show sympathetic activity.
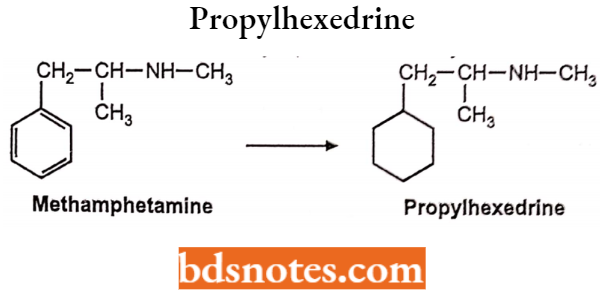
Uses: It is used for the relief of congestion due to colds, allergies, and allergic rhinitis and its euphoric effects.
Mixed Acting Sympathomimetic Agents
Mixed-acting adrenergic agonists are compounds that cause activation of adrenergic receptors by both direct binding as well as release of endogenously stored norepinephrine from presynaptic terminals.
Ephedrine is the prototype mixed-acting agonist. They have no hydroxyls on the aromatic ring but do have a (3-hydroxyl group.
Ephedrine
Chemically, it is 2-methylamino-l-phenyl propane-l-ol. It has both a and (3-adrenergic agonistic effect. Ephedrine is a central nervous system stimulant.
- It is not metabolized by either MAO or COMT and therefore has more oral activity and longer duration of action than E. Ephedrine has two asymmetric carbon atoms, so it has four isomers.
- D(-) isomer is the most active of the four isomers as a presser amine because it has the correct (1R,2S) configuration for optimal direct action at adrenergic receptors.
- Lacking phenolic OH groups, ephedrine is less polar and thus crosses the BBB far better than other CAs.
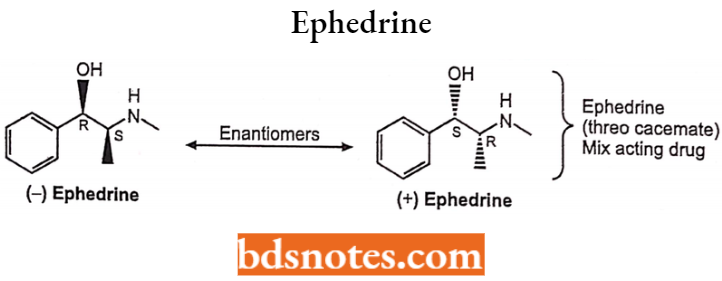
Uses: Used as a bronchodilator, nasal decongestant, orthostatic hypotension, or myasthenia gravis.
Metaraminol
Chemically, it is 3-[-2-amino-l-hydroxy-propyl] phenol. It is an α-adrenergic receptor agonist with some β-receptor effect.
It has a mixed mechanism of action and can be used parenterally as a vasopressor in the prevention of acute hypertension state occurring with spinal anesthesia.
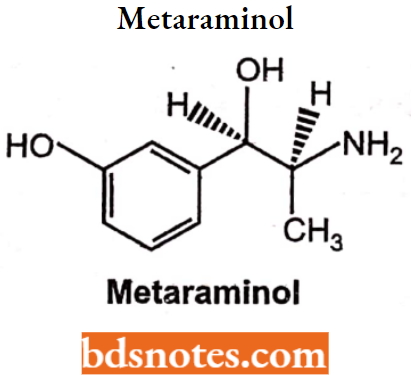
Uses: It is a potent sympathomimetic amine used in the prevention and treatment of hypotension, particularly as a complication of anesthesia.
Adrenergic Antagonists or Adrenerolytic
A sympatholytic (or adrenerolytic) drug is a medication that inhibits the transmission of impulses from the postganglionic functioning of the sympathetic nervous system or inhibits the transmission of nerve impulses in the sympathetic nervous system.
They can block at three different levels:
- Peripheral sympatholytic drugs (a and (3 receptor antagonists) block the action of NA at the effector organ (heart or blood vessel).
- Ganglionic blockers block impulse transmission at the sympathetic ganglia.
- Centrally acting sympatholytic drugs that block sympathetic activity within the brain.
