Cholinergic Drugs And Related Agents Neurotransmitters (NT)
A neurotransmitter is a chemical messenger used by neurons to communicate in one direction with other neurons. Communication between neurons means recognition of the acceptor for a specific chemical messenger.
- They are synthesized primarily in the nerve terminals stored in vesicles of nerve terminals and are released into electrocellular space using Ca2+.
- All neurotransmitters are synthesized at the axon terminals and stored in synaptic vesicles. These synaptic vesicles release neurotransmitters when the presynaptic neuron’s electrical properties change sufficiently (i.e. arrival of an action potential).
- Neurotransmitters are released from the vesicles into a tiny space between neurons called the synapse. A bit of released neurotransmitter diffuses across the synaptic space and binds to receptors on the adjacent neuron, the whole process takes about one millisecond.
- When a neurotransmitter binds to an acceptor on another neuron, ion channels open and ions move in or out of that neuron. This causes a net change in the electrical properties (membrane potential of that neuron and determines its activity).
The change may be inhibitory or excitatory and is determined by the receptor on the post-synaptic neuron.
Cholinergic Neurotransmitters
Acetylcholine is a neurotransmitter parasympathetic nervous system. Acetylcholine (ACh) is the chemical transmitter at both pre and postganglionic synapses in the parasympathetic system.
- Ach is also the neurotransmitter at sympathetic preganglionic synapses, some sympathetic postganglionic synapses, the neuromuscular junction (somatic nervous system), and at some sites in the CNS.
- Acetylcholine is the most widespread autonomic transmitter present in the body. Cholinergic transmission can occur through muscarinic (G protein-coupled) or nicotinic (ionotropic) receptors and the muscarinic receptor is terminated by the action of cholinesterases.
Synthesis Of Acetylcholine (ACh)
The major steps in the synthesis of acetylcholine are illustrated in the figure below. Acetylcholine was also one of the first neurotransmitters identified due to its presence in the peripheral nervous system.
- Acetylcholine is the neurotransmitter at all the motor-targeted synapses in the somatic branch of the peripheral nervous system.
- In addition, it is the neurotransmitter at the parasympathetic postganglionic-target synapses in the autonomic nervous system. (Recall that norepinephrine is the neurotransmitter at the corresponding sympathetic synapses).
- In the central nervous system, acetylcholine is best known for its role in memory and learning. For example, Alzheimer’s disease is associated with a breakdown in acetylcholine neurons.
Acetylcholine is synthesized locally in the choleric nerve ending by the following pathway:
- An amino acid Serine is the precursor for the synthesis of acetylcholine. Serine undergoes decarboxylation in the presence of serine decarboxylase to choline.
- Choline is actively taken up by the axonal membrane by a Na+, choline cotransporter, and acetylated with the help of ATP and coenzyme-A enzyme choline acetyltransferase present in the axoplasm.
- Most of the ACh is stored in ionic solution within small synaptic vesicles, but some free Ca2+ is also present in the cytoplasm of cholinergic terminals. Active transport of ACh into synaptic vesicles is affected by another carrier.
Release of ACh:
When Ca2+ ion concentration is high in intracellular with the cell membrane and release of this content into the synaptic cleft.
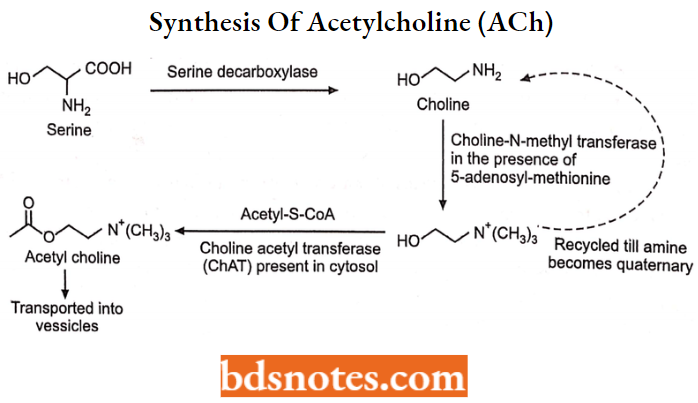
Degradation of ACh:
Acetylcholinesterase cleavages ACh to choline and acetate
Cholinergic Receptors
The acetylcholine (ACh) receptor, at the neuromuscular junction, is a neurotransmitter-gated ion channel that has been fine-tuned through evolution to transduce a chemical signal into an electrical signal with maximum efficiency and speed.
- It is composed of three similar and two identical polypeptide chains, arranged in a ring around a narrow membrane pore.
- Central to the design of this assembly is a hydrophobic gate in the pore, more than 50 A° away from sites in the extracellular domain where ACh binds.
There are two main types of cholinergic receptors.
- Nicotinic cholinergic receptor.
- Muscarinic cholinergic receptor.
Nicotinic and muscarinic ACh receptors are named after two substances that bind to those receptor subtypes. Nicotine binds to nicotinic, but not muscarinic ACh receptors.
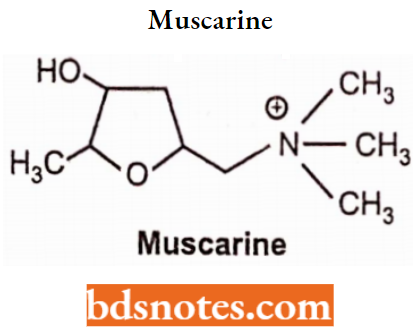
Muscarinic receptors are named after Muscarine, found in some species of mushrooms.
- Nicotinic receptors, when ACh interacts with a nicotinic ACh receptor, it opens a Na+ channel and Na+ ions flow into the membrane. This causes a depolarization, and results in an EPSP.
- Thus, ACh is excitatory on skeletal muscle. The electrical response is fast and short-lived. Nicotinic receptors are mainly located in CNS automatic ganglia and neuromuscular junction (NMJ).
- Muscarinic receptors are located primarily on autonomic effector cells in heart blood vessels, eyes, smooth muscles, and glands of gastrointestinal, respiratory, and urinary tracts, sweat glands, and in the CNS.
- They are classified into sub-types according to their molecular structure, signal transduction, and ligand affinity in M1, M2, M3, M4, and M5.
In M1, M2, and M4 types of muscarinic receptors, ACh acts by stimulating or activating Gq protein and releasing phospholipase C, the G-protein activates channels or enzymes indirectly and responses are diverse, slower, and longer-lived.
