Antianginal Drugs Drugs Acting On the Cardiovascular System
The drugs acting on the cardiovascular system are divided into four groups:
- Cardiotonic drugs.
- Anti-hypertensive drugs.
- Anti-arrhythmic drugs
- Anti-anginal drugs.
Antianginal Drugs
These are the drugs used in the treatment of angina pectoris. Angina pectoris, commonly known as angina, is severe chest pain due to ischemia (a lack of blood.
- Hence a lack of oxygen supply) of the heart muscle, generally due to obstruction or spasm of the coronary arteries. Coronary artery disease, the main cause of angina, is due to atherosclerosis of the cardiac arteries.
- Major risk factors for angina include cigarette smoking, diabetes, high cholesterol, high blood pressure, a sedentary lifestyle, and a family history of premature heart disease.
“Understanding antianginal drug classification through FAQs: Q&A explained”
Read and Learn More Medicinal Chemistry II Notes
Types of angina pectoris
There are three types:
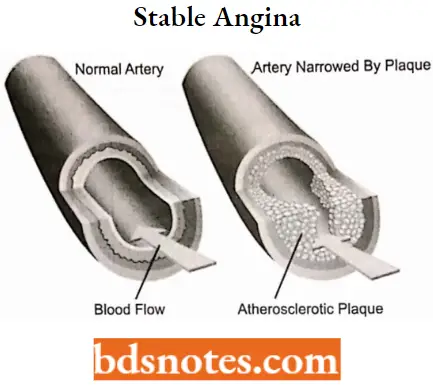
1. Stable angina: In this type the atherosclerotic plaque and inappropriate vasoconstriction (caused by endothelial damage) reduce the blood vessel lumen diameter. Hence there is a reduction in blood flow.
“Importance of studying antianginal drugs for pharmacology students: Questions explained”
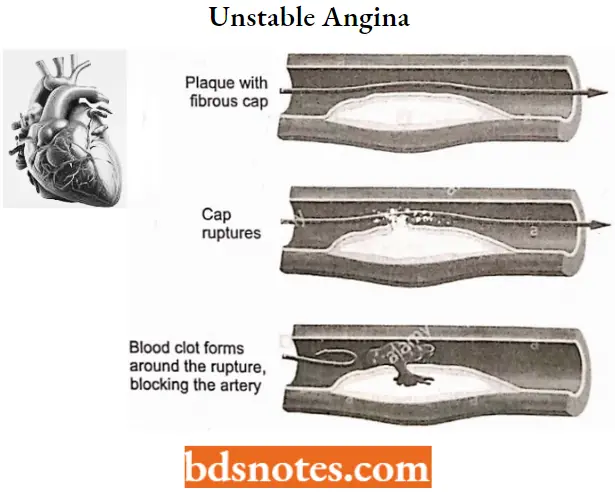
2. Unstable angina: In unstable angina, the rapture of the plaque triggers platelet aggregation, thrombus formation, and vasoconstriction. Depending upon plaque rapture this leads to non-Q wave (non-ST elevation) or Q wave (ST elevation).
“Common challenges in understanding antianginal mechanisms effectively: FAQs provided”
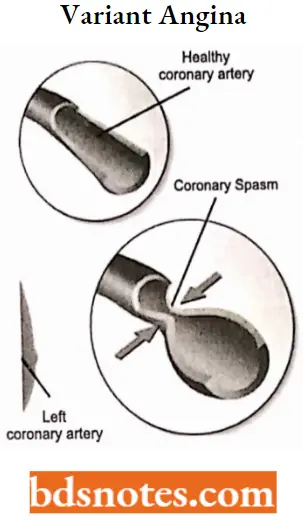
3. Variant angina: In this type atherosclerotic plaques are absent, and ischemia is caused by intense vasospasm. It occurs more in younger women.
Antianginal Drugs Classification
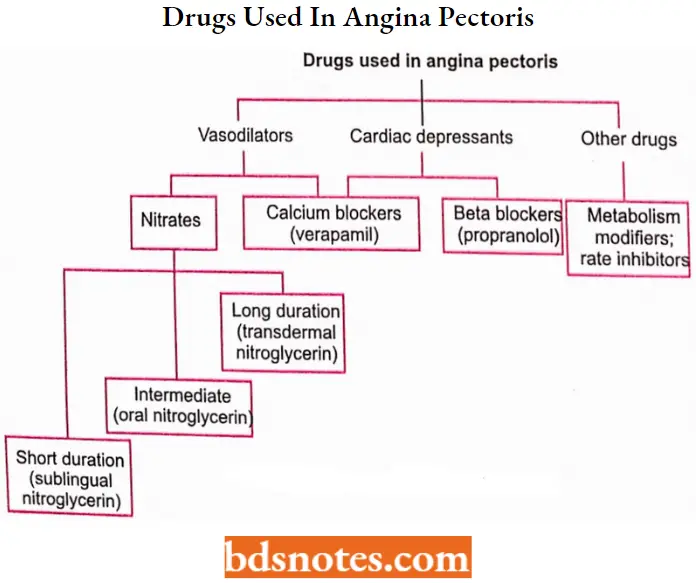
Therapy of angina is directed mainly toward alleviating and preventing anginal attacks by altering the oxygen supply or oxygen demand ratio to the cardiac muscle or dilating the coronary vessels.
Three classes of drugs are found to be very efficient in this regard, although via different mechanisms. These include organic nitrates, calcium channel blockers, and b-adrenergic blockers.
“Factors influencing success with antianginal drug knowledge: Q&A”
Vasodilators
Nitrates
A nitro vasodilator is a pharmaceutical agent that causes vasodilation (widening of blood vessels) by donation of nitric oxide (NO), and is mostly used for the treatment and prevention of angina pectoris.
