Pharmacology Synopsis Notes
General Pharmacology
- Some Important Definitions
- Pharmacology: Study of substances that interact with living systems through chemical processes, especially by binding to regularity molecules.
- Pharmacogenetics: Study of genetic variations that cause individual differences in drug response.
- Pharmacodynamics: Study of biological and therapeutic effects of drugs and their mechanism of action
- Pharmacokinetics: Study of absorption, distribution, metabolism, and excretion of drugs and their relationship to pharmacological response.
- Clinical pharmacology: Scientific study of drugs & includes pharmacodynamic and pharmacokinetic investigations.
- Chemotherapy: Treatment of systemic infection (cancer therapy as well) with specific drugs with no or minimal effects on the host cells.
- Pharmacy: Science of identification, selection, preservation, standardization, compounding ad dispensing of medicinal substances.
- Bioavailatylity: Percentage of drug that is absorbed from a given dosage from reaches the systemic- circulation following non-vascular administration.
- Bio-transformation: Chemical alteration of a drug within a living organism.
- Drug: Is any substance or product that is used or is intended to be used to modify or explore physiological systems or pathological states for the benefit of the recipient.
Pharmacology Synopsis Notes: Key Concepts and Drug Mechanisms
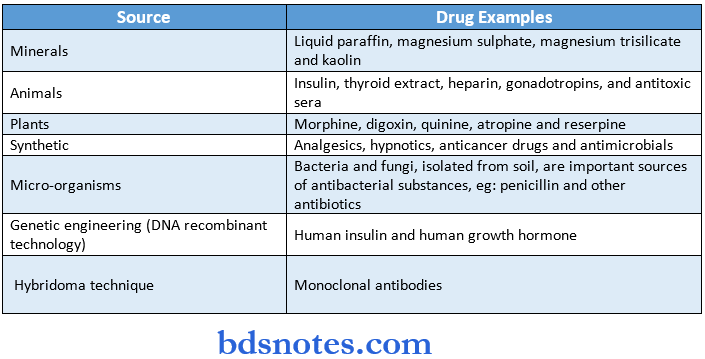
Various sources of drugs:
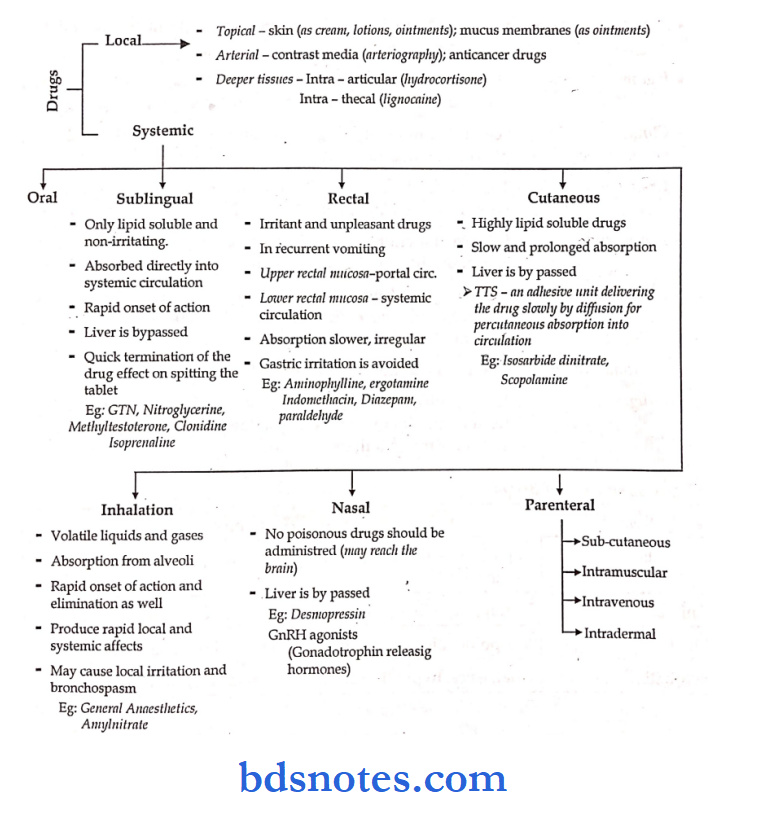
Routes Of Durg Administration:
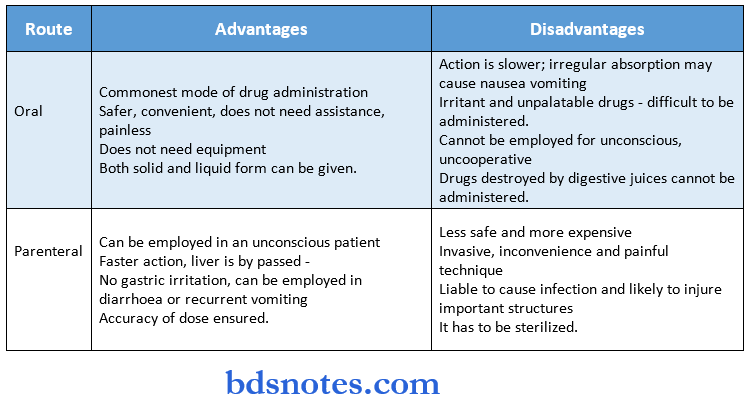
- Factors affecting drug absorption and bio-availability
Drug-related:
- Physical state – liquids > solids; crystalloids > colloids.