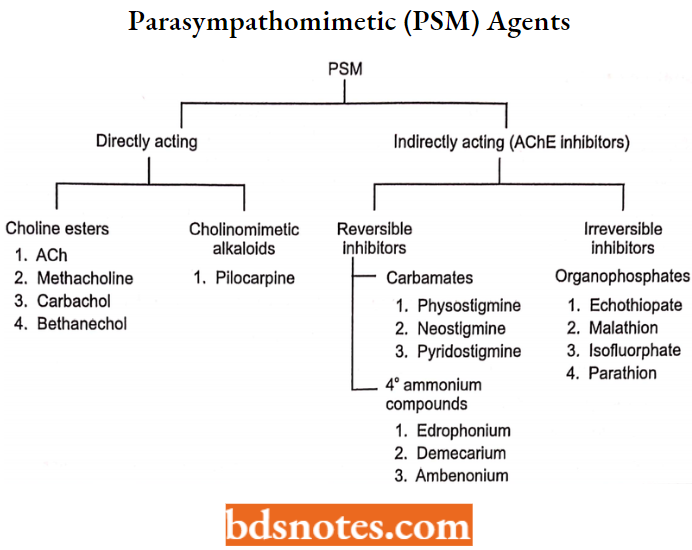
Parasympathomimetic (PSM) Agents
Compounds that mimic the action of ACh in the parasympathetic system are called cholinergic parasympathomimetic agents.
Thus these drugs stimulate the effect of cells innervated by postganglionic parasympathetic cholinergic nerves. They are classified as directly acting and indirectly acting cholinergic.
Sar Of Parasympathomimetic Or Cholinergic Agonist
Acetylcholine the ester of choline, is a quaternary ammonium compound that possesses a cationic (positively charged) part joined by a two-carbon chain to an ester group.
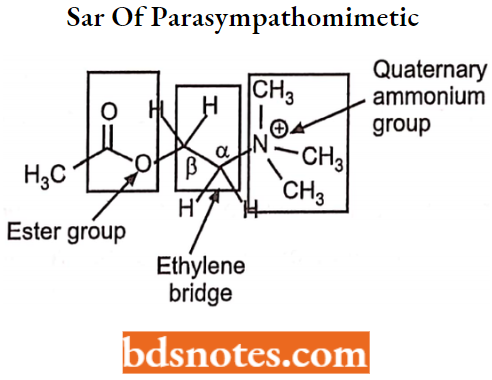
Modification of the Quaternary Ammonium Group:
The quaternary ammonium group is essential for intrinsic activity and contributes to the affinity of the molecule for the receptors, partially through the binding energy and partially because of its action as a detecting group.
- The trimethyl ammonium group is essential for the optimal functional activity. (Although some exceptions which show muscarinic activity, For Example. pilocarpine, nicotine).
- Replacement of Nitrogen (N) by other atoms like Sulphur (S), Arsenic (As), or Selenium (Se) leads to a decrease in the activity.
Placement of primary, secondary, or tertiary amine leads to a decrease the activity.
Modification of Ester Group:
- The ester group of ACh contributes to the binding of the compound to the muscarinic receptors.
- Replacement of ethyl group or bulky alkyl group leads to inactive compounds.
- Ester of higher aromatic acids possesses cholinergic antagonist activity. For Example. Bethanechol, Methancholine.
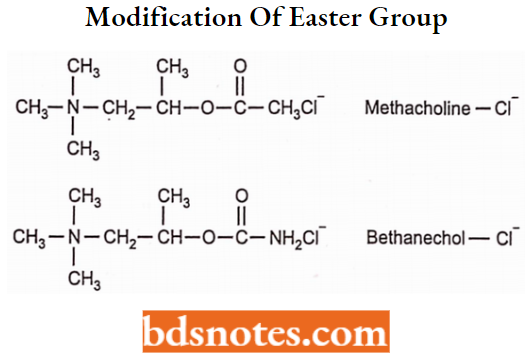
NH2 group can be introduced in place of CH3. It is more stable than carboxylate ester towards hydrolysis.
Modification of Ethylene Bridge:
For maximum muscarinic activity, there should not be more than four atoms in between the quaternary ammonium group and the ester molecule. A series of N-alkyl trimethyl ammonium salts shows optimum muscarinic activity.
- The methyl ester is rapidly hydrolyzed by cholinesterase to choline and acetic acid.
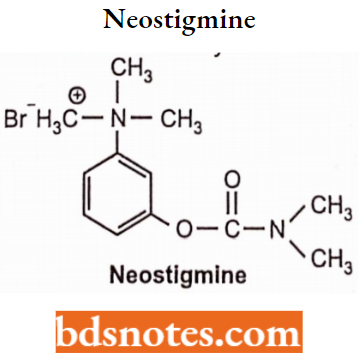
- To reduce susceptibility to hydrolysis, carbamate esters of choline were synthesized and found to be more stable than carboxylated esters. For Example. Neostigmine.
Placement of a-substitution in choline moiety results in a reduction of both muscarinic and nicotinic activity.
- Substitution at (5-position leads to compounds with a decrease in activity. For Example. methacholine.
- Replacement of the ester group with ether or ketone produces chemically stable and potent compounds. For Example. Muscarine, pilocarpine.
Directly Acting Cholinergic Drugs:
The directly acting cholinomimetic drugs are divided based on their chemical structures into:
- Choline esters and
- Cholinomimetic alkaloids
Choline Esters
Choline esters are synthetic derivatives of choline. They stimulate muscarinic receptors affecting the cardiac muscle, smooth muscle, exocrine glands, and the eye.
- Choline esters are lipid insoluble and do not readily enter the CNS (effects occur primarily in the periphery). They are highly resistant to being destroyed by acetylcholinesterase (AChE).
- The choline esters differ in their relative sensitivity to hydrolysis and in their relative nicotinic and muscarinic effects.
- Thus, methacholine, while still susceptible to hydrolysis, is much more stable than ACh, and carbachol and bethanechol are resistant to hydrolysis by AChE.
- Carbachol has a high level of nicotinic activity, methacholine with little nicotinic activity, and bethanechol essentially of no nicotinic activity.
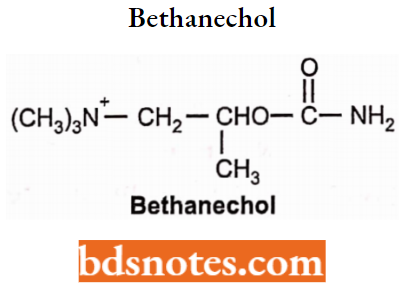
Bethanechol
- Bethanechol is a synthetic derivative of choline.
- Bethanechol is a carbonic ester of β-methylcholine.
- It is not rapidly hydrolyzed by acetylcholinesterase and has strong muscarinic actions but little nicotinic actions.
- The presence of CH3 gives prolonged activity due to steric hindrance.
- It produces smooth muscle contractions. It is not well absorbed from the GI tract (large doses required). It can be given subcutaneously but not by IM or 4 routes because of severe adverse effects.
- It is used for the relief of urinary retention and abdominal distention after surgery.
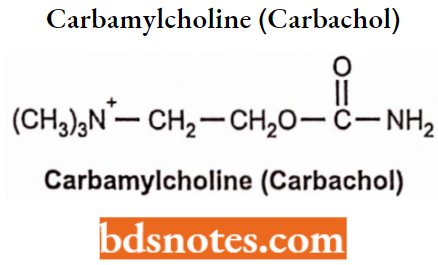
Carbachol
- Carbachol is an ester of carbamic acid. In carbachol, the terminal methyl group of acetylcholine is replaced by the amino (-NH2) group.