Nitrates MOA: Within the body, organic nitrates are chemically reduced to release NO. NO is an endogenous signaling molecule that causes vascular smooth muscle relaxation. The various organic nitrates give rise to NO by different chemical and biochemical mechanisms.
- Organic nitrates have the chemical structure RNO2. The nitro group is reduced to form NO in the presence of specific enzymes and extracellular and intracellular reductants (For Example. thiols).
- The NO activates guanylyl cyclase. The activated guanylyl cyclase increases the formation of cGMP from GTP. The cGMP activates myosin-LC phosphatase.
- The activated myosin-LC phosphatase causes the dephosphorylation of myosin-LC-(P) to myosin-LC. This relaxes the smooth muscle and causes vasodilatation.
Nitrates SAR:
- In a cell-free system, the potency of organic nitrate for guanylate cyclase activation is mainly determined by the number of nitrate groups.
- Since nitrate-induced activation of guanylate cyclase involves the formation of the nitric oxide free radicals, potency, therefore, increases as the nitric group increases in the group.
- An increase in lipophilicity due to esterification of the free OH group in isosorbide mononitrate had no major influence on guanylate cyclase activation.
Amyl nitrite
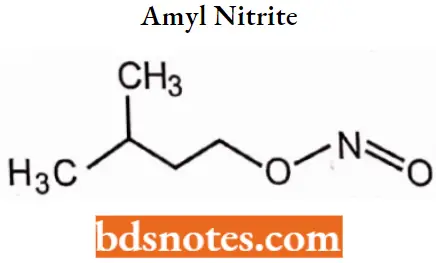
Amyl nitrite IUPAC Name: (3-methylbutyl) nitrite.
MOA: Amyl nitrite’s antianginal action is thought to be the result of a reduction in systemic and pulmonary arterial pressure (afterload) and decreased cardiac output because of peripheral vasodilation, rather than coronary artery dilation.
Amyl nitrite is a source of nitric acid, which accounts for the mechanism described above. As an antidote (to cyanide poisoning), amyl nitrite promotes the formation of methemoglobin, which combines with cyanide to form nontoxic cyanmethemoglobin.
Amyl nitrite SAR: The small lipophilic ester character makes them volatile. Volatility is an important concern in drug formulation because of the potential loss of the active principle from the dosage form.
Amyl nitrite Metabolism: The drug is metabolized rapidly, probably by hydrolytic denitration; approximately one-third of the inhaled amyl nitrite is excreted in the urine.
Amyl nitrite Therapeutic uses: Amyl nitrite is employed medically to treat heart diseases as well as angina. Amyl nitrite is sometimes used as an antidote for cyanide poisoning. It can act as an oxidant, to induce the formation of methemoglobin.
Amyl nitrite Adverse reactions: Headache, giddiness, postural hypotension, flushing, dizziness (cloudness of consciousness). Tolerance develops with continuous use. Tolerance is more common with oral and transdermal routes.
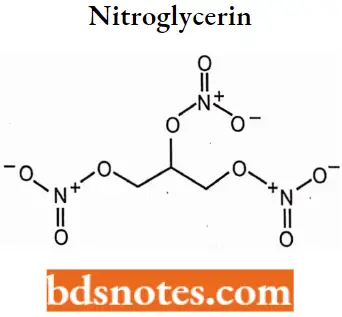
Nitroglycerin
“Steps to explain types of antianginal drugs: Nitrates vs beta-blockers vs calcium channel blockers: Q&A guide”
Nitroglycerin IUPAC Name: Propane-1,2,3-triyl trinitrate.
Nitroglycerin MOA: Similar to other nitrites and organic nitrates, nitroglycerin is converted to nitric oxide (NO), an active intermediate compound that activates the enzyme guanylate cyclase.
- This stimulates the synthesis of cyclic guanosine 3’,5-monophosphate (cGMP) which then activates a series of protein kinase-dependent phosphorylations in the smooth muscle cells.
- Eventually resulting in the dephosphorylation of the myosin light chain of the smooth muscle fiber. The subsequent release of calcium ions results in the relaxation of the smooth muscle cells and vasodilation.
Nitroglycerin Metabolism: Hepatic, cytochrome P450 (P450) is a key enzyme of organic nitrate biotransformation.
Nitroglycerin Therapeutic uses: Nitroglycerin extended-release capsules are used to prevent chest pain (angina) in people with a certain heart condition (coronary artery disease).
- This medication belongs to a class of drugs known as nitrates. Angina occurs when the heart muscle is not getting enough blood.
- This drug works by relaxing and widening blood vessels so blood can flow more easily to the heart.
Nitroglycerin Adverse reactions: Headache, dizziness, lightheadedness, nausea, and flushing may occur as your body adjusts to this medication. If any of these effects persist or worsen, tell your doctor or pharmacist promptly.
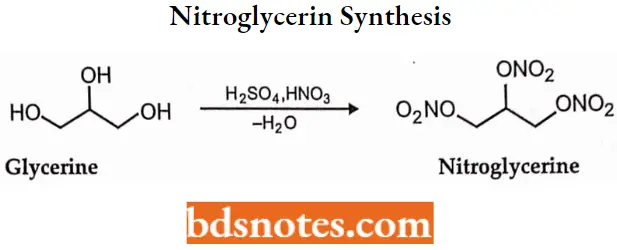
Nitroglycerin Synthesis:
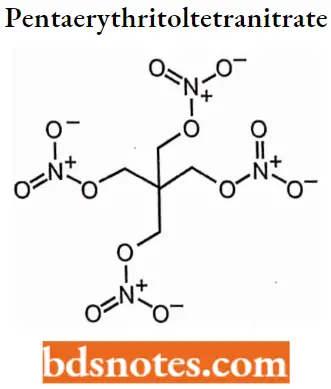
Pentacrythritoltetranitrate
“Role of nitrates in relieving angina symptoms: Questions answered”
Pentacrythritoltetranitrate IUPAC Name: 2,2-Bis[(nitrooxy)methyl]propane-l,3-diyl dinitrate.