Classification Of Adrenerolytic Or Sympatholytic (SL)
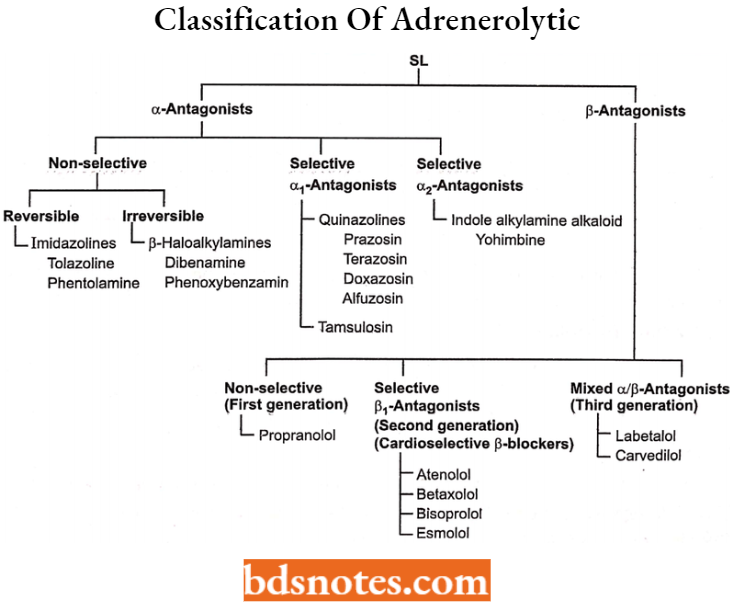
Alpha (α) – Adrenergic Antagonists Or (α) – Adrenergic Blockers
Adrenergic blocker agents prevent the response of effector organs to endogenous as well as exogenous adrenaline and nor-adrenaline.
- The alpha receptor is one of the two receptors through which the sympathetic nervous system. The α-adrenergic receptors are further subdivided into two distinct types: α1, most of which are located post-junctionally on the vascular smooth muscle cell producing vasoconstriction.
- And α2, which is located prejunctionally on the sympathetic nerve ending has negative feedback on norepinephrine release into the synapse.
Tolazoline
Tolazoline belongs to the synthetic non-selective competitive alpha-adrenergic blocking agents known as imidazoline derivative. It is a mixed alpha-1 and alpha-2 adrenergic receptor antagonist.
Chemically it is lH-lmidazole, 4,5-dihydro-2-(phenylmethyl) monohydrochloride and structurally similar to the imidazoline a-agonist like nephazole and xylometazoline.
Synthesis:
It is prepared by condensation of an amino ether (obtained by methanolysis of phenyl acetonitrile) with ethylene diamine.
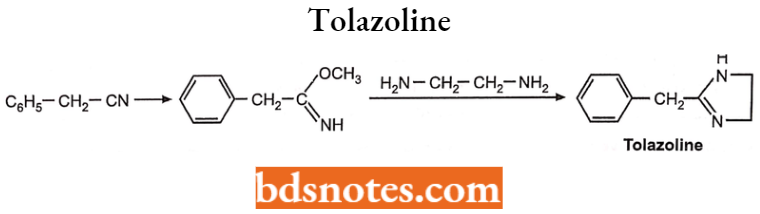
Use: Tolazoline has been used in the treatment of persistent pulmonary hypertension in the newborn.
Phentolamine
Phentolamine is a reversible non-selective a-adrenergic antagonist. Chemically it is 3-[(4,5-Dihydro-lH-imidazol-2-ylmethyl)(4-methylphenyl)amino]phenol. It is an imidazoline derivative and causes vasodilation and alters the effect of adrenergic drug.
Synthesis:
N-(4-methylphenyl)-3-hydroxyaniline on condensation with hydrogen cyanide and formaldehyde gives N-(4-methylphenyl)-3-hydroxy anilinoacetonitrile which on further treatment with ethylene diamine gives phentolamine.
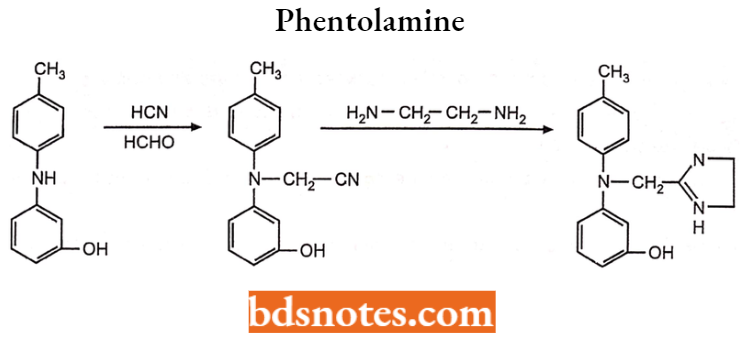
Uses: It is used in the treatment of hypertension and hypertensive emergencies, pheochromocytoma, and vasospasm of Raynaud disease. As a drug of choice when no one responds to benzodiazepines and calcium-channel blockers.
