Carcinogenesis – Types, Effects and Prevention
“What is carcinogenesis and why does it matter?”
The agents which can induce tumors are called carcinogens.

There are four types of carcinogens :
4 Types Carcinogen
- Molecular pathogens: Genes are involved.
- Chemical carcinogens:
1. Initiator Carcinogens
They can initiate the process of neoplastic transformation.
- DirectActing Carcinogens: They do not require metabolic activation.
- Alkylating Agents: Anticancer drugs, epoxides
- Acylating Agents: Acetylimidazole.
- Indirect Acting Agents: They are also called procarcinogens.
“Understanding the role of carcinogenesis in cancer development”
2. Promoter Carcinogens
- Chemicals that promote further clonal proliferation and expansion of initiated cells. These include:
- Phorbol esters
- Phenols
- Certain hormones: Estrogen, contraceptive hormones
- Miscellaneous: Dietary fat in cancer of the colon.
- Physical carcinogens:
- Radiating: UV light, X-rays, etc. cause cancer by damage to DNA resulting in mutagenesis.
- Non-radiating: By injury, mechanical trauma, and burns induce carcinogenesis.
- Biological carcinogens:
- They are chiefly viruses
- Others are parasites, fungi, and bacteria.
“Importance of studying carcinogenesis for better cancer prevention”

“Common challenges in understanding and preventing carcinogenesis”
Carcinogenesis
- It is also known as oncogenesis or tumorigenesis.
- Carcinogenesis is the mechanism of induction of tumors.
- Carcinogenesis is discussed in four parts:
- Molecular pathogenesis of cancer
- Chemical carcinogenesis
- Physical carcinogenesis
- Biological carcinogenesis.
Molecular Pathogenesis of Cancer
“Steps to identify common types of carcinogenesis”
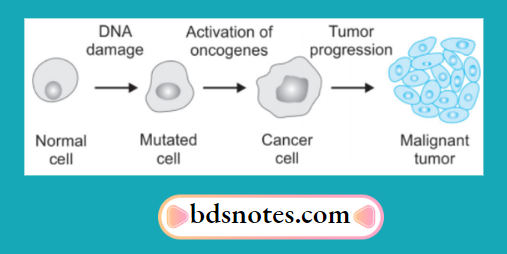
“Role of chemical carcinogens in initiating cancer”
In cancer, transformed cells are produced by abnormal cell growth due to organ damage.
This is due to:
- Activation of growth-promoting oncogenes causes the transformation of the cell. The gene product of oncogenes are called oncoproteins
- Inactivation of cancer suppressor genes permitting the cellular proliferation of transformed cells
- Abnormal apoptosis regulatory genes act as oncogenes or antioncogenes
- Failure of the DNA repair gene and thus there is the inability to repair DNA damage results in a mutation.
“How do viral infections contribute to carcinogenesis?”
Genetic properties or hallmarks of cancer
- Excessive and autonomous growth: Growth promoting oncogenes
- Refractoriness to growth inhibition: Growth suppressing antioncogenes
- Escaping cell death by apoptosis: Genes regulating apoptosis and cancer
- Avoiding cellular aging: Telomeres and telomerase in cancer
- Continued perfusion of cancer: Cancer angiogenesis
- Invasion and distant metastasis: Cancer dissemination
- DNA damage and repair system: Mutator genes and cancer
- Cancer progression and tumor heterogeneity: Clonal aggressiveness.
Chemical Carcinogenesis
The basic mechanism of chemical carcinogenesis is by induction of mutation in proto-oncogenes and anti-oncogenes. It consists of two mechanisms:
- Initiation: It is the first stage in chemical carcinogenesis. Change can be produced by a single dose of initiating agent for a short time, larger doses for a longer duration are more effective. Change is sudden, irreversible, and permanent. Chemical carcinogens acting as initiators are direct-acting carcinogens and indirect-acting carcinogens.