- Dosage form – particle size affects absorption; a tablet containing a large aggregate of tire active compound is absorbed poorly.
- Disintegration time and dissolution rate.
- Metabolism of drug – extensive first-pass metabolism reduces bioavailability.
- Concentration – passive transport depends on the cone.
- Absorption of the drug in a cone.
- Solution dilute solution.
Patient-related:
pH of GIT fluid and blood – weakly acidic drugs are rapidly absorbed.
- GI transmit time – rapid absorption occurs if the drug is given on an empty stomach; increased peristalsis reduces drug absorption.
- Enterohepatic cycling – increases the bioavailability of a drug. Eg: morphine.
- Area of absorbing surface – small intestine > stomach.
- Disease states – absorption affected in conditions like malabsorption, thyrotoxicosis, achlorhydria, and biliary obstruction.
Bio-transformation
- Is necessary to render lipid-soluble compounds lipid insoluble so that they are not reabsorbed at renal tubules and hence more readily excretable
- Metabolic products are often less pharmacologically active than the parent drug.
- By studying bio-transformation, drug metabolizing enzymes have been exploited in the design of pharmacologically inactive prodrugs that are converted to active molecules in the body.
- Sites – Liver, kidney, intestine, lungs, and plasma, (primary site)
Role of Biotransformation – metabolic transformation by enzymes
- Inactivation
- Active metabolite from an active drug (Hofmann elimination)
- Activation of inactive drug (prodrug)
- Spontaneous change into other substances (without enzymes)
Reactions
Phase 1 (Non-synthetic)
- Oxidation – Acetanilide, barbiturates, amphetamine, benzodiazepines
- Reduction – Chloral hydrate, chloramphenicol
- Hydrolysis – Procaine, lignocaine, Oxytocine, pethidine, procainamide
- Cyclization – proguanil
- Decyclization – Barbiturates, phenytoin.
Phase 2 (Synthetic)
- Conjugation – Glucuronidation – aspirin, morphine, diazepam, Metronidazole, chloramphenicol
- Acetylation – Sulfonamides, PAS, Hydralazine, Isoniazid
- Methylation – Adrenaline, histamine, Nicotinic acid
- Sulcate conjugation – chloramphenicol
- Glycine conjugation – salicylates.
Factors affecting drug metabolism
- Age and sex, genetic determinants
- Nutritional states, presence of disease/hepatitis/ renal damage Route and duration of administration
- Simultaneous administration of drugs Environmental factors (altitude, temperature).
Pharmacology Summary Notes: Drugs, Dosages, and Mechanisms
Prolongation of drug action
- Retarding {prolonging) drug absorption
- Sustained release tablets, administration after full stomach (oral)
- Reduction is the solubility of the drug or as an oily solution
- Combination with a protein, esterification, implantation of pellets, TTS.
- Inhibiting drug metabolism in the liver
- Retarding renal excretion – eg: probenecid with ampicillin/penicillin
- Increased protein binding.
Effects of protein binding on drugs:
- Assists enteral absorption of a drug
- Acts as a temporary store for it and thereby prolong its action.
- Causes prolonged, low levels of free drug.
- Delays its metabolic degradation
- Delays its excretion; and diminishes its penetration into the CNS.
Adverse Drug Effects
Adverse drug effects: Any undesirable and unintended consequence of drug administration.
Adverse drug reaction: Suspected to be due to a drug occurring at doses normally used in man.
Classification of adverse drug effects:

Side effects: Occurs at the therapeutic level; are unwanted but often unavoidable pharmacodynamic effects.
Toxic effect: Occurs at overdosage or prolonged dosage due to excessive pharmacological action of the drug
Secondary effect: Indirect consequences of the primary action of the drug.
Examples: Superinfection (Tetracycline Chloramphen icon, Ampicillin, Amoxicillin).
Drug withdrawal reactions: It is a worsening of the clinical condition for which the drug was being used after a sudden interruption of therapy.
Examples: Corticosteroids, Clonidine, p- blockers, and Antiepileptics.
Intolerance: It is the appearance of characteristic toxic effects of a drug in an individual at therapeutic effects due to the low threshold of the individual.
Idiosyncracy: It is the uncharacteristic or bizarre effect due to abnormal reactivity. It is genetically determined.
Examples: Quinine causes cramps diarrhea etc. in some individuals.
Drug allergy: It is an immunologically mediated reaction (unrelated to pharmacodynamic profile; largely independent of dosage).
