Reaction Mechanisms
A reaction mechanism is a detailed step-by-step description of a chemical process in which reactants are converted into products.
It consists of a sequence of bond-making and bond-breaking steps involving the movement of electrons and provides a rationalization for chemical reactions. Above all, following a few basic principles allows one to predict the likely outcome of a reaction.
On the other hand, it must be appreciated that there will be times when it can be rather difficult to actually ‘prove’ the proposed mechanism, and in such instances, we are suggesting a reasonable mechanism consistent with experimental data.
The basic layout of this book classifies chemical reactions according to the type of reaction mechanism involved, not by the reactions undergone within any specific group of compounds. As we proceed, we shall meet several types of general reaction mechanisms.
Initially, however, reactions can be classified as ionic or radical, according to whether bond-making and bond-breaking processes involve two electrons or one electron respectively.
Ionic Reactions
As the name implies, ionic reactions involve the participation of charged entities, i.e. ions. Bond-making and bond-breaking processes in ionic reactions are indicated by curly arrows that represent the movement of two electrons.
The tail of the arrow indicates where the electrons are coming from, the arrowhead where they are going.
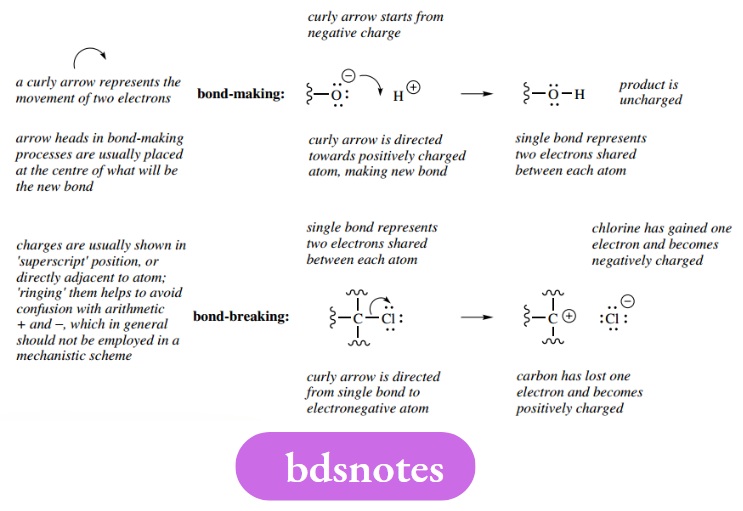
Lone pairs, originally nonbonding electrons, can also be used in bond-making processes.
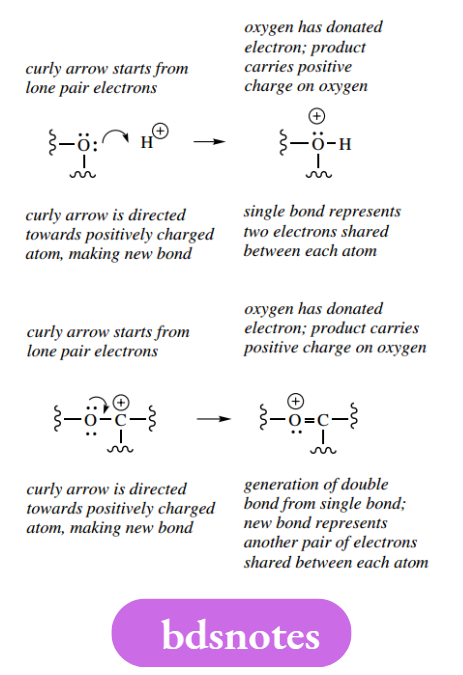
These simple examples illustrate the basic rules for mechanism and the use of curly arrows. The concepts are no different from those we have elaborated for drawing resonance structures:
- Curly arrows must start from an electron-rich species. This can be a negative charge, a lone pair, or a bond.
- Arrowheads must be directed towards an electron-deficient species. This can be a positive charge, the positive end of a polarized bond, or a suitable atom capable of accepting electrons, for example, an electronegative atom or Lewis acid.
If we are to draw sensible mechanisms, putting in the correct number of bonds and assigning the correct charges, then we must know the number of electrons around any particular atom.