- It possesses both muscarinic as well as nicotinic properties by cholinergic receptor stimulation.
- Carbachol is a poor substrate for acetylcholine esterase and therefore is not readily hydrolyzed as acetylcholine.
- The presence of a carbonyl group in carbachol decreases the electrophilicity and forms resonance structures more easily than ACh. Thus less susceptible to hydrolysis, and more stable in an aqueous solution.
- Carbachol has a higher level of nicotinic activity than compared with methacholine.
- It reduces intraocular pressure. Carbachol is used to induce miosis (pupil constriction) and relieve intraocular pressure of glaucoma.
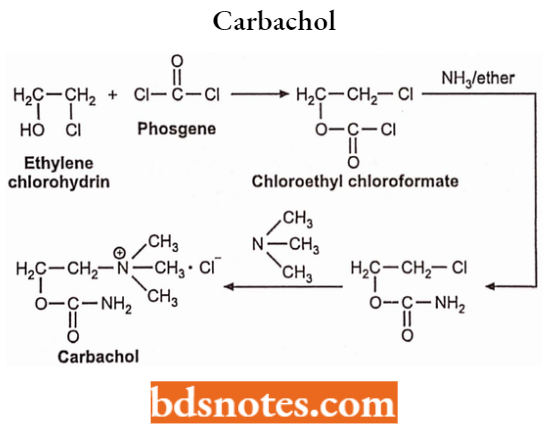
Synthesis: It can be prepared by treating phosgene with ethylene chlorohydrin to form chloroethyl chloroformate. Chloroethylchloroformate then treated in the presence of ammonia and ether gives urethane which is then heated with trimethylamine to yield carbachol.
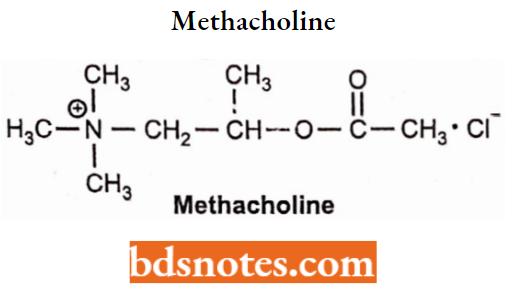
Methacholine
Methacholine is available as methacholine chloride. Chemically methacholine is 2-acetoxypropyltrimethyl ammonium chloride.
- Methacholine is still susceptible to hydrolysis but much more stable than ACh, and carbachol and bethanechol which are resistant to hydrolysis by AChE.
- Methacholine can exist as (S) and (R) enantiomers, but its muscarinic activity resides in the (S) isomer.
- Methacholine was used in the past to control supraventricular tachycardia and is replaced with ectrophonium and other, safer drugs.
- Methacholine is also used for the diagnosis of belladonna poisoning, for diagnosis of familial dysautonomia, and diagnosis of bronchial hyperreactivity.
Cholinomimetic Alkaloids
Muscarine, arecoline, and pilocarpine are natural alkaloids. The actions of these alkaloids are similar to those of choline esters and show more CNS side effects.
Pilocarpine, muscarine, and oxotremorine are relatively specific for muscarinic acetylcholine receptors.
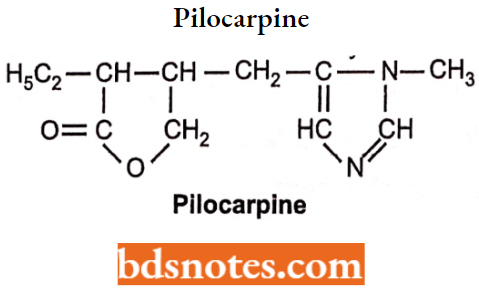
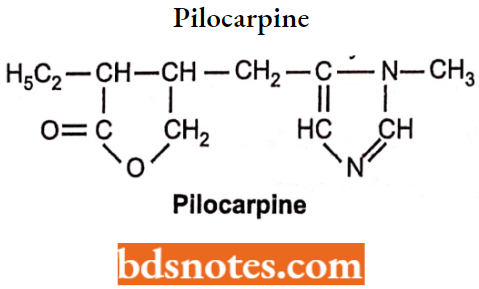
Pilocarpine
Pilocarpine is an alkaloid obtained from the dried leaflets of Pilocarpus jaborandi.
- It is available as hydrochloride and nitrate salt. Chemically it is 3-ethyldihydro-4[(1-methyl-lH-imida zol-5-yl)-methyl] furan-2(3H)-one.
- It binds to the muscarinic receptor as a non-selective agonist.
- It cannot penetrate the membrane because of bulky substituents but penetrates the eye membrane when applied topically.
- These drugs (essentially only pilocarpine) are used in the treatment of glaucoma.
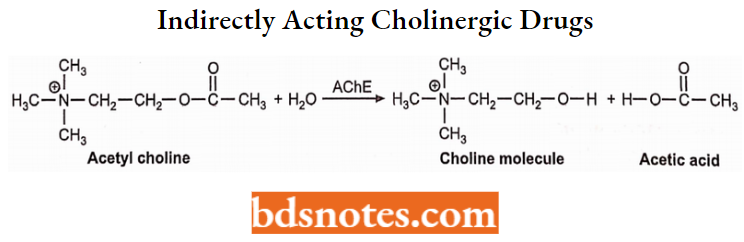
Indirectly Acting Cholinergic Drugs (Acetylcholinesterase Inhibitors)
These drugs inhibit the cholinesterase enzymes and thereby stimulate the production of acetylcholine. It terminates the biological activity of acetylcholine by hydrolyzing acetylcholine into acetic acid and choline molecules.
It is found in RBCs in the brain and other nerve tissue. It is present in high concentration on the presynaptic sites, postsynaptic sites, and at the motor nerve end of the cholinergic neurons. Acetylcholinesterase has three important sites, the anionic, cationic, and esteratic sites.
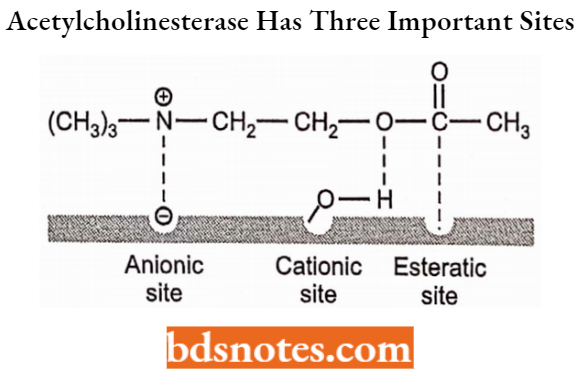
The anionic site of the enzyme binds with the quaternary nitrogen of the ACh through ionic and hydrophobic forces. At this site, the Y-carboxylate group of the glutamic acid residue acts as a strong nucleophile and then attacks the C-carbon of carbonyl moiety to form a tetrahedral intermediate.