Drug Dependence: Is used to cover both drugs of addiction i.e., which cause both psychological and physiological dependence, as well as drugs, which only cause habit formation i.e., which causes only psychological dependence and no physiological dependence.
Pharmacology Notes: Drug Classes, Actions, and Side Effects
Classification Of Drugs
Drugs Acting On Arms
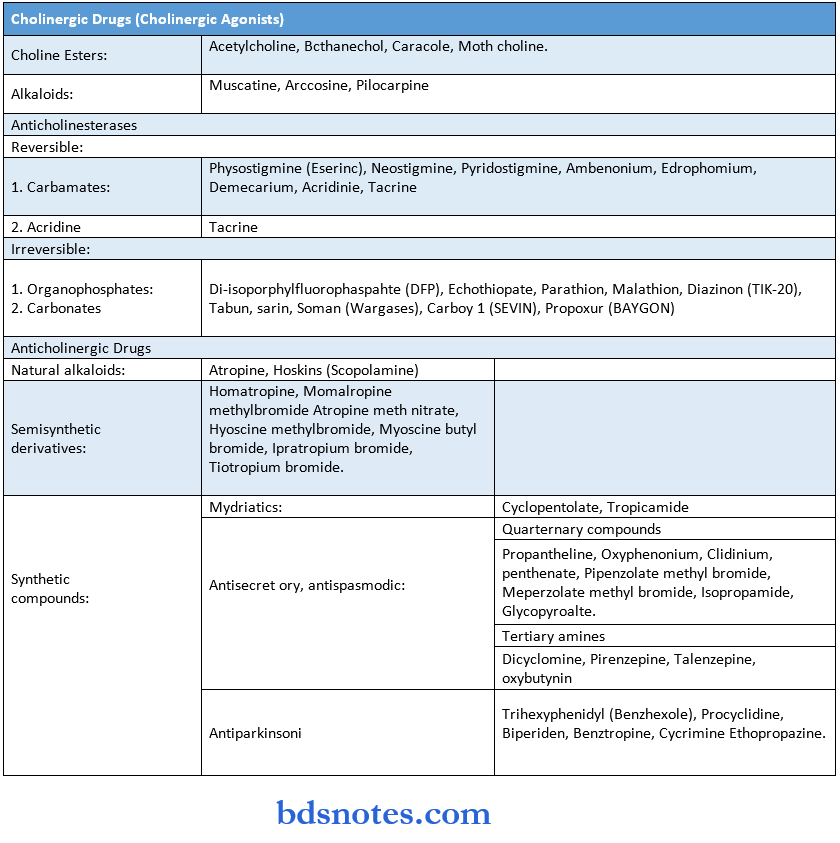
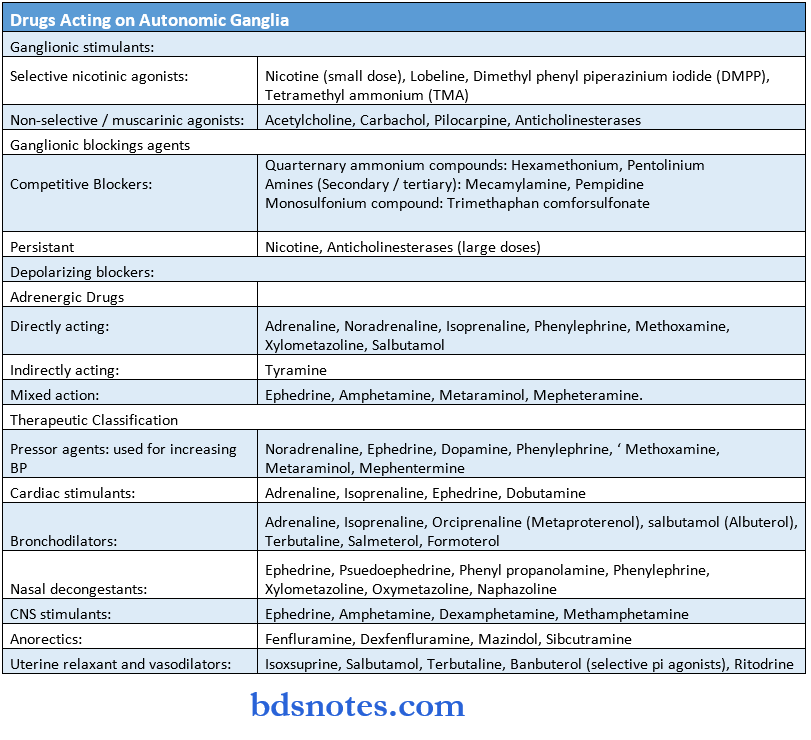
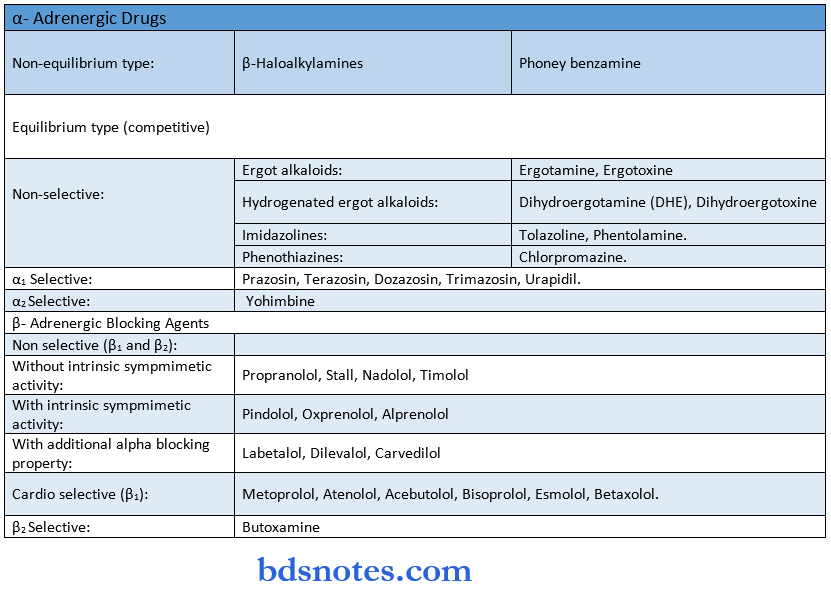
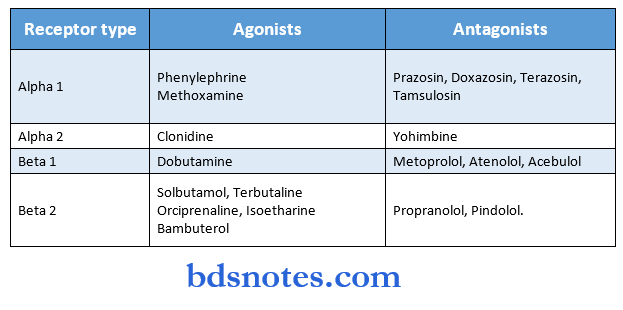
Drugs Acting On Peripheral Nervous System
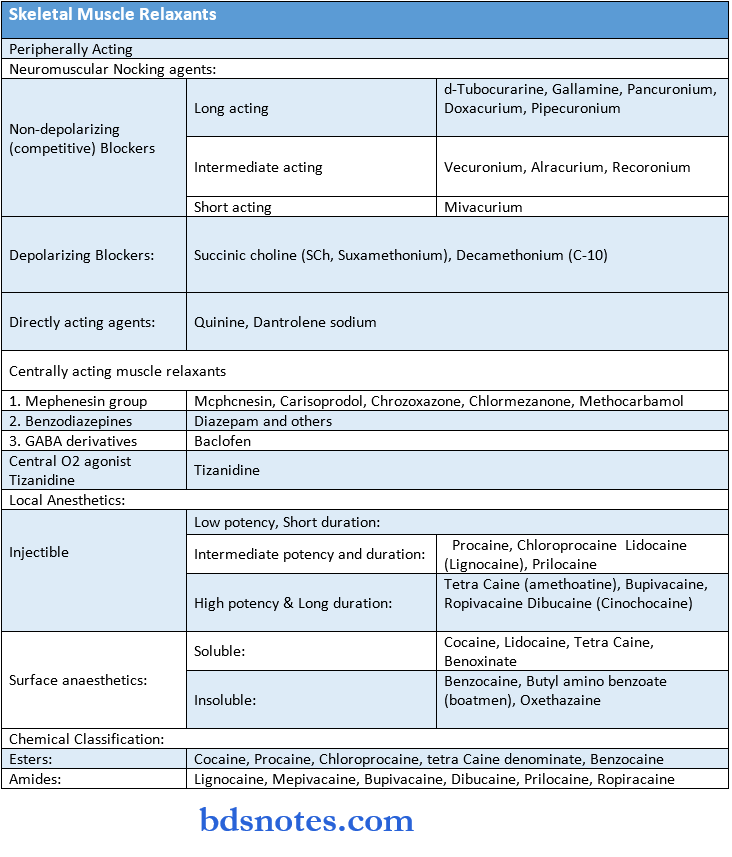
Autocoids And Related Drugs
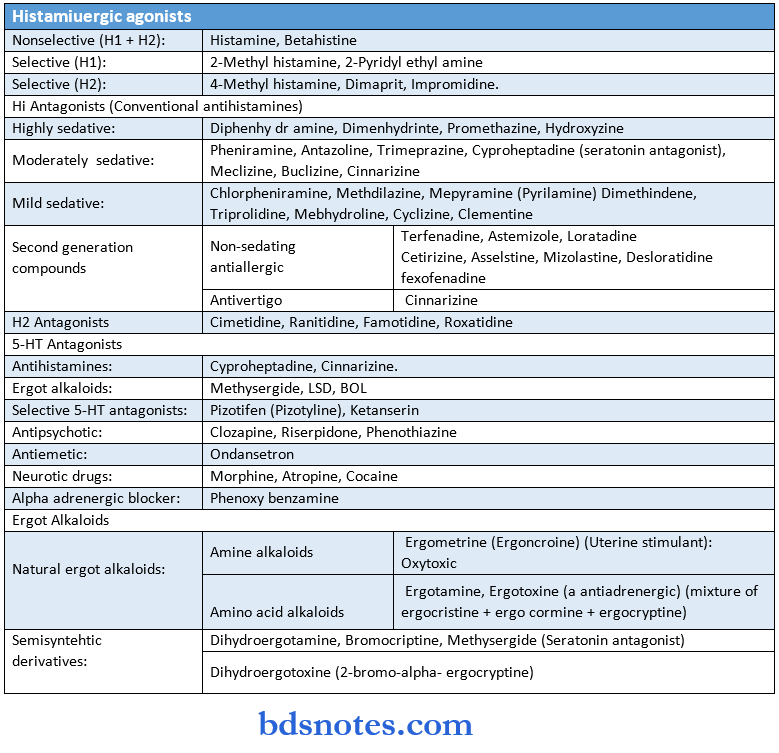

Respiratory System Drugs
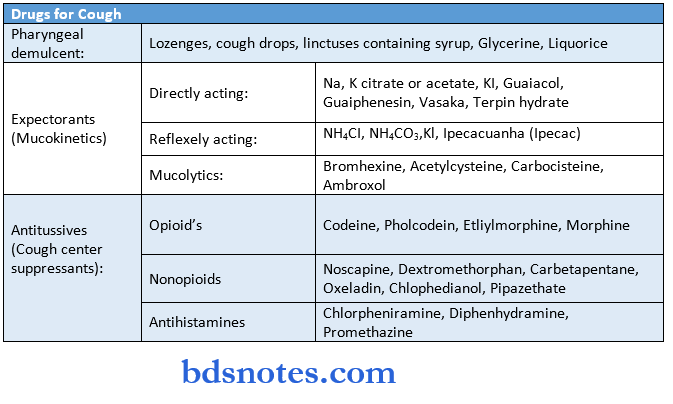