Phenoxybenzamine
- Phenoxybenzamine is a non-selective, irreversible alpha blocker having a long duration of action because it forms a permanent covalent bond with adrenergic receptors.
- It has been used to treat hypertension and as a peripheral vasodilator. Chemically, it is benzyl-N- (2-chloroethyl)-l-phenoxypropan-2-amine. It also blocks acetylcholine, histamine, and serotonin receptors and shows vasodilatory effects.
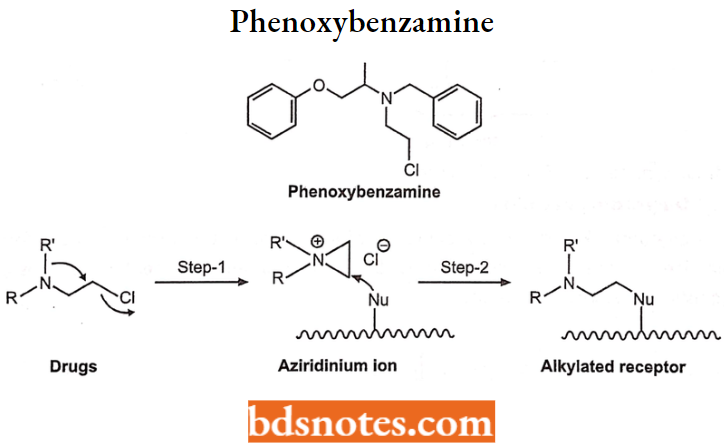
Uses: Used to treat hypertension caused by pheochromocytoma and pulmonary edema.
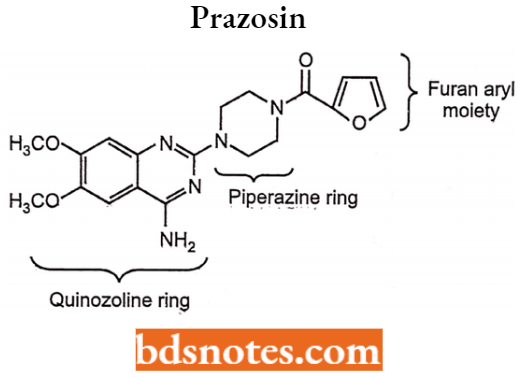
Prazosin
Chemically, it is [4-(4-Amino-6,7-dimethoxy-2-quinazolinyl)-l-piperazinyl](2-furyl) methanone. It is quninazoline derivative, piperazine ring with acyl moiety.
It is a highly selective competitive antagonist of the α1-adrenergic receptor. It lowers blood pressure, heart rate, and cardiac output.
Effects of Prazosin and Analogs on the Cardiovascular System:
- Prazosin and its analogs are selective ai-receptor blockers used to treat hypertension. These agents have similar cardiovascular actions, differing only in pharmacokinetic parameters.
- Doxazosin, trimazosin and terazosin are more widely used than prazosin.
- These agents relax the smooth muscle associated with arteries and veins.
- This results in a decrease in systemic arterial blood pressure due to a decrease in peripheral vascular resistance and venous return.
- The reduction in arterial blood pressure does not result in a significant increase in heart rate.
- Treatment with these drugs can result in fluid retention as a response to the lowering of blood pressure. Thus the drugs can be prescribed with a diuretic in the treatment of hypertension.
- May have beneficial effects on lipid profiles by increasing HDL cholesterol and decreasing LDL cholesterol.
Uses: Hypertension, Raynaud’s disease.
Dihydroergotamine
It is an ergot alkaloid and is a derivative of lysergic acid. It is a semi-synthetic form of ergotamine. It affects adrenoreceptors, DA, and 5-histamine receptors. It possesses α – antagonistic and vasoconstrictor actions.