Pentacrythritoltetranitrate MOA: Pentaerythritoltetranitrate is the lipid-soluble polyol ester of nitric acid belonging to the family of nitro-vasodilators.
- Pentaerythritoltetranitrate releases free nitric oxide (NO) after the denitration reaction, which triggers NO-dependent signaling transduction involving soluble guanylate cyclase (sGC).
- Nitric oxide binds reversibly to the ferrous-heme center of sGC, causing a conformational change and activating the enzyme.
- This enzyme activation results in increased cellular concentrations of cyclic guanosine monophosphate JcGMP) within the vascular smooth muscle, resulting in vasodilation mediated by cGMP-dependent protein kinases.
Pentacrythritoltetranitrate Metabolism: Extensively metabolized in the liver.
Pentacrythritoltetranitrate Metabolites: pentaerythritol trinitrate, pentaerythritol dinitrate, pentaerythritol (inactive).
Pentacrythritoltetranitrate Therapeutic Uses: Like nitroglycerin (glyceryl trinitrate) and other nitrates, PETN is also used medically as a vasodilator in the treatment of heart conditions.
These drugs work by releasing the signaling gas nitric oxide in the body. The heart medicine Lentonitrat is nearly pure PETN.
Pentacrythritoltetranitrate Adverse reactions: The toxicity of nitrate drugs is the result of nitrate conversion to nitrite in the body. Nitrite leads to the autocatalytic oxidation of oxyhemoglobin to both hydrogen peroxide and methemoglobin.
- This increase in methemoglobin levels is a condition known as methemoglobinemia and is manifested by tissue hypoxia, as methemoglobin is unable to bind oxygen.
- Dizziness, redness of the skin (due to vasodilatation), skin irritation, and headache are adverse effects of this drug. Postural hypotension can also occur, especially if this drug is taken at higher doses or while standing.
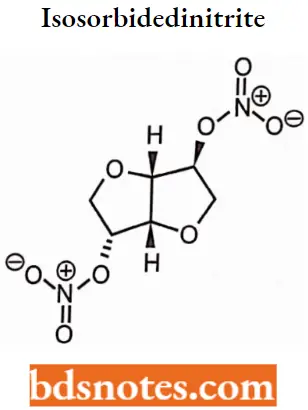
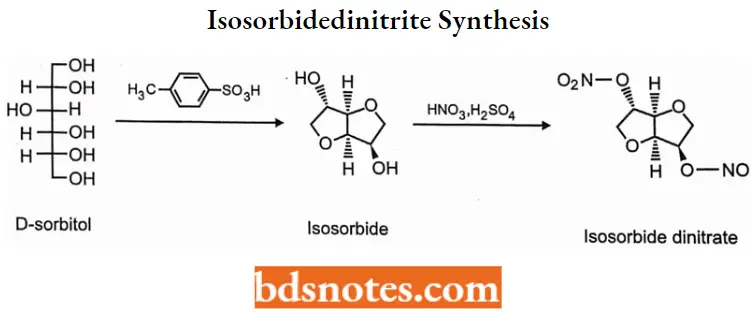
Isosorbidedinitrite
“How do beta-blockers reduce myocardial oxygen demand? FAQ explained”
Isosorbidedinitrite IUPAC Name: l,4:3,6-dianhydro-2,5-di-0-nitro-D-glucitol.
Isosorbidedinitrite MOA: It relaxes the vascular smooth muscle and consequent dilatation of peripheral arteries and veins, especially the latter.
- Dilatation of the veins promotes peripheral pooling of blood and decreases venous return to the heart, thereby reducing left ventricular end-diastolic pressure and pulmonary capillary wedge pressure (preload).
- Arteriolar relaxation reduces systemic vascular resistance, systolic arterial pressure, and arterial pressure.
Isosorbidedinitrite Metabolism: Absorption of isosorbide dinitrate after oral dosing is nearly complete, but bioavailability is highly variable (10% to 90%), with extensive first-pass metabolism in the liver.
Isosorbidedinitrite Therapeutic Uses: Isosorbidedinitrate (ISDN) is a medication used for heart failure and esophageal spasms, and to treat and prevent chest pain from not enough blood flow to the heart.
It is particularly useful in heart failure due to systolic dysfunction together with hydralazine in black people.
Isosorbidedinitrite Adverse Reactions: Common side effects include headache, lightheadedness with standing, and blurred vision. Severe side effects include low blood pressure.
It is unclear if use in pregnancy is safe for the baby. Severe allergic reactions (rash; hives; itching; difficulty breathing; tightness in the chest; swelling of the mouth, face, lips, or tongue); fainting; fast or slow heartbeat; nausea; new or worsening chest pain; vomiting.
Isosorbidedinitrite Synthesis:
Dipyridamole
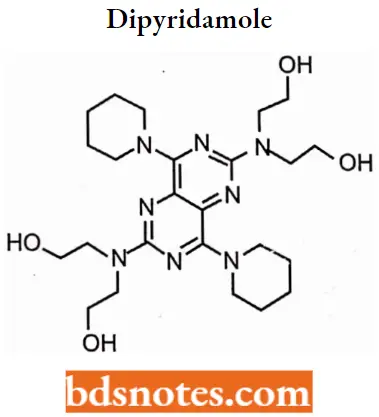
Dipyridamole IUPAC Name: 2,2′,2″,2″,-(4,8-di(piperidin-l-yl)pyrimido[5,4-d]pyrimidine-2,6-diyl) bis (azanetriyl) tetraethanol.