Hormones And Related Drugs
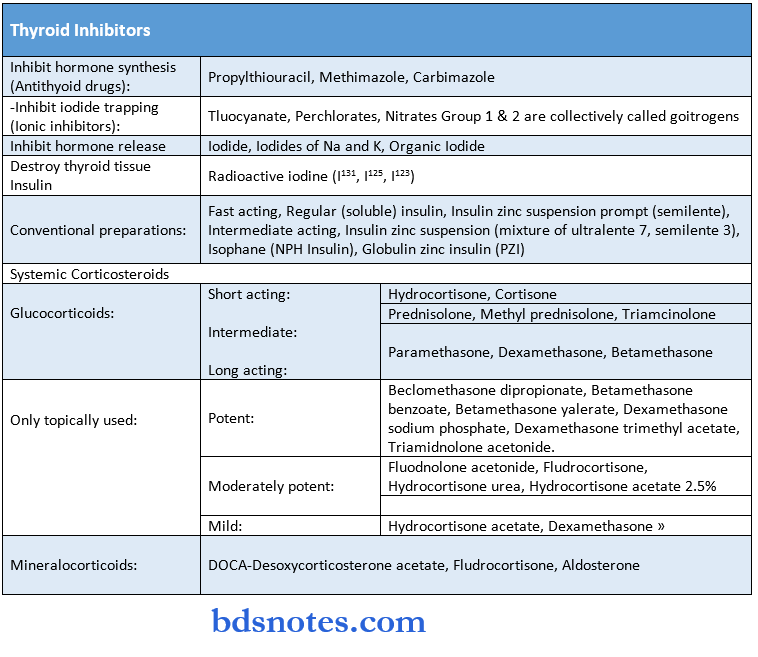
Drugs Actings On CNS
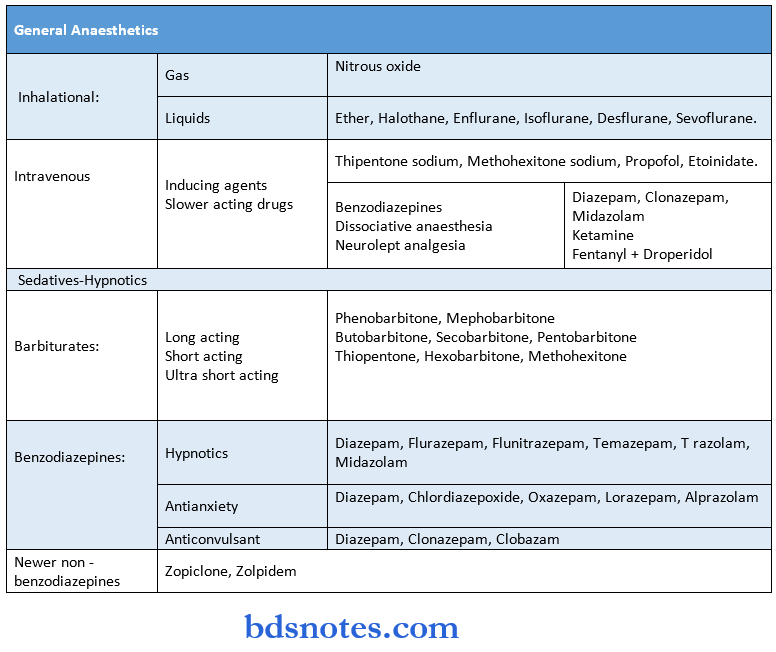
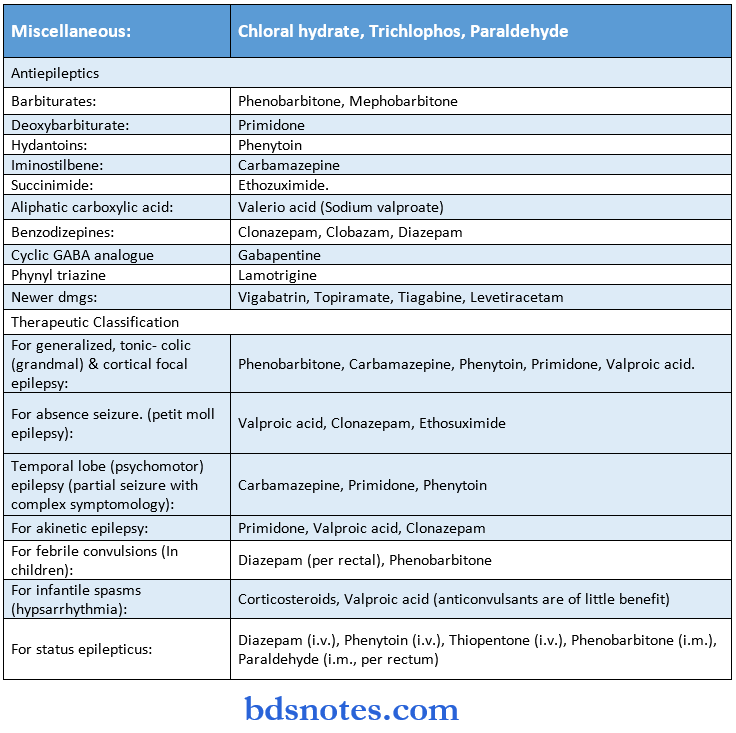
Cardiovascular Drugs
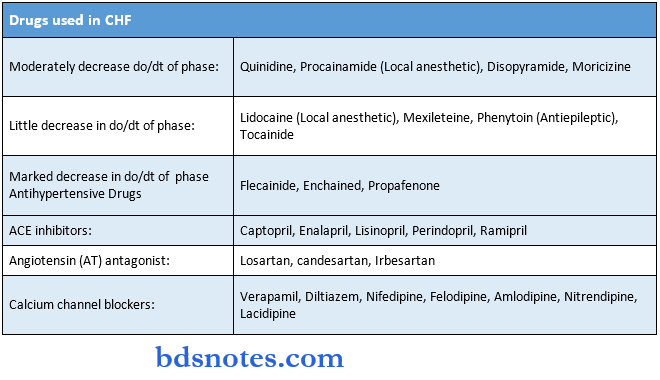
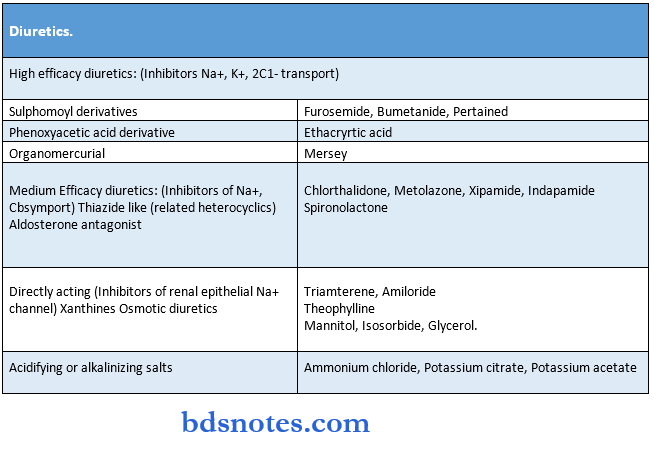
Drugs Acting On Kidney
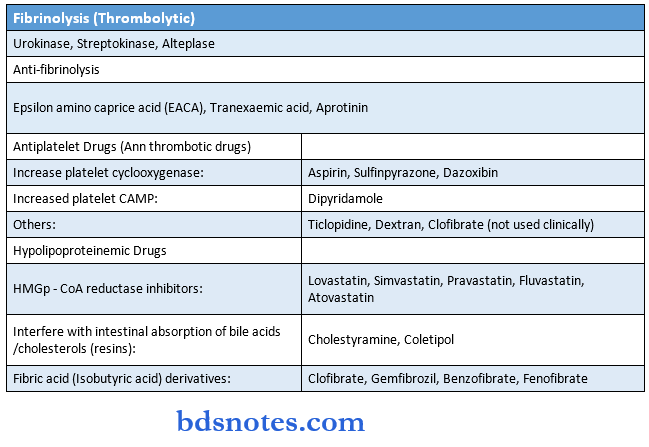
Drugs Affecting Blood And Blood Formation
Gastrointestinal Drugs
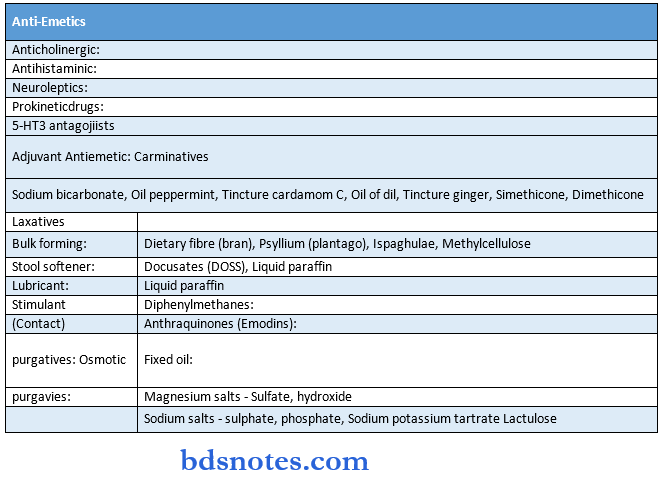
Experiments -1 KMNO4 Solution( Mouth Wash)
Aim:
- Prepare and dispense 60 ml of KMNOi 1 in 4000 for use as a mouthwash.
Calculations:
- 4000 ml→ 1000 mg 60 ml→?
- 60/4000×1000 = 15 mg
- i.e. 15 mg of KMNO4 in 60 ml will give 1 in 4000 dilution.
Ingredients:
- KMNO4—15 mg
- Squaad — 60 ml