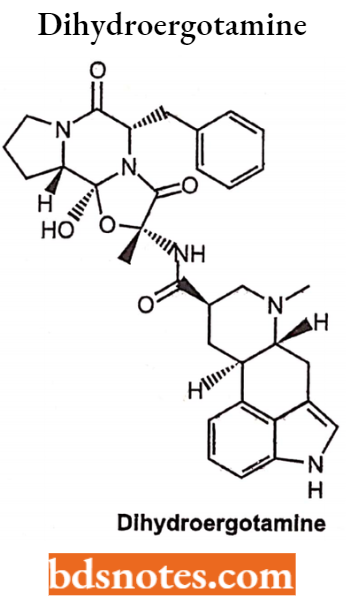
Mechanism of Action:
Dihydroergotamine binds with high affinity to 5-HT1Dα and 5-HT1Dβ receptors. It also binds with high affinity to serotonin 5-HT1A, 5-HT2A, and 5-HT2C receptors, noradrenaline α2A, α2B, and α, receptors, and dopamine D2L and D3 receptors.
- The therapeutic activity of dihydroergotamine in migraine is generally attributed to the agonist effect at 5-HT1D receptors.
- Two current theories have been proposed to explain the efficacy of 5-HT1D receptor agonists in migraine. One theory suggests that activation of 5-HT1D receptors located on intracranial blood vessels leads to vasoconstriction, which correlates with the relief of migraine headaches.
- The alternative hypothesis suggests that activation of 5-HT1D receptors on sensory nerve endings of the trigeminal system results in the inhibition of pro-inflammatory neuropeptide release.
Uses: Effective in headache and severe migraine pain. It can be administered by 4, subcutaneously, or by nasal spray.
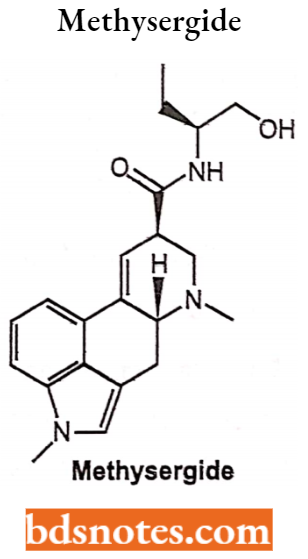
Methysergide
Methysergide is a partially synthetic compound structurally related to lysergic acid butanolamide, well known as methylergonovine.
- Chemically, methysergide maleate is designated as ergoline-8-carboxamide, 9,10-didehydro-N-[l-(hydroxymethyl)propyl]-l,6- dimethyl-(8b)-(Z)-2-butenedioate.
- Methylation in the number position of the ring structure enormously enhances the antagonism to serotonin that is present to a much lesser degree in the partially methylated compound (methylergonovine maleate) as well as altering other pharmacologic properties.
Mechanism of Action:
Methysergide is a serotonin antagonist and acts on the central nervous system (CNS), which directly stimulates the smooth muscle leading to vasoconstriction. Some alpha-adrenergic blocking activity has been reported.
Suggestions have been made by investigators as to the mechanism whereby methysergide produces its clinical effects, but this has not been finally established, although it may be related to the anti-serotonin effect.
Uses: To treat chronic migraine and cluster headaches (prevention or reduction) of intensity and frequency of vascular headaches.
Beta (β) – Adrenergic Blockers
These agents antagonize the effect of the catecholamine at a β-adrenergic receptor. They block the effects of endogenous and exogenous catecholamine.
These agents decrease heart rate (chronotopic) and force of contraction (ionotropic) (β1 effect) and also block the sympathetic stimulation of kidney renin release (β2 effect).
β-blockers are classified into the following groups based on their structure.
Non-selective β-blockers: Propranolol, 4-hydroxypropranolol.
Selective β1 blockers: Metoprolol, atenolol.
Mixed α/β blockers: Labetalol, carvedilol.
Sar Of Beta Blockers
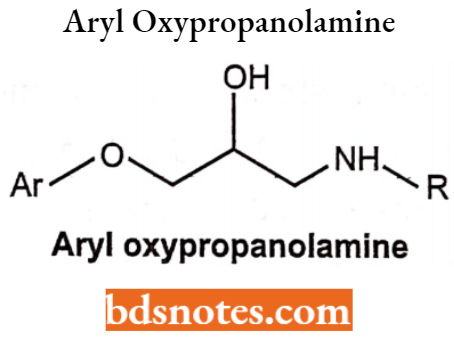
Most of the β-blockers are similar to β agonists and are in a chemical class of aryloxypropanolamines, only the catechol ring is substituted by different rings to enhance the antagonist activity.