- Metabolic activation: The majority of chemical carcinogens are indirect-acting and require metabolic activation while direct acting does not require this activation. Indirect carcinogens are activated in the liver by monooxygenases of cytochrome P-450 in the endoplasmic reticulum.
- Reactive electrophiles: As direct-acting carcinogens are intrinsically electrophilic, indirect-acting substances become electron deficient after metabolic activation, i.e. they become reactive electrophiles.
- Target molecule: Primary target of electrophiles is DNA which produces mutagenesis.
- Initiated Cell: Unrepaired damage produced in the DNA of the cell become permanent and fied if the altered cell undergoes at least one cycle of proliferation.
- Promotion: Promotors of carcinogenesis are substances such as phorbol esters, phenols, hormones, artificial sweeteners, and drugs like phenobarbital.
- Promotors do not produce sudden change.
- They require application or administration following initiator exposure for sufficient time in a sufficient dose.
- Change induced is reversible.
- They do not damage the DNA and they enhance the effect of initiators.
“Early warning signs of untreated carcinogenic exposure”
Physical Carcinogenesis
Physical agents are divided into two groups, i.e. radiation and non-radiation.
Radiation carcinogenesis
Ultraviolet light and ionizing radiation are two main forms of radiation carcinogenesis.
Mechanism
Radiation damages the DNA of cells by two possible mechanisms, i.e.
- It may directly alter the cellular DNA
- It may dislodge ions from water and other molecules of the cell and result in the formation of highly reactive free radicals which may bring damage to DNA which results in mutagenesis. This will lead to chromosomal breakage, translocation, or point mutation.
Ultraviolet light
Ultraviolet radiation leads to the induction of the mutation, inhibition of cell division, and inactivation of enzymes sometimes causing cell death. UV radiation leads to the formation of pyrimidine dimers in DNA.
“Asymptomatic vs symptomatic stages of carcinogenesis development”
Ionizing Radiation
Ionizing radiation of all kinds of rays like X-rays, α, β, and γ rays, radioactive isotopes, protons, and neutrons leads to carcinogenesis.
The most frequent radiation-induced cancers are leukemias, cancers of the thyroid, skin, breast, salivary glands, ovary, etc.
Non-radiation carcinogenesis
Implants of inert materials such as plastic, glass, etc. in prostheses, foreign bodies, mechanical trauma, and burns lead to carcinogenesis.
Biologic Carcinogenesis
The most important biologic agent which leads to carcinogenesis is the virus. Besides these parasites, fungi, and bacteria are also responsible for biological carcinogenesis.
chemical carcinogenesis
Viral carcinogenesis
Oncogenic transmission of viruses is by three routes:
- Vertical transmission: When infection is generally transmitted from infected parents to off spring.
- Horizontal transmission: When infection passes from one to another by direct contact.
- By inoculation as done in experimental patients.
- Oncogenic viruses fall into two categories, i.e. DNA oncogenic virus and RNA oncogenic virus
Mechanism:
- Mode of DNA viral oncogenesis:
- Host cells infected by DNA viruses undergo replication and integration.
- In replication, viruses replicate in the host cells with consequent lysis of infected cells and release of virions.
- This leads to cell death.
- In integration, viral DNA integrates into host cell DNA.
- This leads to neoplastic transformation.
- Mode of RNA viral oncogenesis:
“Can targeted interventions reduce risks of carcinogenesis?”
Following are the steps involved in the transformation of host cells by RNA oncogenic virus are as follows:
- Reverse transcriptase acts as a template to synthesize a single strand of matching viral DNA.
- A single strand of viral DNA is copied by DNA-dependent DNA synthetase to form another strand of complementary DNA resulting in double-stranded viral DNA or provirus.
- Provirus is then integrated into the DNA of the host cell genome and may transfer the cell into a neoplastic cell.
- Integration of provirus brings about replication of viral components which are then assembled and released by budding.’