- This intermediate collapses to form a choline molecule. The hydroxyl group binds at the cationic site. The esteratic site consists of two imidazole rings from histidine and serine moieties (Imi and Im2).
- The imidazole group of Imi accepts a proton from serine and hydroxyl from the tyrosine site to bind with the ether oxygen of acetylcholine. Choline molecule forms, and undergo dissociation and conformational changes.
In the presence of a second imidazole ring which catalyzes hydrolysis of acetylated serine to give acetic acid and serine residue.
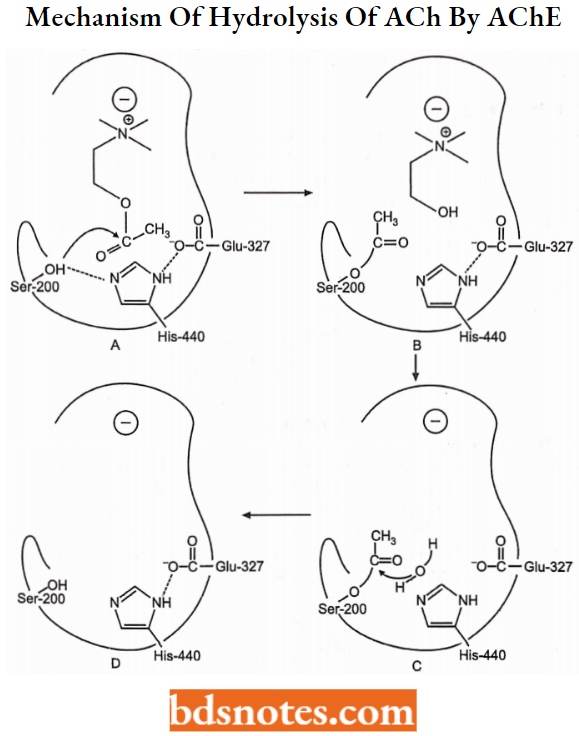
- ACh-AChE-reversible complex,
- Acetylation of a static site,
- General base-catalyzed hydrolysis of the acetylated enzyme,
- Free enzyme
Acetylcholinesterase inhibitors have been used clinically in the treatment of myasthenia gravis. They are also employed to treat glaucoma and more recently Alzheimer’s disease.
Classification Of Anti-Cholinesterases
The anticholinesterases are classified into:
- Reversible anti-cholinesterases.
- Irreversible anti-cholinesterases.
Reversible Anti-cholinesterases
They structurally resemble choline esterases and can compete for the binding sites of cholinesterase receptors. Thus it causes inhibition of the enzyme for a short duration of time.
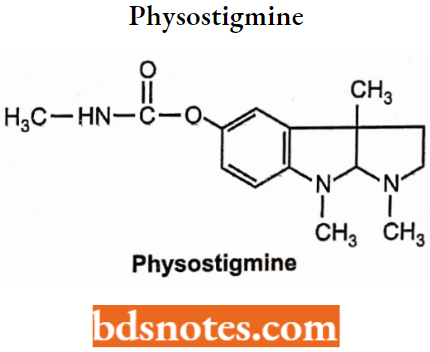
Physostigmine
- Physostigmine, a tertiary amine, is an alkaloid obtained from the physostigma venenosum.
- Chemically, it is 1, 2, 3, 3α, 8, 8α-hexahydro-1, 3α, 8-trimethyl-pyrrolo [2,3-b] indol-5-methylcarbamate. It is a reversible acetylcholinesterase inhibitor.
- Physostigmine reversibly inhibits acetylcholinesterase thus resulting in potentiation of cholinergic activity. It stimulates muscarinic, and nicotinic sites of ANS and nicotinic receptors of neuromuscular junction.
- Used in the treatment of Alzheimer’s disease, and glaucoma and as an antidote during dhatura poisoning.
Neostigmine
- Chemically, it is 3-{[(Dimethylamino)carbonyl]oxy}-N,N,N-trimethylbenzenaminium. Neostigmine is available as bromide and methyl sulfate salt It is a reversible acetylcholinesterases inhibitor and can be administered along with atropine.
- Used in the treatment of Myasthenia gravis, Ogilvie’s syndrome, and cases of snake bites.
- Neostigmine inhibits both AChE and butyrylcholinesterase reversibly and potentiates the nicotinic and muscarinic effects of acetylcholine.
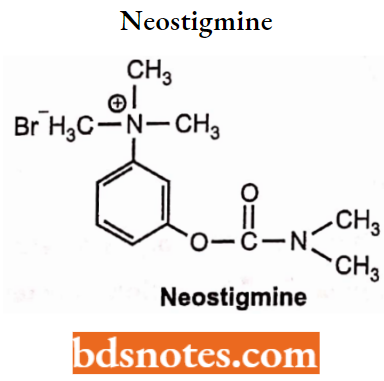
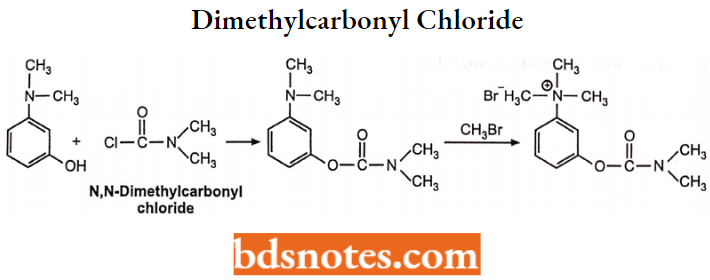
Synthesis: It is synthesized from N, N-Dimethylcarbonyl chloride and 3-Hydroxy N, N-dimethylaniline.
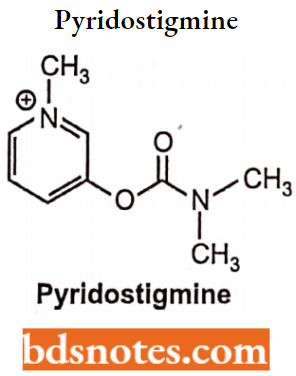
Pyridostigmine
- Pyridostigmine is a pyridine analog of neostigmine.
- Chemically, it is 3-[(dimethylcarbamoyl)oxy]-l-methylpyridinium. It is a reversible acetylcholinesterase inhibitor.
- Pyridostigmine is a potent reversible inhibitor of acetylcholinesterase. By inhibiting the enzyme it permits free transmission of nerve impulses across the neuromuscular junction.
- It is hydrolyzed by acetylcholinesterase but much more slowly than acetylcholine itself.
- Pyridostigmine intensifies both the nicotinic and muscarinic effects of acetylcholine. It can be given and administered by 4.