We have already considered how to assess the formal charge on an atom; the following resume´ e´ covers those occasions that we are most likely to meet.
Carbon has four bonding electrons and can attain a stable octet of electrons by bonding to four other atoms, i.e. it has a valency of four.
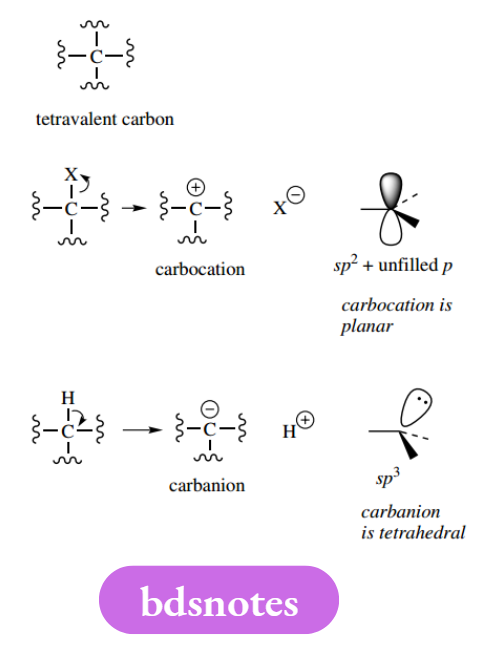
Carbon can also bond to just three other atoms by donating a pair of electrons from the octet to one of the atoms originally bonded, in so doing breaking the bond.
It will then carry a positive charge; it has effectively donated its single electron contribution from the shared pair comprising the single bond. This positively charged carbon is called a carbocation (in older nomenclature a carbonium ion).
Note that, with only six electrons involved in bonding, the carbocation is a planar entity, having two electrons in each of three sp² orbitals and with an unfilled p orbital.
Alternatively, carbon can carry a negative charge if it accepts both electrons from one of the original bonds, leaving the other group electron-deficient and positively charged.
It has effectively gained a single electron and is termed a carbanion. In this case, carbon carries a full octet of electrons and is tetrahedral, as if it had four single bonds.
The lone pair of electrons occupy the fourth sp³ orbital. Remember, carbon cannot form more than four bonds! Nitrogen has three bonding electrons and a lone pair; it can bond to three atoms, i.e. it has a valency of three.
However, it can also bond to four atoms by donating its lone pair, in which case it will then carry a positive charge.
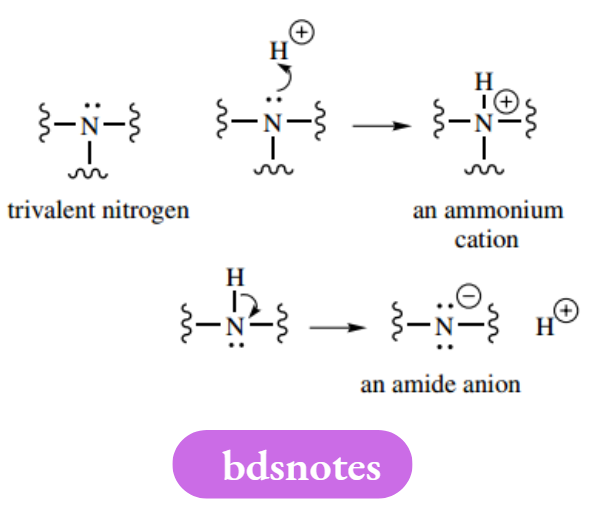

Nitrogen can also bond to just two atoms. Here, it carries a negative charge, since the octet is made up by acquiring one electron. Oxygen has two bonding electrons and two lone pairs. It can bond to two other atoms and is usually divalent.
It can also bond to one atom in a negatively charged form, or to three atoms in a positively charged form. The oxonium cation produced still carries a lone pair, but these electrons will not participate in further bonding, since this would necessitate an unfavourable double-charged oxygen.
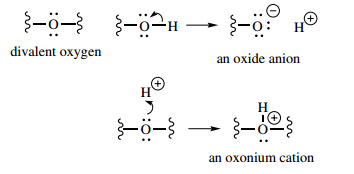
Hydrogen has one bonding electron and can bond to one other atom; it is monovalent. The electrons in this bond can be donated to hydrogen, giving the hydride anion, or can be donated to the other atom, generating a proton.