Carcinogen Staging and Grading of Cancer
Carcinogen Staging
Staging of cancer is done by TNM staging of tumors which is as follows:
TNM classification was given by American Joint Committee on Carcinoma (AJCC)
- T is suggestive of a primary tumor
- N is suggestive of regional lymph nodes
- M is suggestive of distant metastasis.
“Steps to educate patients about carcinogenesis and its risks”
Carcinogen T: Primary Tumor
- TX: Primary tumor cannot be assessed
- T0: No evidence of primary tumor
- Tis: Carcinoma in situ
- T1: Tumor 2 cm or less in greatest dimension
- T2: Tumor more than 2 cm but not more than 4 cm in greatest dimension
- T3: Tumor more than 4 cm in greatest dimension
- T4a (Lip): Tumor invades through cortical bone, inferior alveolar nerve, floor of mouth or skin (chin or nose)
- T4a (Oral Cavity): Tumor invades through cortical bone, into deep/extrinsic muscle of the tongue (genioglossus, hyoglossus, palatoglossus, and styloglossus), the maxillary sinus or skin of the face
- T4b (lip and oral cavity): Tumor invades masticatory space, pterygoid plates, or skull base or encases internal carotid artery.
Carcinogen N: Regional Lymph Nodes
- NX: Regional lymph nodes cannot be assessed
- N0: No regional lymph node metastasis
- N1: Metastasis in a single ipsilateral lymph node, 3 cm or less in greatest dimension
- N2a: Metastasis in a single ipsilateral lymph node, more than 3 cm but not more than 6 cm in the greatest dimension
- N2b: Metastasis in multiple ipsilateral lymph nodes, not more than 6 cm in greatest dimension
- N2c: Metastasis in bilateral or contralateral lymph nodes, not more than 6 cm in greatest dimension
- N3: Metastasis in a lymph node more than 6 cm in greatest dimension.
Carcinogen M: Distant Metastasis
- MX: Distant metastasis cannot be assessed
- M 0: No distant metastasis
- M 1: Distant metastasis.
Carcinogen Stage Grouping
“Role of counseling in improving awareness of cancer prevention”
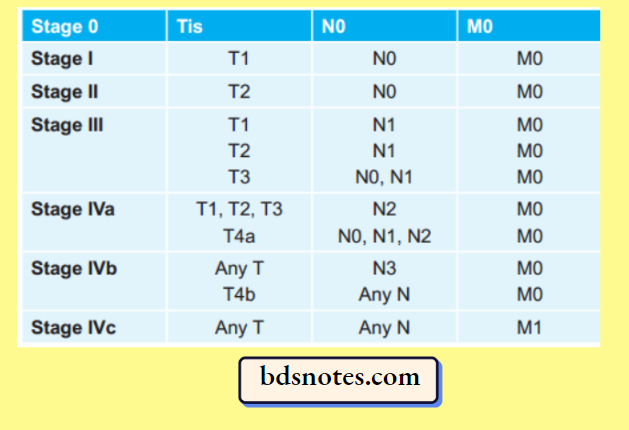
Carcinogen Grading
Grading is defined as the macroscopic and microscopic degree of differentiation of the tumor.
Grading of Cancer is done by Broader’s Grading
- Grade 1: Well differentiated (< 25% anaplastic cell)
- Grade 2: Moderately differentiated (25-50% anaplastic cell)
- Grade 3: Moderately differentiated (50-75% anaplastic cell)
- Grade 4: Poorly differentiated or anaplastic (more than 75% anaplastic cell).
Or - Characteristics of Neoplasia: The characteristics of neoplasia are:
- Rate of growth
- Cancer phenotype and stem cells
- Clinical and gross features
- Microscopic features
- Spread of tumors
- Local invasion or direct spread
- Metastasis or distant spread
“How do educational materials clarify complex carcinogenesis concepts?”
Carcinogen Rate of Growth:
- Tumor cells generally proliferate more rapidly as compared to normal cells. Benign tumors grow slowly and malignant tumors rapidly. But there are exceptions to this generalization. The rate at which the tumor enlarges depends upon two main factors:
- Rate of cell production, growth fraction, and rate of cell loss
- Degree of differentiation of the tumor.
