Dental Plaster
Write a short note on setting of dental plaster.
Answer:
The setting of Dental Plaster:
When 1 g mol of calcium sulfate hemihydrates reacts with 1.5 g mol of water, the hemihydrates revert to dihydrate forming 1 g mol of calcium sulfate dihydrate by hydration, with liberation of 3900 calories of heat.
Here the heat which is evolved in reaction is equal to the heat originally used in calcinations.
- (CaSO4)2.H2O + 3H2O → 2CaSO4.2H2O + unreacted (CaSO4)2.½ H2O + Heat
- Hemihydrate + Water → Dihydrate + unreacted hemihydrates + 3900 cal/g mol
Three theories of settng have been put forward, i.e.
- Colloidal theory
- Hydration theory
- Dissolution-precipitation theory.
Out of these theories most acceptable theory is dissolution precipitation theory which is:
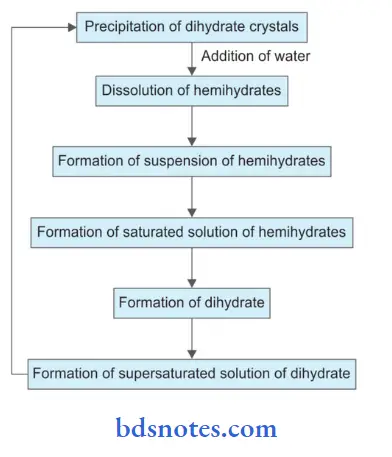
Dissolution-Precipitation Theory (Crystalline Theory):
This theory is widely accepted. According to the theory, the plaster dissolves and reacts to form gypsum crystals which interlock to form the set solid. The setting reaction is explained on the basis of difference in solubility of hemihydrates and dihydrate.
Hemihydrate is four times more soluble than dihydrate. When hemihydrate is mixed in water it forms a flid workable suspension. Hemihydrate dissolves until it forms a saturated solution. Some dihydrate is formed due to the reaction. The solubility of dihydrate is much less than hemihydrate, the saturated hemihydrate is supersaturated with respect to the dihydrate. All supersaturated solutions are unstable.
So the dihydrate crystals precipitate out. As the dihydrate precipitates out, the solution is no longer saturated with hemihydrate and so it continues to dissolve.The process continues until further dihydrate precipitates out of the solution. Initially, there is little reaction and thus little or no rise in temperature.
This time is referred to as the induction period. As the reaction proceeds gypsum is formed in the form of needle-like clusters called spherulites. Continued growth and intermeshing of crystals of gypsum leads to thickening and hardening of the mass into a strong solid structure.
Summary of Dissolution-Precipitation Theory:
Leave a Reply