The proton thus contains no electrons. This seems a rather unnecessary statement, but it means a proton can only be an acceptor of electrons, and can never donate any. Curly arrows may be directed towards protons, but can never start from them! This would be a serious mechanistic error. Nevertheless, most students seem to make this error at some time or other.
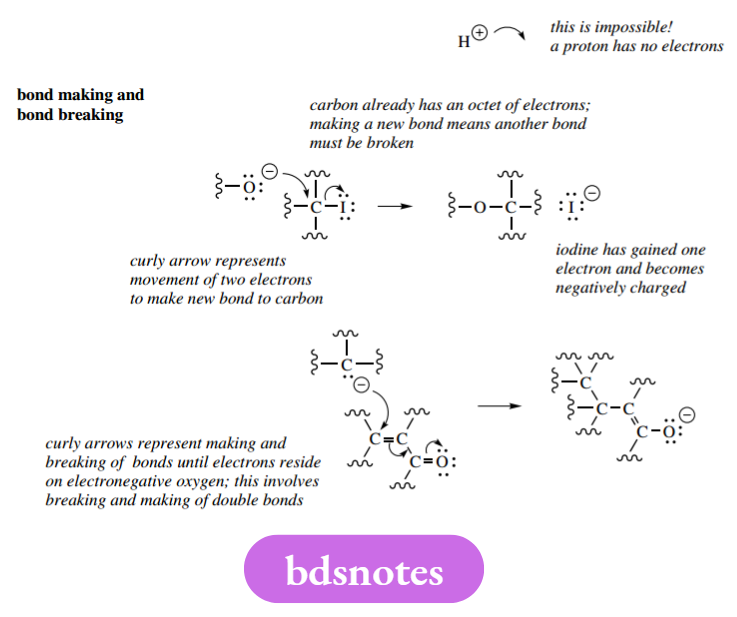
Counting the number of electrons on a particular atom becomes even more important when mechanisms become a little more complex and involve the making and breaking of bonds at the same atom.
This is going to be routine at carbon atoms, and the statement above, that ‘carbon cannot form more than four bonds’, becomes an important guiding principle.
Any mechanism that adds electrons to a carbon atom that is already carrying its full octet of electrons will also require the breaking of a bond and the removal of the excess electrons. Initially, it is a good idea to show nonbonding electrons in a mechanism, so that the number of electrons can be assessed and the correct charges defined.
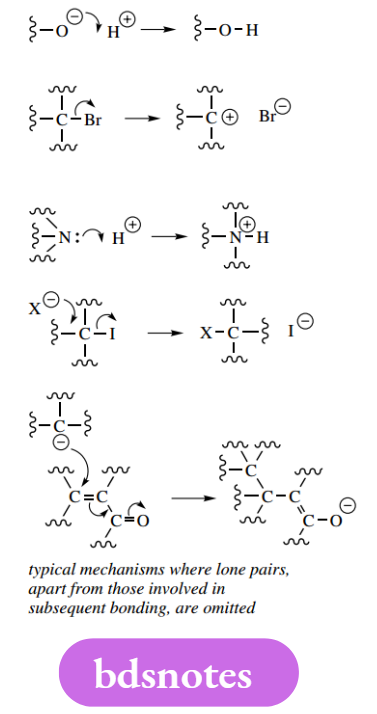
In due course, it is quicker to draw mechanisms without all the lone pairs, and it is normal practice to use representations showing just charges and only the lone pairs involved in subsequent bonding. The following mechanisms omit the lone pairs not involved in bonding but are perfectly acceptable.
Bond polarity
The concept of bond polarity has been discussed in some detail in Chapter 2 (see Section 2.7). Because different atomic nuclei have a particular ability to attract electrons, bonds between unlike atoms may not be shared equally.
This leads to a charge imbalance, with one of the atoms taking more than its share of the electrons. We refer to this as bond polarity.