Dipyridamole MOA: Dipyridamole has Iwo known effects, acting via different mechanisms of action:
- Dipyridamole inhibits the phosphodiesterase enzymes that normally break down cAMP (increasing cellular cAMP levels and blocking the platelet aggregation response to ADP) and or cGMP.
- Dipyridamole inhibits the cellular reuptake of adenosine into platelets, red blood cells, and endothelial cells, leading to increased extracellular concentrations of adenosine.
Dipyridamole Metabolism: Metabolised in the liver mainly excreted as glucoronoids in bile.
Dipyridamole Therapeutic Uses: Dipyridamole likely inhibits both adenosine deaminase and phosphodiesterase, preventing the degradation of cAMP, an inhibitor of platelet function.
- This elevation in cAMP blocks the release of arachidonic acid from membrane phospholipids and reduces thromboxane A2 activity.
- Dipyridamole also directly stimulates the release of prostacyclin, which induces adenylate cyclase activity, thereby raising the intraplate-let concentration of cAMP and further inhibiting platelet aggregation.
Dipyridamole Adverse reactions: Dizziness, stomach upset, diarrhea, vomiting, headache, and flushing may occur as your body adjusts to the medication.
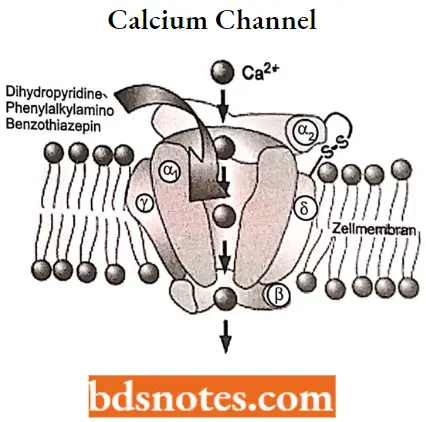
Calcium channel blockers
- Calcium-channel blockers (CCBs for short) are medicines that are often used to treat high blood pressure. There are a few different types of CCB and each works in a slightly different way. Most of these medicines will have names that end in “ipine”.
- These are an important class of cardiovascular drugs that act by inhibiting L-type voltage-sensitive calcium channels in smooth muscle and heart. There are five pharmacologically distinct subclasses of calcium channel blockers.
Mechanism of action: A calcium channel is embedded in a cell membrane.
- In the body’s tissues, the concentration of calcium ions (Ca2+) outside cells is normally about 10000-fold higher than the concentration inside cells. When these cells receive a certain signal.
- The channels open, letting calcium rush into the cell. The resulting increase in intracellular calcium has different effects on different types of cells.
- Calcium channel blockers prevent or reduce the opening of these channels and thereby reduce these effects.
- Several types of calcium channels occur, with several classes of blockers, but almost all of them preferentially or exclusively block the L-type voltage-gated calcium channel.
- Voltage-dependent calcium channels are responsible for the excitation-contraction coupling of skeletal, smooth, and cardiac muscle and for regulating aldosterone.
And cortisol secretion in endocrine cells of the adrenal cortex. In the heart, they are also involved in the conduction of the pacemaker signals.
“Early warning signs of gaps in understanding antianginal basics: Common questions”
Classification of Calcium Channel blockers
- Class 1 Phenylalkylamines
- For Example, Verapamil
- Class 2 Benzothiazepines
- For Example, Diltiazem
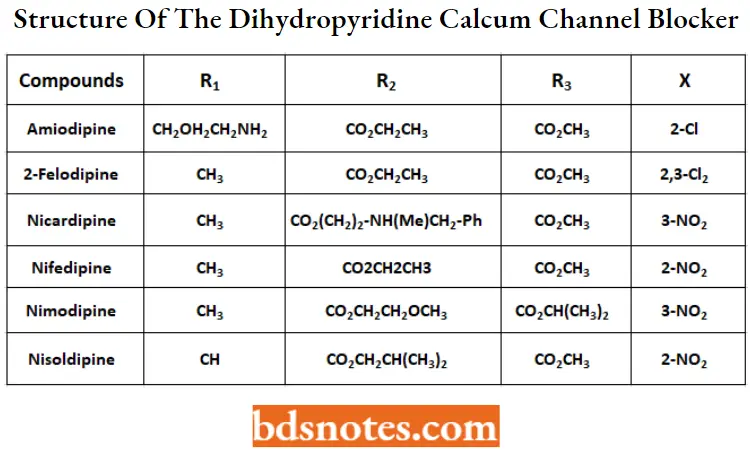
- Class 3 Dihydropyridines.
- First generation – Nifedipine
- Second generation – Felodipine
- Third generation – Amlodipine
Class 1 Phenylalkylamines
Verapamil
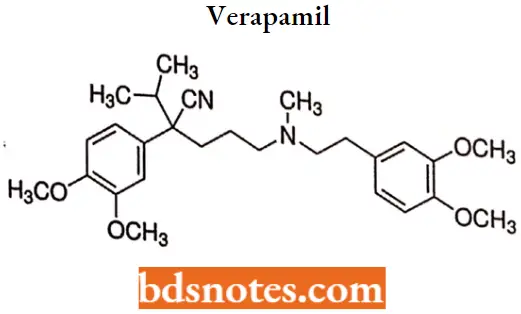
Verapamil IUPAC Name: (RS)-2-(3,4-Dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl]-(methyl)aminol}-2-prop-2-ylpentanenitrile.