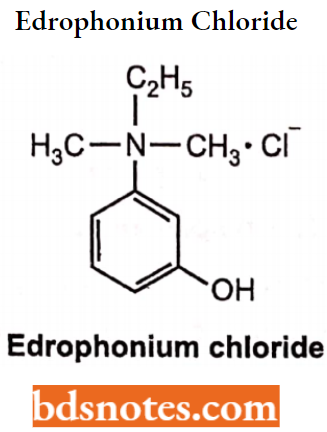
Edrophonium Chloride
- Edrophonium is available as edrophonium chloride. Chemically, it is N-Ethyl-3-hydroxyN, N-dimethylbenzenaminium. It is a reversible acetylcholinesterase inhibitor.
- It acts by inhibiting competitively the enzyme acetylcholinesterase at the neuromuscular junction.
- Quaternary ammonium compounds inhibit the enzyme reversibly by either binding with the esteratic site or the peripheral anionic site.
- Edrophonium chloride is an anti-AChE agent with a rapid onset but short duration of action. Edrophonium binds reversibly and selectively to the active center; this reversible binding and its rapid renal elimination result in its short duration of action. It has been used in myasthenia gravis.
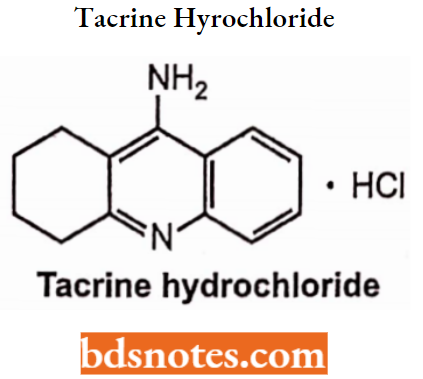
Tacrine Hydrochloride
- Chemically, it is l,2,3,4-tetrahydroacridin-9-amine. It is a reversible inhibitor of acetylcholinesterases.
- It has a longer duration of action.
- Used in the treatment of Alzheimer’s disease and analeptic agents to promote mental awareness.
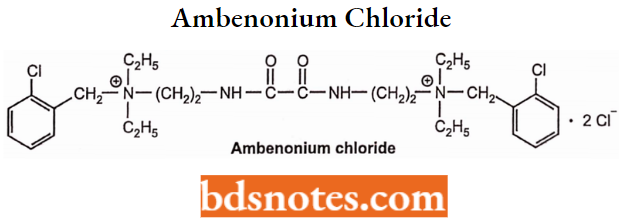
Ambenonium Chloride
- Ambenonium occurs as ambenonium chloride. Chemically, it is [oxalylbis(iminoethylene)] bis[(o-chlorobenzyl)diethylammonium]dichloride.
- It acts by suppressing the activity of acetylcholinesterase by competitive reverse inhibition. It is not hydrolyzed by AChE.
- Used: to treat myasthenia gravis in patients who do not respond satisfactorily to neostigmine or pyridostigmine.
Irreversible Anti-cholinesterases
These compounds are generally organophosphorus and combined with the ecstatic site of cholinesterase and phosphorylated. The process is slow and produces a long-term inhibition of cholinesterases.
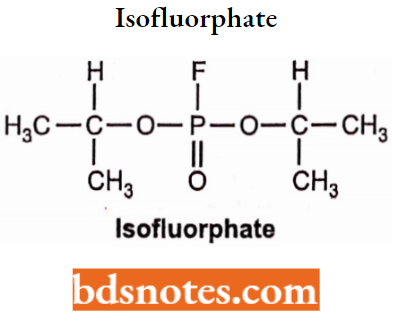
Isofluorphate
- Isofluorphate is an organophosphate compound and chemically it is bis(propane-2-yl) fluorophosphate.
- Isofluorphate irreversibly inhibits cholinesterase, its activity lasts for days or even weeks.
- Used in the treatment of glaucoma.
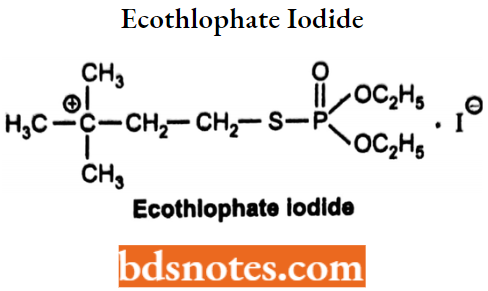
Echothiophate Iodide (Phospholine)
- Echothiophate is an organophosphate available as echothiophate iodide. Chemically, it is 2-(Diethoxyphosphorylsulfanyl)ethyl-N, N, N-trimethylammonium iodide.
- It is A long-acting Irreversible acetylcholinesterase Inhibitor.
- It has a short duration of action with rapid onset and elimination of the drug, Used In the treatment of glaucoma and as an Irreversible cholinesterase,
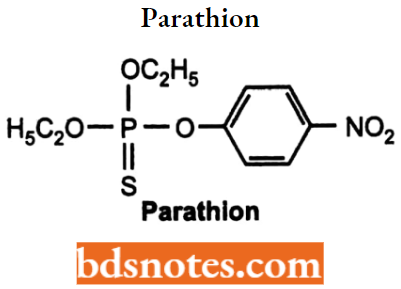
Parathion
- Parathlon is. 0, O-diethyl O-p-nitrophenyl phosphorothloate. It is an
organothlophosphate compound having weak antlchollnostoraso activity. - Used ns agricultural Insecticide and Irreversible cholinesterase.
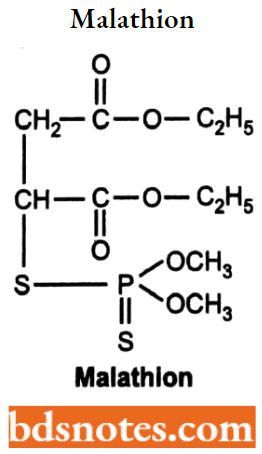
Malathion
- Malathion is a phosphodithioate ester, chemically 2 – [(dimethoxyphosphinothioyl) thio-] – butanedioic acid diethyl ester. It is an organophosphate compound and acts as an acetylcholinesterases inhibitor.
- Malathion is used extensively for controlling insects on vegetables, fruits, and cereal crops. It is also used for controlling insects affecting man and animals (e.g. Mosquitoes.)
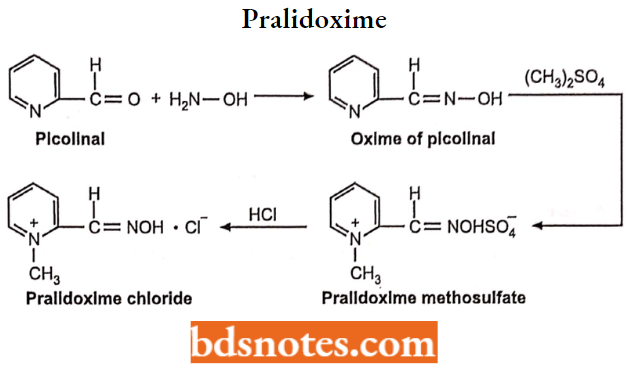
Pralidoxime
- Pralidoxime is an aldoxime and is available as pralidoxime chloride. Chemically it is 2-formyl-l-methylpyridinium chloride oxime.