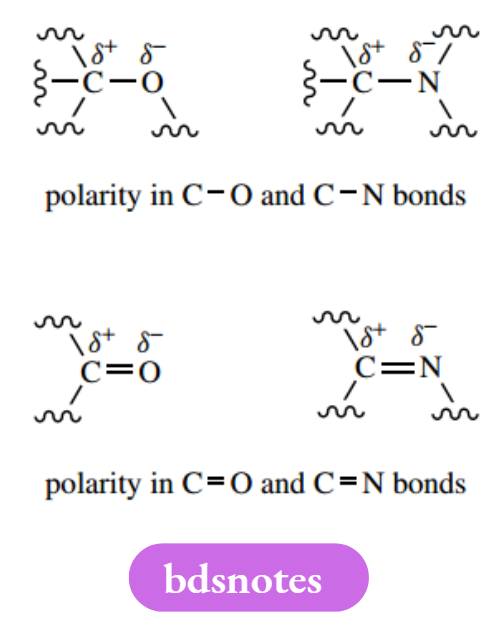
An atom that is more electronegative than carbon will thus polarize the bond, and we can consider the atoms as being partially charged.
This is indicated in a structure by putting partial charges ( δ+ and δ−) above the atoms. It can also be represented by putting an arrowhead on the bond in the direction of electron excess.
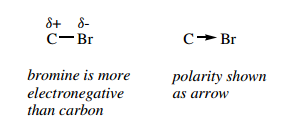
The relatively small difference in electronegativities between hydrogen and carbon means there is not going to be much polarity associated with a C–H bond. Most atoms other than hydrogen and carbon when bonded to carbon are going to be electron-rich and bonds may therefore display considerable polarity.
This is illustrated by carbon-oxygen and carbon-nitrogen single bonds. Double bonds show even greater polarity. This polarity helps us to predict chemical behaviour and is crucial to our prediction of chemical mechanisms.
Nucleophiles, Electrophile Les, And Leaving Groups
Reagents are classified as nucleophiles or electrophiles. Nucleophiles are electron-rich, nucleus-seeking reagents, and typically have a negative charge (anions) or a lone pair.
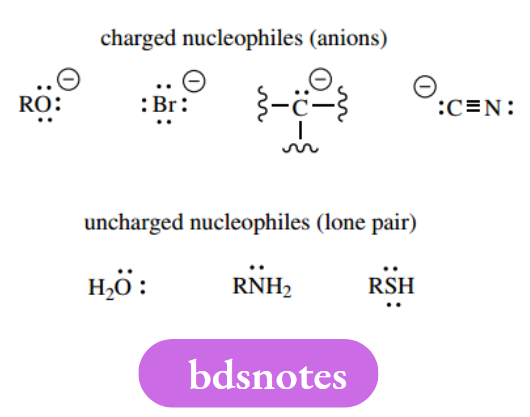
Compounds with multiple bonds, for example, alkenes, alkynes, and aromatics can also act as nucleophiles in so-called electrophilic reactions.
Electrophiles are electron-deficient, electron-seeking reagents, and typically have a positive charge (cations) or are polarizable molecules that can develop an electron-deficient centre.
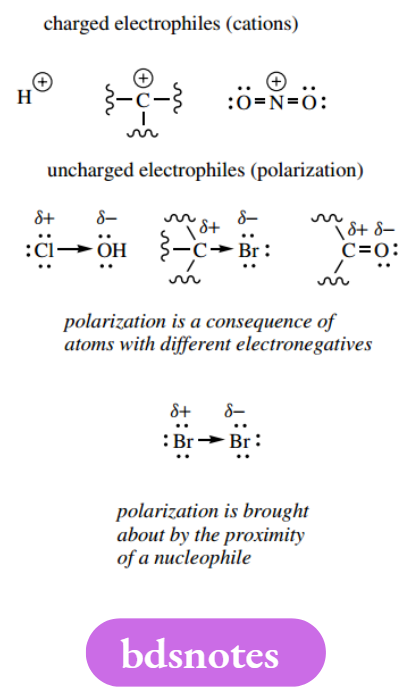
Many reactions will involve both nucleophiles and electrophiles. These may then be classified as nucleophilic if the main change to the substrate involves the attack of a nucleophile, or electrophilic if the principal change involves an attack of the substrate onto an electrophile. This distinction will become clearer in due course. The electron-rich species is always regarded as the attacking agent
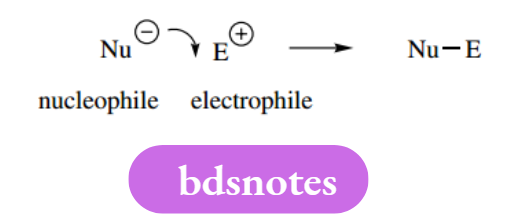
Leaving the group is the terminology used for ions or neutral molecules that are displaced from a reactant as part of a mechanistic sequence. Frequently, this displacement is the consequence of a nucleophile attacking an electrophile, and where the electrophile carries a suitable leaving group.