Verapamil MOA: Verapamil inhibits voltage-dependent calcium channels. Specifically, its effect on L-type calcium channels in the heart causes a reduction in ionotropy and chronotropy, thus reducing heart rate and blood pressure.
Verapamil’s mechanism of effect in cluster headaches is thought to be linked to its calcium-channel blocker effect, but which channel subtypes are involved is presently not known.
Verapamil SAR: Verapamil is a synthetic compound possessing structural similarity to papaverine. It is a chiral compound where the (+)-isomer is more potent than the (-)-isomer as a calcium channel blocker.
- Removal of the orthomethoxy group gives inactive species.
- The essential structural features of this class of drugs are The benzene ring. Tertiary amino nitrogen, which is almost completely charged at physiological pH. The isopropyl group is not essential for activity.
Verapamil Metabolism: It is metabolized in the liver to at least 12 inactive metabolites (though one metabolite, or verapamil, retains 20% of the vasodilating activity of the parent drug).
Preferential metabolic tests involve N-dealkylation, followed by O-demethylation and subsequent conjugation of product.
Verapamil Therapeutic Uses: Verapamil is also used to prevent chest pain (angina). It may help to increase your ability to exercise and decrease how often you may get angina attacks.
Verapamil is also used to control your heart rate if you have a fast or irregular heartbeat (such as atrial fibrillation). It helps to lower the heart rate, helping you to feel more comfortable and increase your ability to exercise.
Verapamil Adverse reactions: The most common side effect of Verapamil is constipation (7.3%). Other side effects include dizziness (3.3%), nausea (2.7%), low blood pressure (2.5%), and headache 2.2%.
Other side effects seen in less than 2% of the population include edema, congestive heart failure, pulmonary edema, fatigue, elevated liver enzymes, shortness of breath, low heart rate, atrioventricular block, rash, and flushing.
Bepridil hydrochloride
“Asymptomatic vs symptomatic effects of ignoring antianginal principles: Q&A”
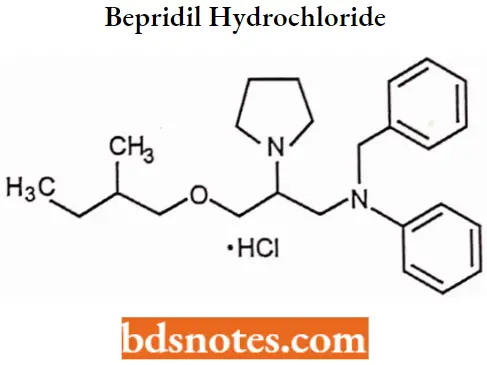
Bepridil hydrochloride IUPAC Name: N-benzyl-N-[3-(2-methylpropoxy)-2-(pyrrolidin-l-yl)propyl]aniline hydrate hydrochloride.
Bepridil hydrochloride MOA: Bepridil hydrochloride is a long-acting calcium-blocking agent with significant anti-anginal activity.
The drug produces significant coronary vasodilation and modest peripheral effects. Bepridil Hydrochloride is the hydrochloride salt form of bepridil, a calcium antagonist.
Bepridil hydrochloride Metabolism: It is rapidly and completely absorbed after oral administration. The time to peak bepridil plasma concentration is about 2 to 3 hours.
Over ten days, approximately 70% of a single dose of VASCOR (bepridil) is excreted in the urine and 22% in the feces, as metabolites of bepridil. The excretion of unmetabolized drugs is negligible.
Bepridil Hydrochloride Therapeutic Uses: Bepridil Hydrochloride is a medicine that is used for the treatment of Hypertension, Atrial fibrillation, and other conditions.
Bepridil hydrochloride Adverse Reactions: The most commonly reported side-effects of Bepridil Hydrochloride are gastrointestinal disorders. Bepridil also can cause increased or decreased heart rate and other abnormal rhythms.
Other side effects that can occur among patients taking bepridil include flu-like symptoms, muscle aches, fever, nausea, vomiting, loss of appetite, constipation, gas, stomach ache dry mouth, diarrhea, and headache.
Class 2 Benzothiazepines
Diltiazem hydrochloride
“Can targeted interventions improve outcomes using antianginal knowledge? FAQs provided”
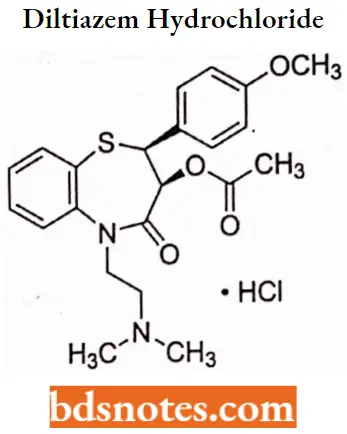
Diltiazem hydrochloride IUPAC Name: cis-(+)-[2-(2-Dimethylaminoethyl)-5-(4-methoxyphenyl)-3-oxo-6-thia-2-azabicyclo[5.4.0]undeca-7,9,11-trien-4-yl]ethanoate.
Diltiazem hydrochloride MOA: It relaxes the smooth muscles in the walls of arteries, which opens (dilates) the arteries, allows blood to flow more easily, and lowers blood pressure.
Additionally, it lowers blood pressure by acting on the heart itself to reduce the rate, strength, and conduction speed of each beat.
Diltiazem hydrochloride SAR: The tertiary basic nitrogen is essential for activity. N-demethyl derivatives as well as quaternization products are either less active or not active compared to the parent compound.
Diltiazem hydrochloride Metabolism: Diltiazem is metabolized by and acts as an inhibitor of the CYP3A4 enzyme.
Diltiazem hydrochloride Therapeutic Uses: Diltiazem is used in the treatment of hypertension, angina pectoris, and some types of arrhythmia.