- Pralidoxime is a useful antidote for intoxication with cholinesterase inhibitors such as organophosphates.
Synthesis: Picolinal is converted into its oxime. The formed oxime is converted to pralidoxime methosulfate with dimethylsulphate, which on treatment with hydrochloric acid forms pralidoxime chloride.
Solanaceous Alkaloids And Analogues
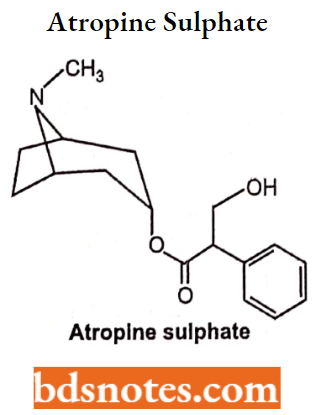
Atropine Sulphate
- Chemically, it is (8-Methyl-8-azabicyclo[3.2.1]oct-3-yl) 3-hydroxy-2-phenylpropanoate.
- It is an antimuscarinic agent and acts by inhibiting the parasympathetic nervous system. It can be administered by 4 or IM.
- Hyoscyamine sulfate: It is a tropane alkaloid. 8-methyl-8-azabicyclo[3.2.1]octan-3-yl 3- hydroxy-2-phenylpropanoate.
- It is an antagonist of the muscarinic acetylcholine receptor. It blocks the action of acetylcholine at parasympathetic sites.
- Used to treat spasms caused by peptic ulcer, irritable bowel syndrome, and pancreatitis. Also to treat Parkinson’s disease and abnormal respiratory symptoms.
Solanaceous Alkaloids And Analogues
Atropine Sulphate
- Chemically, it is (8-Methyl-8-azabicyclo[3.2.1]oct-3-yl) 3-hydroxy-2-phenylpropanoate.
- It is an antimuscarinic agent and acts by inhibiting the parasympathetic nervous system.
- It can be administered by 4 or IM.
- Hyoscyamine sulfate: It is a tropane alkaloid. 8-methyl-8-azabicyclo[3.2.1]octan-3-yl 3- hydroxy-2-phenylpropanoate.
- It is an antagonist of the muscarinic acetylcholine receptor. It blocks the action of acetylcholine at parasympathetic sites.
- Used to treat spasms caused by peptic ulcer, irritable bowel syndrome, and pancreatitis. Also to treat Parkinson’s disease and abnormal respiratory symptoms.
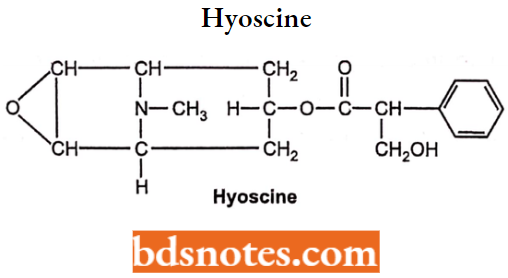
Scopolamine Hydrobromide (Hyoscine)
- Chemically, it is 3-Hydroxy-2-phenylpropionic acid (lR,2R,4S,5S,7a,9S)-9-methyl-3-oxa-9- azatricyclo[3.3.1.02,4]non-7-yl ester.
- It is a natural alkaloid and is considered to be one of the safest drugs.
- Used to treat motion sickness and postoperative nausea- vomiting. It is also given as a pre-anesthetic drug. It has an antispasmodic activity.
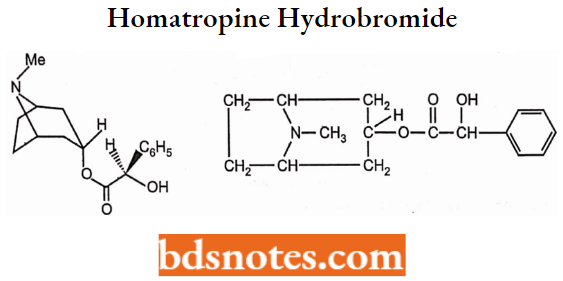
Homatropine Hydrobromide
- Chemically, it is N-Methyl-8-azabicyclo[3.2.1]oct-3-yl) 2-hydroxy-2-phenylacetate.
- It is a muscarinic antagonist receptor.
- It is a synthetic alkaloid. It is available as a colorless, crystalline, and odorless solid. It has a rapid onset of action as compared to atropine and used in mydriasis.
- Used to treat mydriasis. Also to treat peptic ulcers and gastrointestinal spasms.
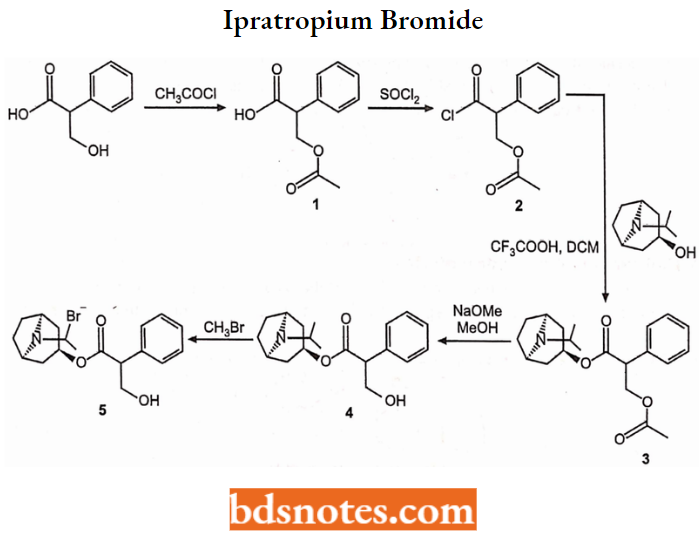
Ipratropium Bromide
- Chemically, it is [8-methyl-8-(l-methylethyl)-8-azoniabicyclo[3.2.1] oct-3-yl] 3-hydroxy-2- phenyl-propanoate.
- It’s a muscarinic antagonist and is used to treat chronic obstructive pulmonary disease (COPD) and asthma COPD.
Synthesis: It is synthesized from acetyl chloride treated with acid chloride to give compound-1 which is subsequently treated with acid chloride to give compound-2.
- Isopropyl alcohol with an organic acid denture base product was dissolved in dichloromethane and added via acylation of compound 2 to give compound 3.
- Compound-3 to give Compound-4 alcoholysis reaction. 4-bromo-methyl compound to give compound-5 i.e. ipratropium bromide.