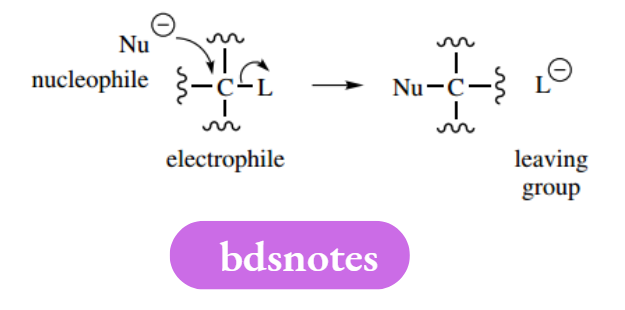
Good leaving groups are those that form stable ions or neutral molecules after they leave the substrate. We shall frequently need to write mechanisms involving general nucleophiles, electrophiles or leaving groups.
Standard abbreviations are Nu− or Nu: for a nucleophile (charged or uncharged), E+ for an electrophile, and L− or L: for a leaving group. In many instances, an electrophile containing a leaving group would simply be represented by C–L.
Radical Reactions
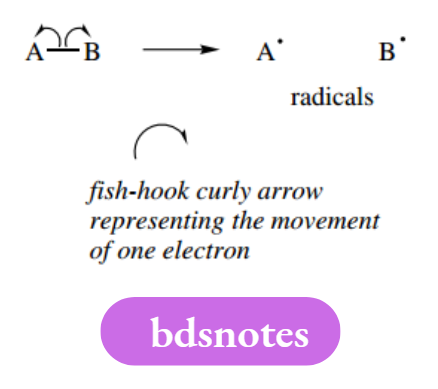
Radicals (sometimes termed free radicals) are uncharged high-energy species with an unpaired electron, and may contain one or more atoms:

For clarity, nonbonding electrons are usually omitted, though to propose meaningful mechanisms it is important to remember how many electrons are associated with each atom. The unpaired electron must always be shown.
In the formation of radicals, a bond is broken and each atom takes one electron from the pair constituting the bond. Bond-making and bond-breaking processes are indicated by single-headed (fishhook) curly arrows representing the movement of one electron.
A radical mechanism sequence requires three distinct types of process: initiation, propagation, and termination.
Initiation is the formation of two radical species by bond fission, whereas propagation involves the reaction of a radical with a neutral molecule, a process that leads to the generation of a new radical. Because radicals are so reactive, the propagation process may continue as long as reagent molecules are available.

Finally, the reaction is brought to a conclusion by the combination of two radical species, so that the unpaired electrons, one from each species, are combined into a new single bond. The radical pairing termination step is analogous to a reversal of the initiation step.
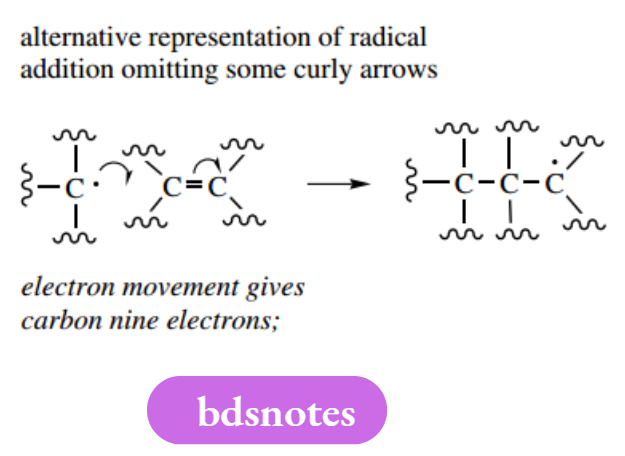
It occurs readily because of the reactivity of radicals; it follows, therefore, that the initiation step will require the input of a considerable amount of energy to dissociate the single bond. In the propagation steps shown above, the radical propagates a further radical by causing the fission of a single bond in the substrate. Many important radical reactions involve compounds with double bonds as substrates, and the π bond is cleaved during the radical addition reaction.
