Oxidative Deamination Explained: From Glutamate To Ammonia
Question 1. Give one example each for transamination and deamination of amino acids.
Answer:
Example of transamination reaction:
- Transfer of amino group to coenzyme pyridoxal phosphate to form pyridoxamine phosphate
Example of deamination of amino acids:
1. Oxidative deamination
- Catalyzation of glutamate by glutamate dehydrogenase to liberate ammonia
2. Non-oxidative deamination
- Deamination of hydroxy amino acids by PLP-dependent hydrates
Question 2. Oxidative deamination
Answer:
It is the liberation of free ammonia from the amino group of amino acids along with oxidation
Example:
- Amino acids oxidases act on respective amino acids to produce a ketoacid and ammonia.
Question 3. Classify amino acids based on their nutritional importance and give two examples of each.
Answer:
1. Essential amino acids
- They cannot be synthesized by the body
- Example: valine, leucine, lysine
2. Semi-essential amino acids
- Can be synthesized by adults but not by children
- Examples: arginine, histidine
3. Nonessential amino acids
- They are synthesized in the body
- Examples: glycine, serine, aspartate, alanine
Question 4. Define isoelectric pH and denaturation.
Answer:
Isoelectric pH:
- It is defined as the pH at which a molecule exists as a Twitter ion and carries no net charge.
- The molecule is neutral with
- Minimum solubility
- Maximum precipitability
- Least buffering capacity
- Value
- Casein – 4.6
- Albumin – 4.7
- Haemoglobin – 6.7
Denaturation of proteins:
- It is the phenomenon of the disorganization of native protein structures.
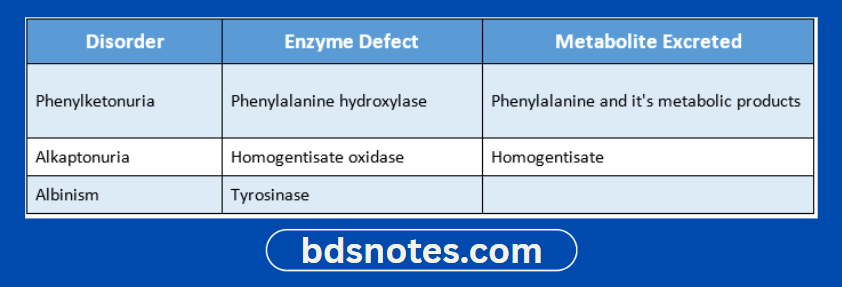
Question 5. Write the enzyme defect and the metabolite excreted in the urine of the following disorder
- Phenylketonuria
- Alkaptonuria
- Albinism
Answer:
Question 6. Name the various products formed from glycine
Answer:
- Products formed from glycine:
- NH4
- Carbon dioxide
- One carbon fragment as N5,
- N10 methylene THF.
Leave a Reply