Rate of Cell Production, Growth Fraction and Rate of Cell Loss
The rate of growth of a tumor depends upon 3 important parameters:
- Doubling time of tumor cells,
- Number of cells remaining in the proliferative pool (growth fraction)
- Rate of loss of tumor cells by cell shedding.
“Early warning signs of knowledge gaps in patient understanding”
In general, malignant tumor cells have increased mitotic rates and slower death rates, i.e. cancer cells do not follow normal controls in the cell cycle and are immortal. If the rate of cell division is high, the tumor cells in the center of the tumor do not receive adequate nourishment and undergo ischemic necrosis.
At a stage when malignant tumors grow relentlessly, they do so because a larger proportion of tumor cells remain in the replicative pool but due to lack of availability of adequate nourishment, these tumor cells are either lost by shedding or leave the cell cycle to enter into G0 (resting phase) or G1 phase.
Degree of Differentiation
The rate of growth of malignant tumors is directly proportional to the degree of differentiation. Poorly differentiated tumors show m aggressive growth patterns as compared to better-differentiated tumors.
Some tumors, after a period of slow growth, may suddenly show an increase in their growth due to the development of an aggressive clone of malignant cells.
On the other hand, some tumors may cease to grow after some time. The regulation of tumor growth is under the control of growth factors secreted by the tumor cells.
Cancer Phenotype and Stem Cells
Normally growing cells in an organ grow under normal growth controls, perform their assigned function and there is a balance between the rate of cell proliferation and the rate of cell death including cell suicide, i.e. apoptosis.
So, normal cells are socially desirable. However, cancer cells exhibit the following antisocial behavior:
- Cancer cells do not follow growth-controlling signals in the body and thus proliferate rapidly.
- Cancer cells escape death signals and achieve immortality.
- The imbalance between cell proliferation and cell death in cancer causes excessive growth.
- Cancer cells lose properties of differentiation and thus perform little or no junction.
- Due to the loss of growth controls, cancer cells are genetically unstable and develop newer mutations.
- Cancer cells over-run their neighboring tissue and invade locally.
- Cancer cells have the ability to travel from the site of origin to other sites in the body where they colonize and establish distant metastasis.
“Can interactive tools improve adherence to carcinogenesis prevention?”
Cancer cells originate by clonal proliferation of a single progeny of a cell. Cancer cells arise from stem cells normally present in the tissues in small numbers and are not readily identifiable.
These stem cells have the properties of prolonged self-renewal, symmetric replication, and transdifferentiation. These cancer stem cells are called tumor-initiating cells.
Clinical and Gross Features
Clinically, benign tumors are generally slow growing, and de- pending upon the location, may remain asymptomatic or may produce serious symptoms but on the other hand, malignant tumors grow rapidly, may ulcerate on the surface, and invade locally into deeper tissues.
May spread to distant sites (metastasis), and also produce systemic features such as weight loss, anorexia, and anemia. There are three cardinal clinical features of malignant tumors, i.e. anaplasia, invasiveness, and metastasis.
The gross appearance of benign and malignant tumors may be quite variable and the features may not be diagnostic on the basis of gross appearance alone.
However, certain distinctive features characterize almost all tumors compared to neighboring normal tissue of origin—they have a different color, texture, and consistency.
Gross terms such as papillary, fungating, infiltrating, hemorrhagic, ulcerative, and cystic are used to describe the macroscopic appearance of the tumors.
Following are the general gross features of benign and malignant tumors:
- Benign tumors are generally spherical or ovoid in shape. They are encapsulated or well-circumscribed, freely movable, more often fim and uniform, unless secondary changes like hemorrhage or infarction supervene.
- Malignant tumors, on the other hand, are usually irregular in shape, poorly circumscribed, and extend into the adjacent tissues. Secondary changes like hemorrhage, infarction, and ulceration are seen more often. Sarcomas typically have fish-flesh-like consistency while carcinomas are generally firm.