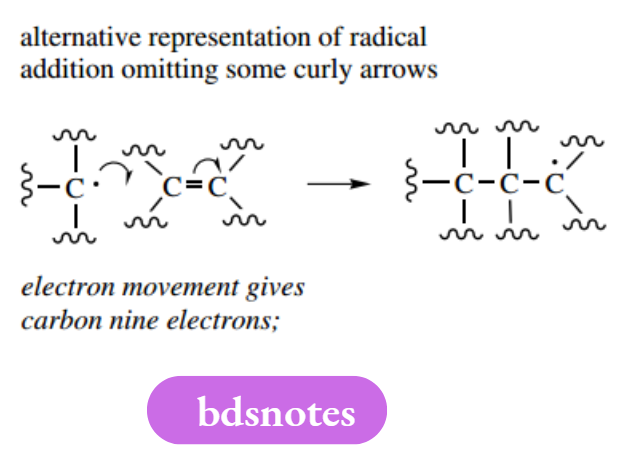
It makes good sense to draw free-radical mechanisms in the manner shown by these examples. However, shorter versions may be encountered in which not all of the arrows are drawn.
These versions bear considerable similarity to two-electron curly arrow mechanisms, in that a fishhook arrow is shown attacking an atom, and a second fishhook arrow is then shown leaving this atom. The other electron movement is not shown but is implicit.
This type of representation is quite clear if the complement of electrons around a particular atom is counted each time; but, if in any doubt, use all the necessary fishhook arrows.
Reaction Kinetics And Mechanism
One of how we can obtain information about a mechanistic sequence is to study the rate of reaction.
The dependence of the reaction rate on the concentration of reagents and other variables indicates the number and nature of the molecules involved in the rate-determining step of the reaction.
The rate-determining step is defined as the slowest transformation in the sequence, with all other transformations proceeding much faster than this. Consider a turnstile at a football match.
This limits the rate at which spectators enter the ground. How rapidly people walk towards the turnstile or away from it once they are in the ground cannot influence the rate at which they get through the turnstile.
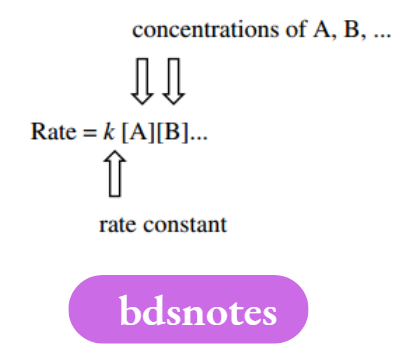
The rate of reaction is given by the equation in which k is the rate constant, and A, B, etc. are the variables on which the rate depends. Square brackets are used to indicate concentrations. It is rare for more than two variables to be involved, and often it is only one.
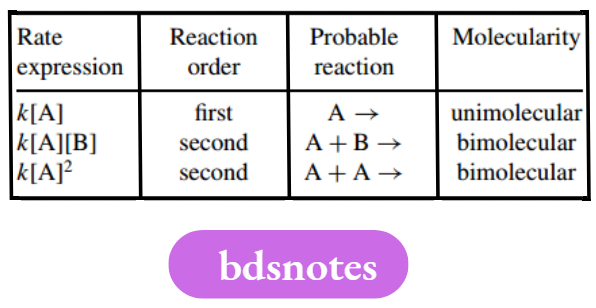
The most common types of rate expression. In first-order reactions, the rate expression depends upon the concentration of only one species, whereas second-order reactions show dependence upon two species, which may be the same or different.
The molecularity, or number of reactant molecules involved in the rate-determining step, is usually equivalent to the kinetic reaction order, though there can be exceptions.
