Increased Mercury Emissions From Modern Dental Amalgams
Write a short note on the amalgamation reaction of low copper alloy.
Answer:
- Low-copper alloy is a type of silver amalgam alloy.
- Low-copper alloy contains less than 6% of copper.
Mercury Emissions From Modern Dental Amalgam
Amalgamation (Setting Reaction):
- When alloy powder and mercury are triturated, the silver and tin in the outer portion of the particle dissolve into the mercury.
“Understanding mercury emissions through FAQs: Sources, impacts, and solutions explained”
Modern Amalgam Mercury Release
- At the same time, mercury diffuses into the alloy particles and starts reacting with the silver and tin present in it, forming silver-mercury and tin-mercury compounds.
- Silver-tin compound is known as the gamma phase (γ).
- The silver-mercury compound is known as the gamma one (γ1) phase and the tin-mercury as the gamma two (γ2) phase.
“Importance of studying mercury emissions for dental professionals: Questions explained”
A simple reaction is:
Ag3Sn + Hg → Ag2 Hg3 + Sn8Hg + Ag3Sn unreacted
(γ) (γ1) (γ2) (γ)
The alloy particles do not react completely with mercury. About 27% of the original Ag3Sn remains as unreacted particles.
Dental Amalgam And Environmental Mercury Pollution
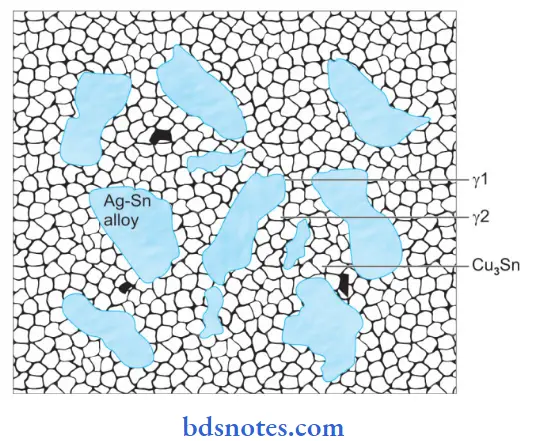
Microstructure:
Set amalgam consists of unreacted core particles, i.e. γ surrounded by a matrix of the reaction products γ1 and γ2
“Common challenges in addressing mercury emissions effectively: FAQs provided”
Leave a Reply