Microscopic Features
These features appreciated in histologic sections are as under:
- Microscopic pattern
- Histomorphology of neoplastic cells (differentiation and anaplasia)
- Tumor angiogenesis and stroma
- Inflammatory reaction.
1. Microscopic Pattern
The patterns or arrangements of tumor cells are best appreciated under low-power microscopic examination of the tissue section.
Some of the common patterns in tumors are:
- Epithelial tumors generally consist of acini, sheets, columns, or cords of epithelial tumor cells that may be arranged in a solid or papillary pattern.
- Mesenchymal tumors have mesenchymal tumor cells arranged as interlacing bundles, fascicles, or whorls lying separated from each other usually by the intercellular matrix substance such as hyaline material in leiomyoma, cartilaginous matrix in chondroma, osteoid in osteosarcoma, reticulin network in soft tissue sarcomas, etc.
- Certain tumors have mixed patterns For Example. teratoma arising from totipotent cells, pleomorphic adenoma of the salivary gland (mixed salivary tumor), and fibroadenoma of the breast.
- Hematopoietic tumors such as leukemias and lymphomas often have no or little stromal support.
2. Cytomorphology of Neoplastic Cells: (Differentiation and Anaplasia)
- Differentiation is defined as the extent of the morphological and functional resemblance of parenchymal tumor cells to corresponding normal cells.
- If the deviation of the neoplastic cells in structure and function is minimal as compared to a normal cell, the tumor is described as ’well differentiated such as most benign and low-grade malignant tumors.
Poorly differentiated, undifferentiated, or dedifferentiated are synonymous terms for poor structural and functional resemblance to corresponding normal cells. - Anaplasia is a lack of differentiation and is a characteristic feature of most malignant tumors. Depending upon the degree of differentiation, the extent of anaplasia is also variable i.e. poorly differentiated malignant tumors have a high degree of anaplasia. Due to anaplasia, noticeable morphological and functional alterations in the neoplastic cells are observed which are best appreciated in higher magnification of the microscope.
These features are as follows:
- Loss of polarity: Normally, the nuclei of epithelial cells are oriented along the basement membrane which is termed basal polarity. Early in malignancy, tumor cells lose their basal polarity so that the nuclei tend to lie away from the basement membrane.
- Pleomorphism: Pleomorphism means variation in the size and shape of the tumor cells. The extent of cellular pleomorphism generally correlates with the degree of anaplasia. Tumor cells are often bigger than normal but in some tumors, they can be of normal size or smaller than normal.
- Nuclear: Cytoplasmic ratio: Generally, the nuclei of malignant tumor cells show more conspicuous changes. Nuclei are enlarged disproportionately to the cell size so that the nuclear-cytoplasmic ratio is increased from normal 1:5 to 1:1.
- Anisonucleosis just like cellular pleomorphism, the nuclei too, show variation in size and shape in malignant tumor cells.
- Hyperchromatism: Characteristically, the nuclear chromatin of malignant cells is increased and coarsely clumped. This is due to an increase in the amount of nucleoprotein resulting in dark staining nuclei, referred to as hyperchromatism.
- Nucleolar changes: Malignant cells frequently have a prominent nucleolus or nucleoli in the nucleus reflecting increased nucleoprotein synthesis. This may be demonstrated as Nucleolar Organiser Region (NOR) by silver staining called AgNOR material.
- Mitotic figures: The parenchymal cells of poorly differentiated tumors often show a large number of mitoses as compared with benign tumors and well-differentiated malignant tumors.
- Functional (Cytoplasmic) changes: Structural anaplasia in tumors is accompanied by functional anaplasia as appreciated by the cytoplasmic constituents of the tumor cells. The functional abnormality in neoplasms may be quantitative, qualitative, or both.