Instruments:
- Weighing balance, measuring box, bottles.
Procedure:
- The sensitivity of the balance provided 100 mg. Ti means one cannot weigh less than 100 mg in balance. To overcome this difficulty, weigh 100 Mg of KMNO4 and dissolve in 100 ml of H2O from this take 150 ml and add the required quantity of water to make up to 60 ml. The solution is dispensed in a bottle and labeled suitably.
Mechanism of action:
KMNO4 is an oxidizing agent. Because of this, it is used as an antiseptic, disinfectant, and deodorant. It is also used as a cleaning agent in eczematous, ulcers, and abscesses.
Uses:
- 1 in 4000 solutions is used for mouthwash and gargling.
- 1 in 1000 is for cleaning skin wounds and ulcers.
- 1 in 500 is used for the irrigation of cavities.

Experiment- 2 Normal Saline
Aim:
- Prepare and dispense 100 ml of normal saline and give directions for use.
Principle:
- 100 ml of normal saline contains 900 mg of NaCl.
Procedure:
- Weight 900 mg of NaCl and dissolve in 50 ml of distilled water and transfer it to a plain white bottle and label it.

Experiment- 3 Dentrifices<Powders>
Aim:
- To dispense 30 gm of powders for cleaning the artificial denture.
Ingredients:
- Sodium Perborate: 10gm.
- Borax: 20 gm
- Menthol: 0.1 gm.
Procedure:
- Weight sodium perborate and borax. Take into a mortar and triturate well. Finally, add menthol, then pack it up and label it.
Uses and actions of Ingredients:
- Sodium perborate is an antiseptic, deodorant, and oxidizing agent.
- Borax is used as an abrasive agent.
- Menthol is used as a deodorant and flavoring agent.
- Artificial dentures should be cleaned with a brush and cold water to remove food particles. The denture then should be soaked in a detergent solution cleaned and sterilized it.
Experiment-4 Lotion Calamine <Lotions>
Aim:
- To dispense 30 ml of lotion calamine.
Ingredients:
- Calamine Powder: 4.5 gms.
- Zno: 1.5 gms.
- Bentonite: 0.9 gms
- Sodium Citrate: 150 gms
- Liquid Phenol: 0.15ml
- Glycerine: 1.5 ml
- Aqua add: 30ml
Procedure:
- Take the required quantities of calamine powder ZnO and bentonite in the mortar, finely powder, and mix thoroughly. Measure about 3/4th of the vehicle and dissolve in it, the required quantity of sodium citrate.