- It relaxes the smooth muscles in the walls of arteries, which opens (dilates) the arteries, allows blood to flow more easily, and lowers blood pressure.
- Additionally, it lowers blood pressure by acting on the heart itself to reduce the rate, strength, and conduction speed of each beat.
Adverse Reactions: Dizziness, lightheadedness, weakness, nausea, flushing, constipation, and headache may occur.
Class 3 Dihydropyridines (DHP)
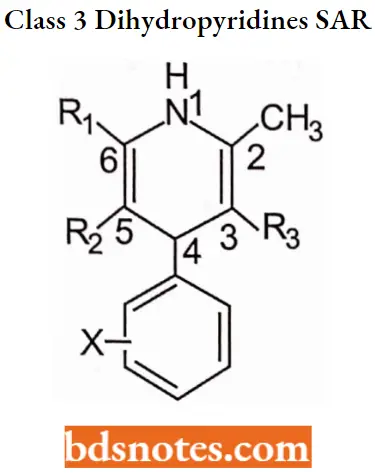
Class 3 Dihydropyridines SAR: The structure-activity relationships for 1, 4-DHP derivatives indicate that the following structural features are important for activity.
“Differential applications of short-acting vs long-acting nitrates: Questions answered”
- A substituted phenyl ring at the C4, position optimizes activity (heteroaromatic rings, such as pyridine, produce similar therapeutic effects; but are not used due to observed animal toxicity). C4 substitution with a small nonplanar alkyl or cycloalkyl group decreases activity.
- Phenyl ring substitution (X) is important for size and position rather than for electronic nature. Compounds with ortho or meta substitutions possess optimal activity, while those that are unsubstituted or contain a para-substitution show a significant decrease in activity.
- Electron-withdrawing ortho or meta-substituents or electron-donating groups demonstrated good activity. The importance of the ortho and meta-substituents is to provide sufficient bulk to “lock” the conformation of the 1, 4-DHP such that the C4 aromatic ring is perpendicular to the 1, 4-dihydropyridine ring. This perpendicular conformation has been proposed to be essential for the activity.
- The 1, 4-dihydropyridine rings are essential for activity. Substitution at the Nl, position or the use of oxidized (piperidine) or reduced (pyridine) ring systems greatly decreases or abolishes activity.
- Ester groups at the C3 and C5 positions optimize activity. Other electron withdrawing groups show” decreased antagonist activity and may even show agonist activity. For example, the replacement of the C3 ester of isradipine with a NO2 group produces a calcium channel activator or agonist.
- Except for amlodipine, all 1, 4-DHPs have C2 and C6 methyl groups. The enhanced potency of amlodipine (vs. Nifedipine) suggests that the 1, 4-DHP receptor can tolerate larger substituents at this position and that enhanced activity can be obtained by altering these groups.
Structure of the Dihydropyridine Calcium Channel Blockers
“Difference between nitrates and beta-blockers: Q&A explained”
Nifedipine
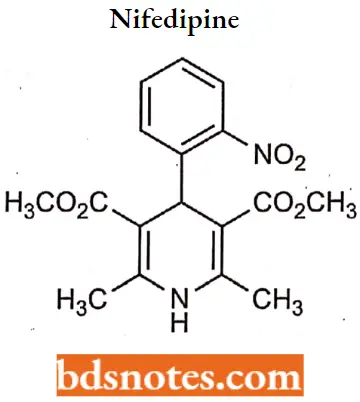
Nifedipine IUPAC Name: 3,5-dimethyl 2,6-dimethyl-4-(2-nitrophenyl)-l,4-dihydropyridine-3,5-dicarboxylate.
Nifedipine MOA: Nifedipine is a calcium channel blocker. Although nifedipine and other dihydropyridines are commonly regarded as specific to the L-type calcium channel, they also possess nonspecific activity towards other voltage-dependent calcium channels.
Nifedipine has additionally been found to act as an antagonist of the mineralocorticoid receptor, or as an antimineralocorticoid.
Nifedipine Metabolism: Hepatic metabolism via cytochrome P450 system. Predominantly metabolized by CYP3A4, but also by CYP1A2 and CYP2A6 isozymes.
Nifedipine Therapeutic Uses: Nifedipine, is a medication used to manage angina, high blood pressure, Raynaud’s phenomenon, and premature labor. It is one of the treatments of choice for Prinzmetal angina.
It may be used to treat severe high blood pressure in pregnancy. Its use in preterm labor may allow more time for steroids to improve the baby’s lung function and provide time for the transfer of the mother to a well-qualified medical facility before delivery.
Nifedipine Adverse reactions: Common side effects include lightheadedness, headache, feeling tired, leg swelling, cough, and shortness of breath. Serious side effects may include low blood pressure and heart failure.
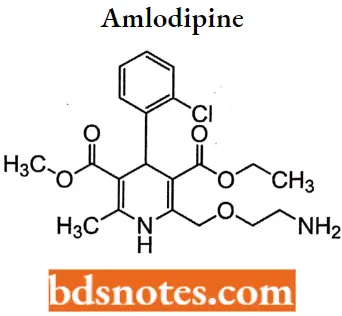
Amlodipine
“Most common complications of poorly understood antianginal concepts: FAQs”
Amlodipine IUPAC Name: (RS)-3-ethyl 5-methyl 2-[(2-aminoethoxy)methyl]-4-(2-chlorophenyl)-6-methyl-l,4- dihydropyridine-3,5-dicarboxylate.
Amlodipine MOA: Amlodipine decreases arterial smooth muscle contractility and subsequent vasoconstriction by inhibiting the influx of calcium ions through L-type calcium channels.
- Calcium ions entering the cell through these channels bind to calmodulin. Calcium-bound calmodulin then binds to and activates myosin light chain kinase (MLCK).