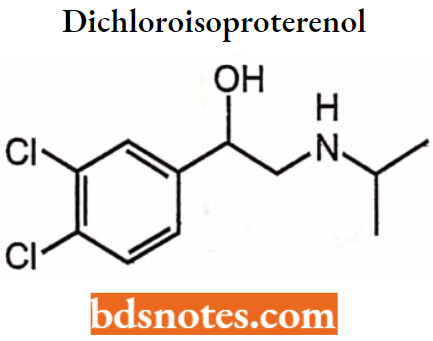
Substituting the 3’4′ di-hydroxyl group with two chlorine groups leads to dichloro isoproterenol. Unfortunately, dichloro isoproterenol has a poor antagonist activity but a good agonist activity.
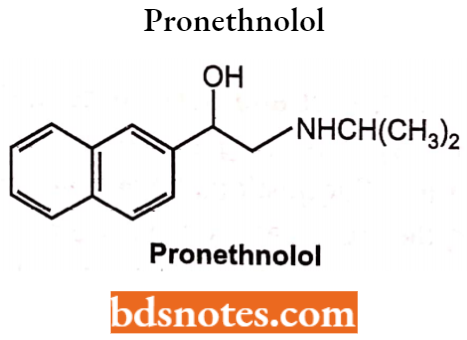
The Catechol group has been replaced by naphthalene to give compounds like pronethnolol. It was a weak antagonist and was withdrawn from clinical testing because it tended to form tumors (mice).
An assumption is made that the oxymethylene bridge (O-CH2-) group is responsible for the potent antagonist activity, and the side chain has been moved from C2 of the naphthyl group to the C1 position.
It has been introduced in between the aromatic ring and ethylamine side chain to give more potent β blockers. For example, propranolol.
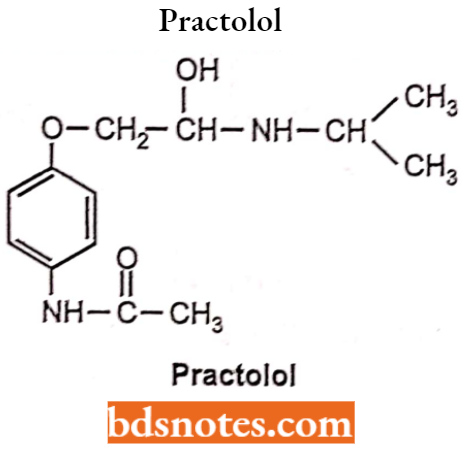
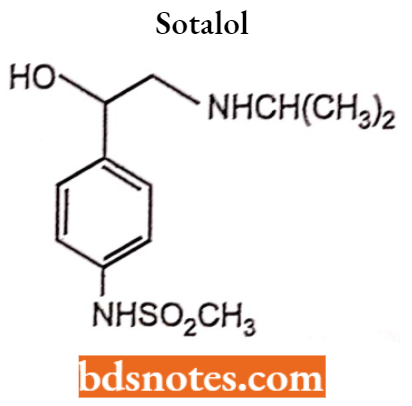
However, most of the drugs containing the 0-CH2– group are β-agonist. The presence of para substituent on the aromatic ring along with the absence of meta substituent gives β1 selective antagonist For Example. Practolol (cabonamide). Sotalol (Methylsulphonamide).
The bulky aliphatic groups such as tertiary butyl and isopropyl groups (which give optimal basicity or nucleophilicity to the amine group for receptor activity) are normally found on the amino functional group of the oxypropanololamine β receptor antagonist.
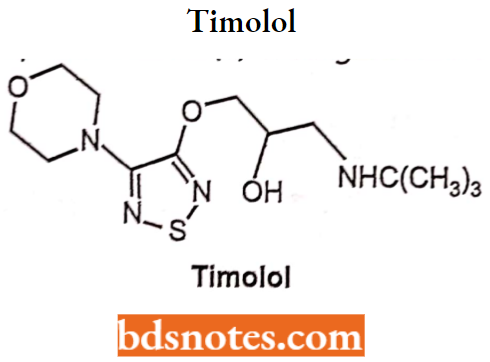
B-blockers show high stereoselectivity and for optimal activity, the carbon of the side chain possessing hydroxyl group must be in (S)-configuration. For Example. Timolol, levobunolol.