Synthetic Cholinergic Blocking Agents
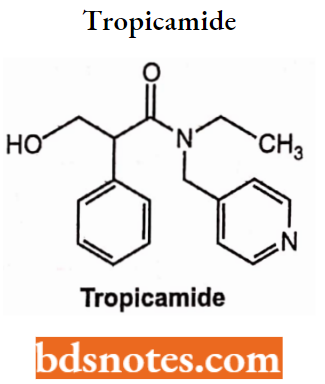
Tropicamide
- Chemically, it is N-ethyl-3-hydroxy-2-phenyl-N-(pyridin-4-ylmethyl)propanamide. It is an anticholinergic drug and produces short-acting mydriasis.
- Used as a mydriatic agent.
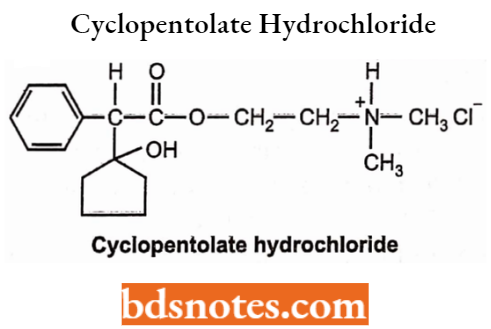
Cyclopentolate Hydrochloride
- Chemically, it is 2-(dimethylamino) ethyl (1-hydroxycyclopentyl) (phenyl) acetate. It is a muscarinic antagonist. It dilates the eye by blocking the muscarinic receptors. It can be used as a substitute for atropine.
- Used as a mydriatic agent. Also used to treat keratitis, choroiditis, and iritis.
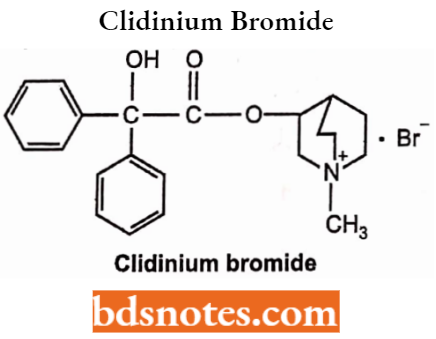
Clidinium Bromide
- Chemically, it is 3-[(2-hydroxy-2,2-diphenylacetyl)oxy]-l-methyl-l-azabicyclo[2.2.2]octan- 1-ium bromide. It is an anticholinergic muscarinic antagonist.
- Used in the treatment of peptic ulcer, hyperchlorhydria, and ulcerative or spastic colon.
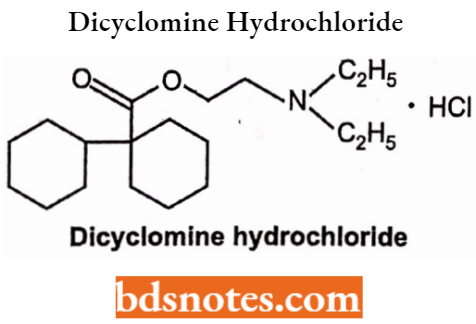
Dicyclomine Hydrochloride
- Chemically, it is 2-(Diethylamino)ethyl 1-cyclohexylcyclohexane-l-carboxylate. It is an anticholinergic, antispasmodic agent, and a smooth muscle relaxant in the GI and urinary tract.
- Used to treat renal, intestinal, irritable bowel, and dysmenorrhoea.
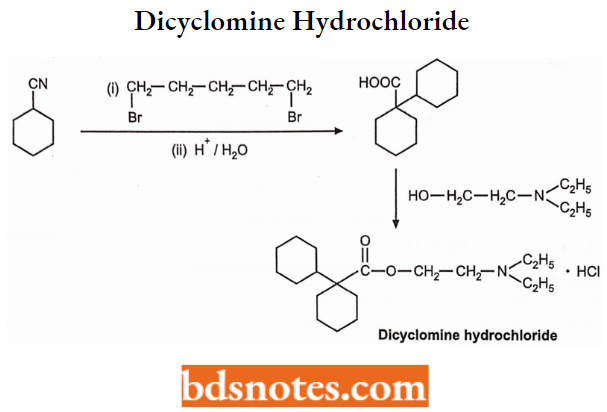
Synthesis: Cyclohexylnitrile on alkylation with 1,5-dibromopentane gives a cyclohexane intermediate which on hydrolysis gives acid. It is then esterified with N, N-diethylamino ethanol to get dicyclomine.
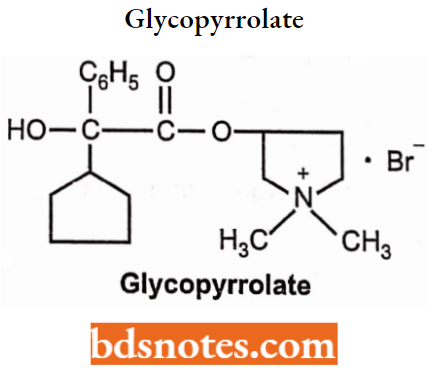
Glycopyrrolate
- Chemically, glycopyrrolate is 3-[2-Cyclopentyl(hydroxy)phenylacetoxy]-l,ldimethylpyrrolidinium bromide.
- It is a muscarinic antagonist. It can be administered orally and by 4 routes.
- Used as anticholinergic, and antispasmodic agent in gastrointestinal spasm.
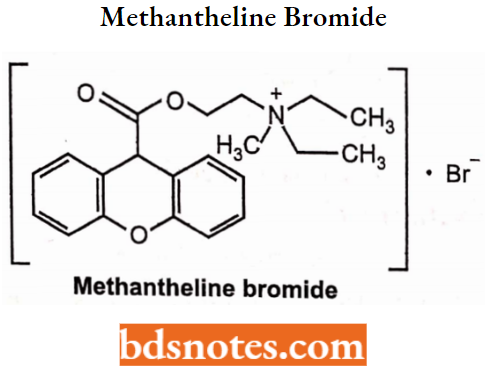
Methantheline Bromide
- Chemically, it is N, N-diethyl-N-methyl-2-[(9H-xanthen-9-ylcarbonyl)oxy]-ethanaminium. It is an anti-muscarinic and acts on the postganglionic cholinergic nerve.
- Used to relieve spasms or cramps of the stomach, intestine, and bladder. Also used to treat peptic ulcers.
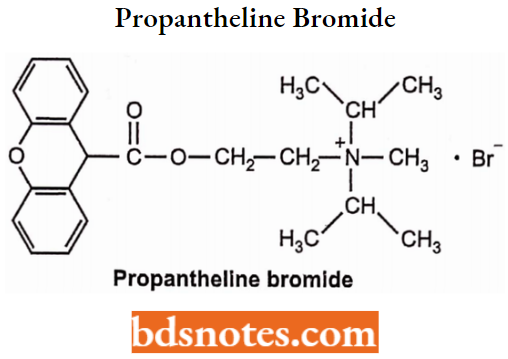
Propantheline Bromide
- Chemically, 3-[2-Cyclopentyl(hydroxy)phenylacetoxy]-l,l-dimethylpyrrolidinium bromide. It is a muscarinic antagonist and has a poor ability to cross the blood-brain barrier.