- Activated MLCK catalyzes the phosphorylation of the regulatory light chain subunit of myosin, a key step in muscle contraction.
Amlodipine Metabolism: Metabolized extensively (90%) to inactive metabolites via the cytochrome P450 3A4 isozyme.
Amlodipine Therapeutic uses: Amlodipine is used in the management of hypertension and coronary artery disease in people with either stable angina (where chest pain occurs mostly after physical or emotional stress) or vasospastic angina (where it occurs in cycles) and without heart failure.
It can be used as either monotherapy or combination therapy for the management of hypertension or coronary artery disease.
Amlodipine Adverse reactions: Some common dose-dependent side effects of amlodipine include vasodilatory effects, peripheral edema, dizziness, palpitations, and flushing.
Peripheral edema (fluid accumulation in the tissues) occurs at a rate of 10.8% at a 10-mg dose (versus 0.6% for placebos) and is three times more likely in women than in men.
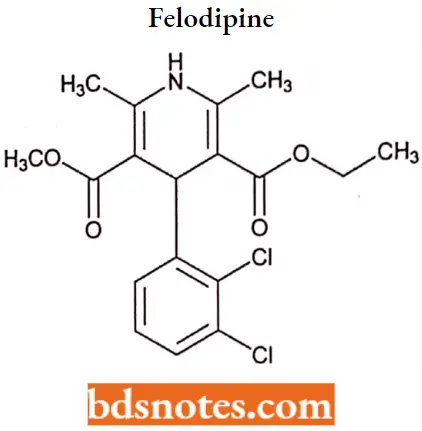
Felodipine
“Treatment scope in adults vs children for antianginal therapies: FAQs”
Felodipine IUPAC Name: (RS)-3-ethyl 5-methyl 4-(2,3-dichlorophenyl)-2,6-dimethyl-l,4-dihydropyridine-3,5- dicarboxylate.
Felodipine MOA: Felodipine is a calcium channel blocker. Felodipine has additionally been found to act as an antagonist of the mineralocorticoid receptor, or as an antimineralocorticoid.
Felodipine Metabolism: Hepatic metabolism primarily via cytochrome P450 3A4. Six metabolites with no appreciable vasodilatory effects have been identified.
Felodipine Therapeutic Uses: Felodipine is used to treat high blood pressure (hypertension). Lowering high blood pressure helps prevent strokes, heart attacks, and kidney problems.
Felodipine is known as a calcium channel blocker. By blocking calcium, this medication relaxes and widens blood vessels so blood can flow more easily.
Felodipine Adverse Reactions: The only very common side effect, occurring in more than 1/10 people, is pain and swelling in the arms and legs.
Common side effects, occurring in between 1% and 10% of people, include flushing, headache, heart palpitations, dizziness, and fatigue. Felodipine can exacerbate gingivitis.
Nicardipine
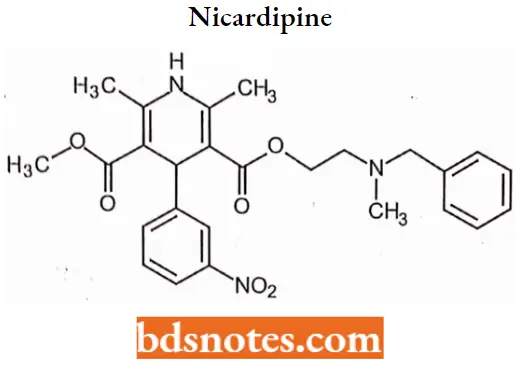
Nicardipine IUPAC Name: 2-[benzyl(methyl)amino]ethylmethyl-2,6-dimethyl-4-(3-nitrophenyl)-l,4-dihydropyridine-3,5-dicarboxylate.
Nicardipine MOA: Its mechanism of action and clinical effects closely resemble those of nifedipine and the other dihydropyridines (amlodipine, felodipine), except that nicardipine is more selective for cerebral and coronary blood vessels. Nicardipine also has a longer half-life than nifedipine.
Nicardipine Metabolism: Nicardipinel-ICl is metabolized extensively by the liver.
Nicardipine Therapeutic Uses: Nicardipine is used with or without other medications to treat high blood pressure (hypertension). Lowering high blood pressure helps prevent strokes, heart attacks, and kidney problems.
Nicardipine is called a calcium channel blocker. It works by relaxing blood vessels so blood can flow more easily.
Nicardipine Adverse reactions: Dizziness, lightheadedness, headache, flushing, or swelling of ankles or feet may occur.
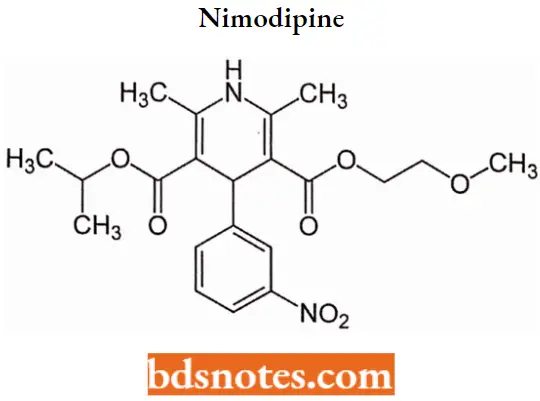
Nimodipine
“Why are antianginal mechanisms often misunderstood in practice? Questions answered”
Nimodipine IUPAC Name: 3-(2-methoxyethyl) 5-propan-2-yl, 2,6-dimethyl-4-(3-nitrophenyl)-l/4-dihydropyridine-3,5-dicarboxylate.
Nimodipine MOA: Nimodipine binds specifically to L-type voltage-gated calcium channels. There are numerous theories about its mechanism in preventing vasospasm.