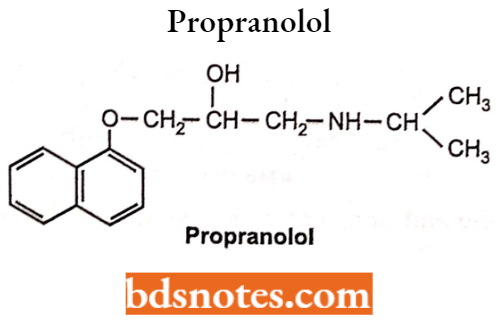
Propranolol
Chemically, it is l-(-isopropylamino)-3-(-l-napthloxy)-2-propranol. It belongs to the class of oxy-propranolol amine. It is a non-seiective β1 and β2-blocker. Its (S)-isomer is more potent.
- It has structural similarities to pronethalol (OCH3– bridge between aromatic and ethylamine side chain). It has relatively high lipid solubility and allows distribution to the CNS.
- One of the metabolites is 4-hydroxy propranolol which shows a potent β-antagonist because of its half-life, a single dose from 3-4 hours to 5-6 hours.
Synthesis: It can be synthesized from naphthol and epichlorohydrin in the presence of hydrochloric acid.
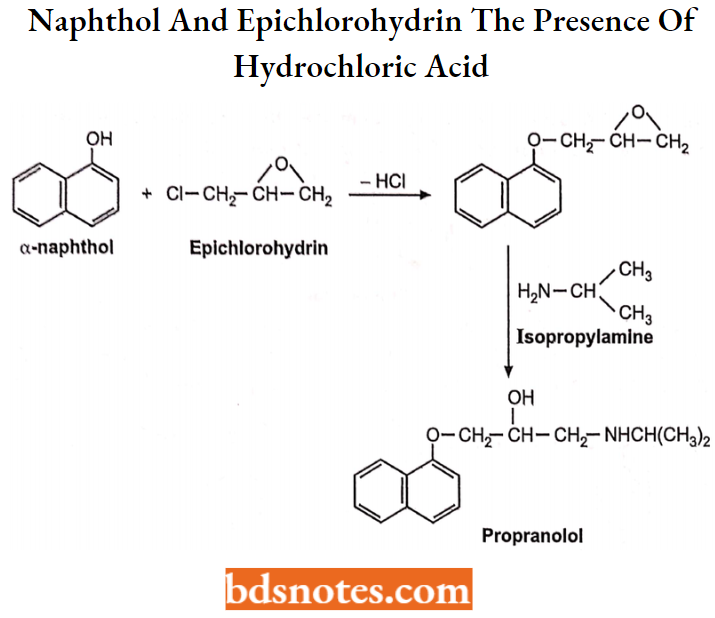
Uses: In the treatment of hypertension, cardiac arrhythmia, angina pectoris, and myocardial infarction.
Metibranolol
A beta-adrenergic antagonist is effective for both beta-1 and beta-2 receptors. Chemically, it is 4-{[-2-hydroxy-3-(isopropylamino)propyl]oxy}-2,3,6-trimethylphenyl acetate.
It is a non-selective (3-blocker and is metabolized into desacetylmetipranolol.
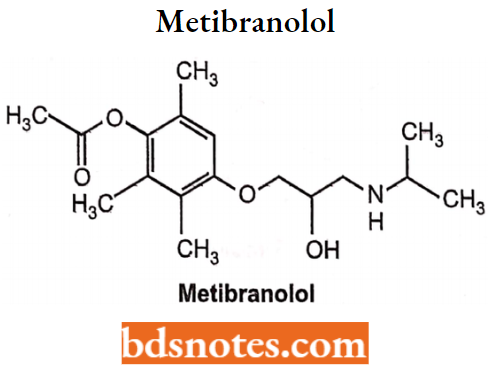
Uses: It is used as an antiarrhythmic, antihypertensive, and antiglaucoma agent.
Atenolol
Chemically atenolol (R S)-2-[4-(2-Hydroxy-3-isopropylaminopropoxy)-phenyl]- acetamido) is a chiral drug from the group of the selective β1– adrenoreceptor blockers.