- Power a portion of it in mortar and triturate to form a smooth cream. Add the remaining amount of sodium citrate solution, and mix well. Add the required quantity of glycerine and liquid phenol info it.
- Add more vehicles to produce the prescribed volume. Transfer it to wide mouth bottle cork and label it.
Mechanism of Action:
1. Calamine is the principle ingredient. It contains
- ZnCo3 < 98%> and Fe203 <2%> .
- ZnCo3 → astringent action.
- Fe203 → gives pink color.
2. ZnO → acts as an adjuvant.
3. Bentonite → Fine particles of Mucl. It’s chemically hydrated aluminum silicate.
- It is used as suspending agent.
- It does not mix with water but absorbs water and its volume increases 12 times.
- It has got thixotropic properties.
4. Sodium citrate: It acts systematically.
- It acts as an anticoagulant.
- It also alkalinizes the urine.
- It is also used to neutralize the phenol.
5. Phenol → It has antiseptic and local anesthetic actions.
6. Glycerine → It is also suspending agent because of bentonite, lotions are viscous and thick. So to liquefy it, glycerine is added.
- Glycerine is also used to prevent the drying of lotion.
- It is hygroscopic.
Uses:
- As antipruritic.
- Uses as impetigo.
- Used as a dusting powder in napkin rashes occurring in children
- To prevent bed sores.
- In urticaria, eczema, and psoriasis.

Experiment -5 Mandler’s paint
Aim:
- To dispense 10 gm of pulp capping paste.
Ingredients:
- ZnO: 6.7 gms.
- Thymol: 3.3 gms.
Instruments:
- Hot water bath.
- Porcelain crucible.
- Porcelain tube.
- Knife blade.
- Small bottle.
Procedure:
- Melt thymol in a porcelain crucible and gradually add ZnO, while stirring, in a warm state, spread it on the tile. Let it cool. Scrap it off with a knife blade, and keep it in a well-stoppered bottle or in a plastic blade.
Mechanism of action:
- ZnO → acts as an astringent, and antiseptic that has soothing and protective action, ha some sedative properties.
- Thymol → acts as a deodorant.

Experiment -9 Cretakaolin mixture
Aim:
- To dispense 15 ml of antiseptic throat paint for a patient suffering from sore throat Ingredients:
Ingredients:
- Sodium salicylates: 1gm
- Sodium Bicarbonate: 1gm
- Liquid extracts of licorice: 03 gm (aromatic agent)
- Aqua add: 30ml.
Procedure:
- Dissolve 12 and KI in a portion of glycerine. Add the oil of peppermint and sufficient glycerine to produce the required volume. Mix well with a glass rod. Transfer to a suitable bottle and label.
Uses:
- Used as a local application in sore throat and tonsillitis.
Mechanism of action:
- H2 is a disinfectant and antiseptic.
- It increases the solubility of I2 in the vehicle.
- Oil of peppermint is added as a flavoring agent.
- Glycerine is a demulcent and sweetening agent. Because of its viscosity, it keeps the medicament at the site of application for a long time.
Experiment-8 Tartr Solvent
Aim:
- To dispense 40 ml of Tartar solvent.
Formulation:
- Citric acid : 3 gm. Liquid phenol: 1 ml. Aqua add: 40 ml.
Procedure:
- Citric acid is dissolved in Wh amount of H2O to this add liquid phenol and make u to desired volume with distilled water. Transfer it to a plain white bottle and label it.
Experiment-9 Creta kaolin Mixture
Aim:
- To dispense 30 ml of crema kaolin mixture.
Ingredients:
- Creba: 1.5gm.
- Light kaolin: 3.5 gm.
- Gum acacia: 600 mg.
- Tincture opi camphorata.
- Tincture car domain.
- Aqua add: 30 ml.
Uses:
- Cxeta: Native CaCCb – In differentiable insoluble in water CaCC>3 exhibits absorbent and demulcent properties.
Light kaolin:
- It is an adjuvant (Hydrated aluminum silicate).
- Demulcent and absorbent.
- Gum acacia – acts as suspending agent, and decreases viscosity.
- Tincture opi camphorate – constipating agent.
- Tinclnre cardamom – flavoring agent and carminative.
Procedure:
- As it is an indisputable mixture of mortar and pestle is used 600 mg of gum acacia is weighed and taken into mortar 20 m of water is added and triturated with the pestle 1.5 gm of Creta is weighed and added to mortar and triturated well 3.5 gm of light kaolin is weighted added and mixed well and the mixture is taken in an ounce glass. 1 ml of tincture opium camphorate is added and mixed well with glass road. Tincture cordmum is added and mixed well and water is added up to 30 ml.
Uses:
- Kaolin mixture – Non-specific diarrhea.
- These components decrease the sequence of diarrhea.
- They act by decreasing irritants to the intestine by toxic products. Thus reducing peristalsis.
- Also act as an adsorbent, and antacid to protect GIT.
Leave a Reply