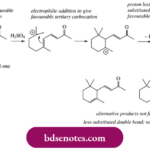
Electrophilic Reactions In the preceding chapters we have seen how new bonds may be formed between nucleophilic reagents and various substrates that have electrophilic centres, the latter typically arising as a result of uneven electron distribution in the molecule. The nucleophile was considered to be the reactive species. In this chapter, we shall consider reactions in … [Read more...] about Electrophilic Reactions
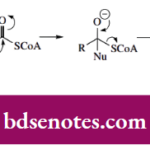
Nucleophilic Reactions Of Carbonyl Groups
Nucleophilic Reactions Of Carbonyl Groups Nucleophilic Reactions Of Carbonyl Groups: Aldehydes And Ketones The carbon-oxygen double bond C=O is termed a carbonyl group and represents one of the most important reactive functional groups in chemistry and biochemistry. Since oxygen is more electronegative than carbon, the electrons in the double bond are not shared equally and … [Read more...] about Nucleophilic Reactions Of Carbonyl Groups
Radical Reactions
Radical Reactions Formation Of Radicals The ionization of HBr distributes the two electrons of the single H–Br bond so that the electronegative bromine accepts electrons while hydrogen loses electrons, and the resultant ions are thus H+ and Br−. This process is termed heterolytic cleavage, in that the two atoms of the bond suffer different fates and that the two electrons … [Read more...] about Radical Reactions
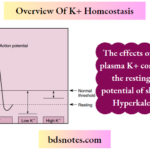
Regulation Of Potassium Balance
Regulation Of Potassium Balance "What is the regulation of potassium balance?" Potassium, which is one of the most abundant cations in the body, is critical for many cell functions, including cell volume regulation, intracellular pH regulation, DNA and protein synthesis, growth, enzyme function, resting membrane potential, and cardiac and neuromuscular activity. Despite … [Read more...] about Regulation Of Potassium Balance
Acid Base Balance and Progression of Kidney Disease
Regulation Of Acid-Base Balance "What is acid-base balance in kidney disease? A detailed question and answers guide" The Concentration Of H+ In The Body Fluids Is Low Compared With That Of Other Ions. For example, Na+ is present at a concentration of some 3 million times greater than that of H+ ([Na+] = 140 Meq/L And [H+] = 40 Neq/L). Because Of The Low [H+] Of The Body … [Read more...] about Acid Base Balance and Progression of Kidney Disease