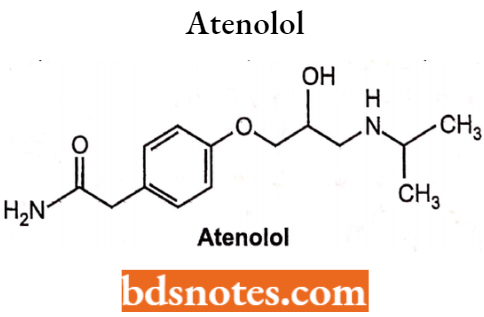
It has low lipid solubility and does not cross blood blood-brain barrier. Thus it has very less CNS side effects.
Synthesis:
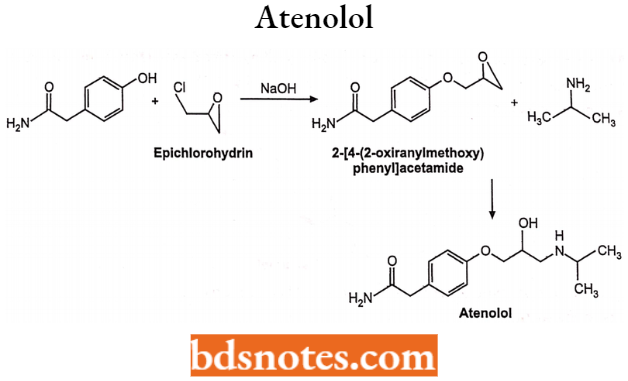
Betaxolol
A cardioselective beta-l-adrenergic antagonist with no partial agonist activity. Chemically, it is l-{4-[2-(cyclopropylmethoxy) ethyl]-phenoxy}-3-(isopropylamino) propan-2-ol. It is a selective (3-blocker and shows more affinity towards 1-receptor.
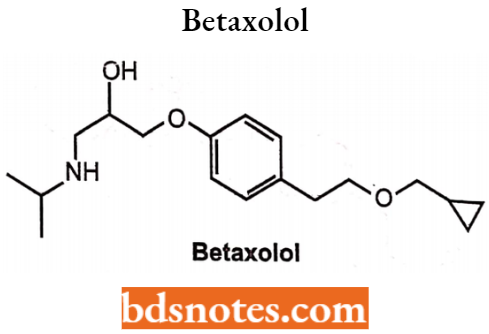
Mechanism of Action:
- Betaxoloi selectively blocks catecholamine stimulation of beta-l-adrenergic receptors in the heart and vascular smooth muscle.
- This results in a reduction of heart rate, cardiac output, systolic and diastolic blood pressure, and possibly reflex orthostatic hypotension.
- Betaxoloi can also competitively block beta-2-adrenergic responses in the bronchial and vascular smooth muscles, causing bronchospasm.
Uses: In hypertension and glaucoma.
Bisoprolol
Bisoprolol is a cardioselective β1-adrenergic blocking agent used for secondary prevention of myocardial infarction (MI), heart failure, angina pectoris, and mild to moderate hypertension.
- Bisoprolol is structurally similar to metoprolol, acebutolol, and atenolol in that it has two substituents in the para position of the benzene ring.
- The β1-selectivity of these agents is thought to be due in part to the large substituents in the para position. It also shows little activity against β2-adrenergic receptors of the lungs and vascular smooth muscle.
- Bisoprolol possesses a single chiral center and is administered as a racemic mixture. Only I-bisoprolol exhibits significant [3-blocking activity. Chemically, it is l-{4-[(2- isopropoxyethoxy) methyl]phenoxy}-3-(isopropylamino)propan-2-ol.
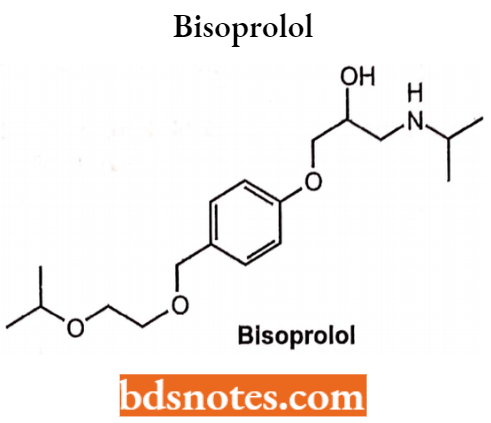
Uses: To treat hypertension, heart attacks, and kidney problems.