Nimodipine has additionally been found to act as an antagonist of the mineralocorticoid receptor, or as an antimineralocorticoid.
Nimodipine Metabolism: Nimodipine is metabolized in the first pass metabolism. The dihydropyridine ring of the nimodipine is dehydrogenated in the hepatic cells of the liver, a process governed by cytochrome P450 isoform 3A (CYP3A).
This can be completely inhibited, however, by troleandomycin (an antibiotic) or ketoconazole (an antifungal drug).
Nimodipine Therapeutic Uses: Nimodipine’s main use is in the prevention of cerebral vasospasm and resultant ischemia, a complication of subarachnoid hemorrhage (a form of cerebral bleeding).
Specifically from ruptured intracranial berry aneurysms irrespective of the patient’s post-ictus neurological condition.
Nimodipine Adverse reactions: For the high dosage group (90 mg) less than 1% of the group experienced adverse conditions including itching, gastrointestinal hemorrhage, thrombocytopenia, neurological deterioration, and vomiting.
Diaphoresis, congestive heart failure, hyponatremia, decreasing platelet count, disseminated intravascular coagulation, and deep vein thrombosis.
Antianginal Drugs Multiple Choice Questions And Answers
Question 1. Nitric oxide half-life:
- Less than five seconds
- About a minute
- Ten minutes
- Sn hour
Answer: 1. Less than five seconds
Question 2. Which of the following types of angina can occur when lying flat?
- Unstable angina
- Variant angina
- Prinzmetal angina
- Decubitus angina
Answer: 4. Decubitus angina
Question 3. Which of the following advice would you not give a patient Who had just had angina 3ild IPs how stable?
- Advice the patient to increase the frequency and intensity of exercise
- Advise the patient to have moderate regular exercise
- Advice patient to adhere to prescribed medications
- Advise patient to in future call an ambulance if GTN is unable to relieve chest pain during angina episodes.
“Cost of ignoring antianginal principles vs benefits of systematic approaches: Q&A”
Answer: 1. Advice the patient to increase the frequency and intensity of exercise
Question 4. How is angina diagnosed?
- Exercise tolerance test
- ECG
- Clinical criteria
- Cardiac makers in the blood
Answer: 3. Clinical criteria
Question 5. Why is it generally advised not to give verapamil with a beta blocker?
- Risk of hypertension
- They can safely be used together, there is no risk
- Risk of tachycardia
- Risk of bradycardia
Answer: 4. Risk of bradycardia
Question 6. All of the following medicines can be given as first-line regular treatment to prevent stable angina except?
- Metoprolol 25 – l00mg bd
- Aspirin 75 mg od
- Diltiazem 30-120mg tablets tds
- Atenolol 25-100mg od
Answer: 2. Aspirin 75 mg od
Question 7. Which of the following types of angina is caused by coronary spasm?
- Stable angina
- Unstable angina
- Crescendo angina
- Prinzmetal angina
Answer: 4. Prinzmetal angina
“Is antianginal-related risk reversible if addressed promptly? Answer provided”
Question 8. N’on-dihydropyridine calcium channel blockers relax vascular smooth muscles and dilate coronary and peripheral arteries. They also act as beta-adrenoceptor blockers. Which of the following medicines belong to this class of drugs?
- Verapamil
- Nifedipine
- Felodipine
- Nicardipine hydrochloride
Answer: 1. Verapamil
Question 9. The aim of angina treatment includes all of the following?
- To relieve pain
- To prevent pain
- To improve prognosis
- To improve the outcome of drug therapy
Answer: 4. To improve the outcome of drug therapy
Question 10. Organic nitrates relax the vascular smooth muscle by
- Increasing intracellular cyclic AMP
- Increasing intracellular cyclic GMP
- Decreasing intracellular cyclic AMP
- Both (2) and (3)
Answer: 2. Increasing intracellular cyclic GMP
Antianginal Drugs Short Question Answers
Question 1. What is angina pectoris?
Answer:
Angina pectoris, commonly known as angina, is severe chest pain due to ischemia (a lack of blood, hence a lack of oxygen supply) of the heart muscle, generally due to obstruction or spasms of the coronary arteries.
Coronary artery disease, the main cause of angina, is due to atherosclerosis of the cardiac arteries.
Question 2. What are the different types of Angina?
Answer:
- Stable! angina-In thin type the atherosclerotic plaque and inappropriate vasoconstriction (caused by endothelial damage) reduce the blood vessel lumen diameter. Hence there is a reduction in blood flow.
- Unstable angina-In unstable angina, the rapture of the plaque triggers platelet aggregation, thrombus formation, and vasoconstriction. Depending upon plaque rapture this leads to non-Q wave (non-ST elevation) or Q wave (ST elevation).
- Variant angina-In this type atherosclerotic plaques are absent, and ischemia is caused by intense vasospasm. It occurs more In younger women.
“Success rate of interventions using modern antianginal techniques: FAQ”
Question 3. Give MOA of organic nitrates.
Answer:
Within the body, organic nitrates are chemically reduced to release NO. No is an endogenous signaling molecule that causes vascular smooth muscle relaxation.
- The various organic nitrates give rise to NO by different chemical and biochemical mechanisms. Organic nitrates have the chemical structure RN02.
- The nitro group is reduced to form NO in the presence of specific enzymes and extracellular and intracellular reductants (For Example. thiols). The NO activates guanylyl cyclase.
- The activated guanylyl cyclase increases the formation of cGMP from GTP. The cGMP activates myosin-LC phosphatase. The activated myosin-LC phosphatase causes the dephosphorylation of myosin-LC-(P) to myosin-LC. This relaxes the smooth muscle and causes vasodilatation.
Leave a Reply