- Used to reduce excessive saliva. It has a high ratio of ganglionic blocking activity to muscarinic activity. It is used as an antispasmodic and in the treatment of peptic ulcers.
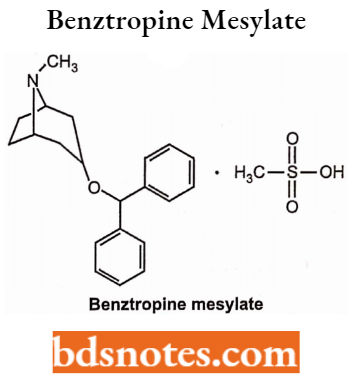
Benztropine Mesylate
- Chemically, it is 3-(Diphenylmethoxy)-8-methyl-8-azabicyclo[3.2.1]octane. It is an anticholinergic drug.
- Used in the treatment of Parkinson’s disease. As an antihistaminic and local anesthetic.
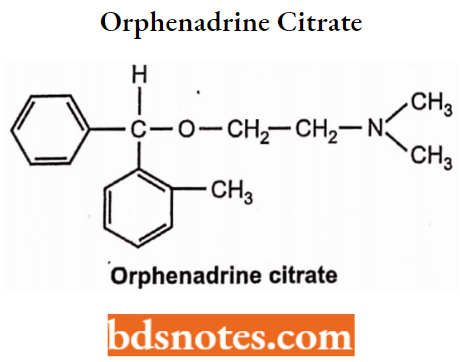
Orphenadrine Citrate
- Chemically, it is N, N-Dimethyl-2-[(2-methylphenyl)-phenyl-methoxy]-ethanamine. It is a cholinergic antagonist. It is a colorless, odorless, crystalline powder.
- Used in Parkinson’s disease and as a skeletal muscle relaxant. It is an antihistaminic agent.
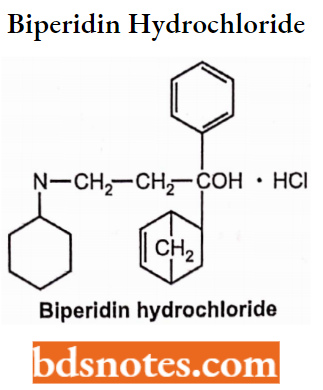
Biperidine Hydrochloride
- Chemically, it is l-(bicyclo[2.2.1]hept-5-en-2-yl)-l-phenyl-3-(piperidin- l-yl)propan-l-ol.
- Used to treat Parkinson’s disease, tremor and rigidity. It is an antipsychotic drug.
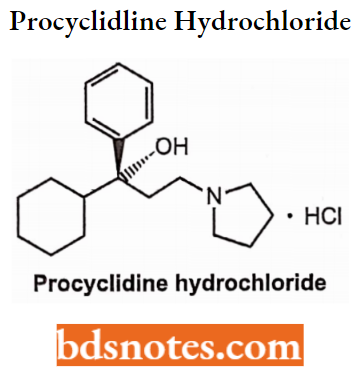
Procyclidine Hydrochloride
- Chemically, it is l-cyclohexyl-l-phenyl-3-pyrrolidin-l-yl-propan-l-ol hydrochloride. It is a cholinergic antagonist and is effective peripherally.
- Used in the treatment of Parkinson’s disease.
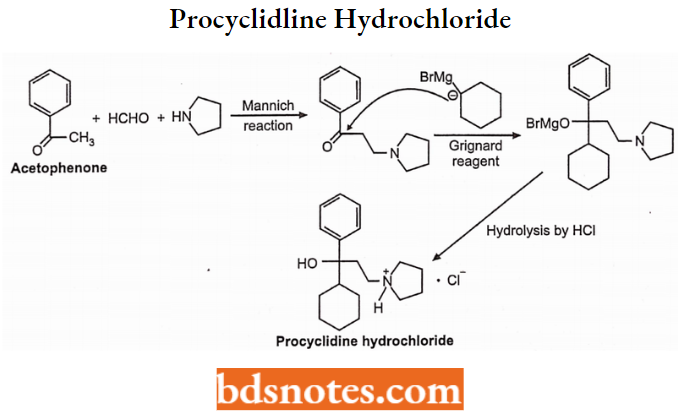
Synthesis: It is synthesized from acetophenone, formaldehyde, and pyrrolidine which is then treated with Grignard reagent to give procyclidine hydrochloride.
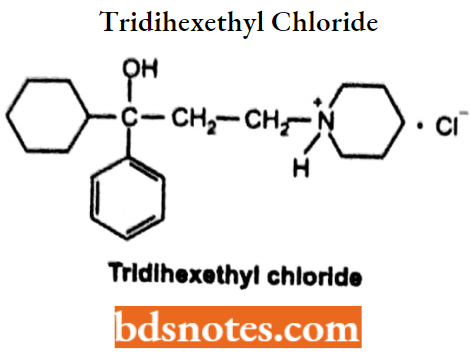
Tridihexethyl Chloride
- Chemically, it is 3-cyclohexyl-N, N, N-triethyl-3-hydroxy-3-phenylpropaml-aminium. It is a muscarinic anticholinergic agent.
- Used in Parkinson’s disease and ulcers. Also used as an antispasmodic.
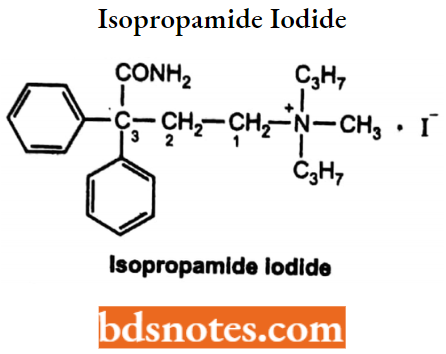
Isopropamide Iodide
- Chemically, it is 4-amino-N, N-diisopropyl-N-methyl-4-oxo-3,3-dlphenylbutan-1-aminium.
- It is an anticholinergic, antispasmodic drug and has a longer duration of action.
- Used to treat peptic ulcer and GI disorders.
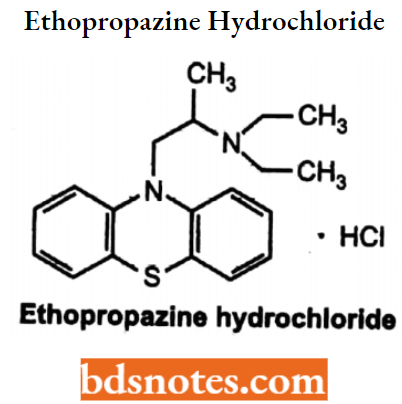
Ethopropnzine Hydrochloride
- Chemically, it is 10-[2-(Diethylamino)propyl]phenothiazine hydrochloride. It is an inhibitor of butyryl cholinesterases.
- Used in the treatment of Parkinson’s disease.
Leave a Reply