- Chromosomal abnormalities: All tumor cells have abnormal genetic composition and on division, they transmit the genetic abnormality to their progeny. The chromosomal abnormalities are more marked in more malignant tumors which include deviations in both morphology and number of chromosomes.
3. Tumor Angiogenesis and Stroma
The connective tissue along with its vascular network forms the supportive framework on which the parenchymal tumor cells grow and receive nourishment.
In addition to a variable amount of connective tissue and vascularity, the stroma may have nerves and metaplastic bone or cartilage but no lymphatics.
Tumor Angiogenesis:
In order to provide nourishment to growing tumors, new blood vessels are formed from pre-existing ones (angiogenesis). Its related morphologic features are as under:
- Microvascular density: The new capillaries add to the vascular density of the tumor which has been used as a marker to assess the rate of growth of tumors and hence grade the tumors. This is done by counting the density in the section of the tumor.
- Central necrosis: If the tumor outgrows its blood supply as occurs in rapidly growing tumors or tumor angiogenesis fails, its core undergoes ischemic necrosis.
Tumor Stroma
The collagenous tissue in the stroma may be scanty or excessive. If the stroma may be scanty or excessive. If the stroma is soft and fleshy (For Example. in sarcomas, lymphomas), while if it is excessive the tumor is hard and gritt (for example, Infiltrating duct carcinoma breast).
“Asymptomatic vs symptomatic effects of poor communication”
The growth of fibrous tissue in tumors is stimulated by basic fibroblast growth factors elaborated by tumor cells.
If the epithelial tumor is almost entirely composed of parenchymal cells, it is called a medullary For Example. medullary carcinoma of the breast, medullary carcinoma of the thyroid.
If there is excessive connective tissue stroma in the epithelial tumor, it is referred to as desmoplasia and the tumor is hard or scirrhous, For Example. infiltrating duct carcinoma breast.
4. Inflammatory Reaction
At times, a prominent inflammatory reaction is present in and around the tumors. It could be the result of ulceration in the cancer when there is a secondary infection. The inflammatory reaction in such instances may be acute or chronic.
However, some tumors show chronic inflammatory reactions, chiefly of lymphocytes, plasma cells, and macrophages, and in some instances granulomatous reaction, as a part of the morphologic features of the tumor, in the absence of ulceration.
Spread of Tumors:
One of the cardinal features of malignant tumors is their ability to invade and destroy adjoining tissues (local invasion or direct spread) and disseminate to distant sites (metastasis or distant spread).
Local Invasion (Direct Spread):
- Benign tumors: Most benign tumors form encapsulated or circumscribed masses that expand and push aside the surrounding normal tissues without actually invading, infiltrating, or metastasizing.
- Malignant tumors: Malignant tumors also enlarge by expansion and some well-differentiated tumors may be partially encapsulated as well, for example, Follicular carcinoma thyroid. But characteristically, they are distinguished from benign tumors by invasion, infiltration, and destruction of the surrounding tissue, besides spreading to distant sites or metastasis.
In general, tumors invade via the route of least resistance, though eventually, most cancers recognize no anatomic boundaries. Often, cancers extend through tissue spaces, permeate lymphatics, blood vessels, and perineural spaces, and may penetrate a bone by growing through nutrient foramina.
Metastasis (Distant Spread):
Metastasis is defined as the spread of a tumor by invasion in such a way that a discontinuous secondary tumor mass forms at the site of lodgment. Cancers may spread to distant sites by the following pathways:
- Lymphatic spread
- Hematogenous spread
- Spread along body cavities and natural passages.
Lymphatic spread: In general carcinomas metastasize by lymphatic route. Virchow’s lymph node is nodal metastasis to the supraclavicular lymph node from cancers of the abdominal organ.
Hematogenous spread: It is a common root for sarcomas but certain carcinomas are also spread by this mode. The common sites are the lung, breast, thyroid, kidney, liver, prostate, and ovary.
Spread along body cavities and natural passages: The routes of distant spread are trans coelom, epithelial lined surfaces, CSF, and implantation.
Leave a Reply