Dental Plaster
Add in detail about the setting reaction of dental plaster and add a note on the uses of different gypsum products.
Or
Write in detail the setting reaction of calcium sulphate dihydrate.
Answer:
Setting Reaction of Dental Plaster:
When 1 g mol of calcium sulfate hemihydrates reacts with 1.5 g mol of water, the hemihydrates reverts to dihydrate forming 1 g mol of calcium sulfate dihydrate by hydration, with liberation of 3900 calories of heat. Here the heat which is evolved in the reaction is equal to the heat originally used in calculations.
- (CaSO4)2.H2O + 3H2O→ 2CaSO4.2H2O + unreacted (CaSO4)2.½ H2O + heat
- Hemihydrate + Water → Dihydrate + unreacted hemihydrates + 3900 cal/g mol
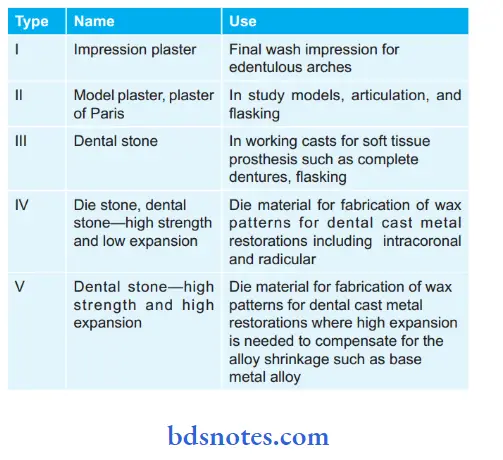
Uses of Different Gypsum Products:
Leave a Reply