Esmolol
Esmolol (trade name Brevibloc) is a cardioselective Pi receptor blocker with rapid onset, a very short duration of action, and no significant intrinsic sympathomimetic or membrane stabilizing activity at therapeutic dosages.
Chemically it is methyl (RS)-3-{4-[2-hydroxy-3- (propan-2-ylamino)propoxy]phenyl}propanoate. It is a short-acting β-blocker with a rapid onset of action.
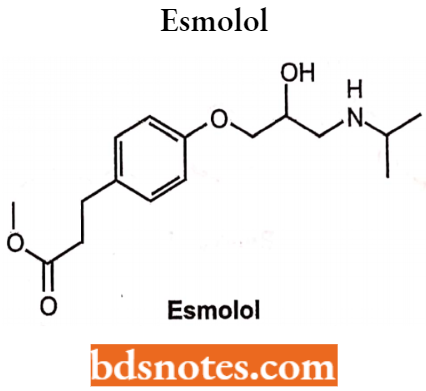
Uses: Controlling heart rate during surgery in operation.
Metoprolol
Chemically, it is l-[4-(2-Methoxyethyl)phenoxy]-3-[(propan-2-yl)amino]propan-2-ol. It is a selective β1-blocker.
It is lipophilic with intrinsic sympathomimetic activity. It reduces heart rate, heart contractility, and blood pressure.
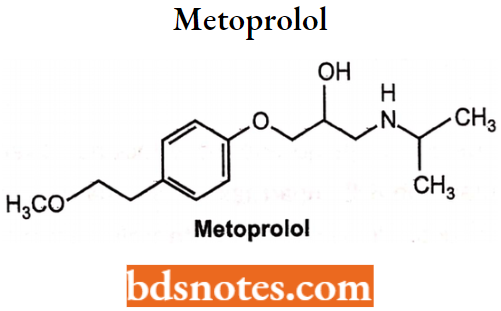
Uses: In angina pectoris, myocardial infarction, and hypertension. Also used in diabetic patients.
Labetalol
Chemically it is 2-hydroxy-5-[l-hydroxy-2-[(4-phenylbutan-2-yl)amino]ethyl] benzamide. It has both α and β adrenergic blocking activity.
In the short term, it can be used to reduce systemic vascular resistance whereas in the long term, it reduces the cardiac output.
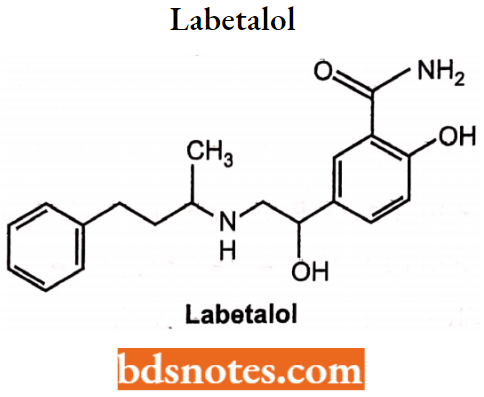
Uses: Used for the management of hypertension emergencies.
Carvedilol
Carvedilol is a non-selective beta blocker indicated in the treatment of mild to moderate congestive heart failure (CHF).
It blocks beta-1 and beta-2 adrenergic receptors as well as the alpha-1 adrenergic receptors. Chemically, it is [3-(9H-carbazol-4-yloxy)-2-hydroxypropyl] [2-(2-methoxyphenoxy)ethyl]amine.
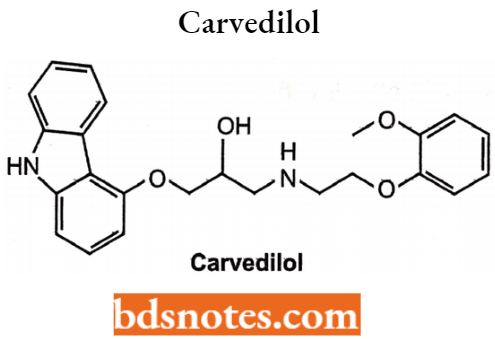
Uses: To treat congestive heart failure and hypertension.
Leave a Reply