For instance, a bimolecular reaction can appear to be first-order if there is no apparent dependence on the concentration of one of the reagents. Such a situation might occur when the solvent was also one of the reagents.
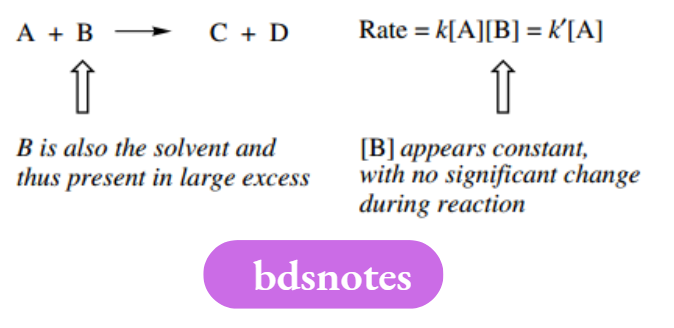
Despite occasional apparent anomalies such as this, the rate expression gives us valuable information about the likely reaction mechanism. If the reaction is unimolecular, the rate-determining step involves just one species, whereas the rate-determining step involves two species if it is bimolecular.
We can then deduce the probable reaction, and our proposed mechanism must reflect this information. The kinetic rate expressions will be considered further as we meet specific types of reactions.
Intermediates And Transition States
Any realistic mechanism will include several postulated structures, perhaps charged structures or radicals, which lie on the pathway leading from reactants to products. Some of these intervening structures are termed intermediates, and others transition states.
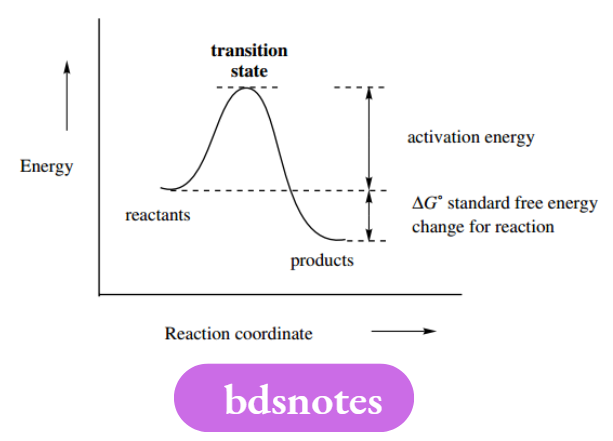
These are differentiated by their stability, and whether they can be detected by appropriate analytical methods. A diagram that follows the energy change during the reaction can illustrate their involvement.
The xcoordinate is usually termed the ‘reaction coordinate’, and in many cases equates to time, though the possibility that the reaction is reversible prevents us from showing this as a simple time coordinate.
In which reactants are converted into products. The difference between the energy of the reactants and products is called the standard free energy change for the reaction. As shown, the change in energy is negative, so that the reaction liberates energy and is potentially favourable.
It does not occur spontaneously, however, since the reactants need to acquire sufficient energy to collide and react.
This energy is termed the activation energy – even gunpowder needs a match to set off the explosion! The high-energy peak in the curve is termed the transition state or sometimes, activated complex. This material cannot be isolated, or even detected.
In an alternative scenario, again with a negative energy change, the energy profile may appear different, In this case, there is again an activation energy required to set the reaction off, but this energy maximum is then followed by an energy minimum.
The energy minimum represents an intermediate in the reaction pathway. It is converted into the products by overcoming a second activation energy, though this is likely to be considerably less than the first activation energy.
Because the intermediate is at an energy minimum, this material may be stable and can be isolated, or it may be reactive and short-lived, but detectable.
The two energy maxima represent different transition states. The energy diagrams shown here are merely generalized examples. We shall meet some specific examples as we consider various reaction mechanisms.
Types Of Reaction
At first glance, there appear to be an infinite number of different chemical reactions, all of which will have to be remembered.
A cursory look through any textbook of organic chemistry does little to dispel this fear. However, the beauty and strength of the mechanism is that it allows us to predict chemical behaviour without having to remember lots of chemical reactions.
A further reassuring fact is that virtually all of the chemical reactions can be classified according to a reaction type, and the number of distinct reaction types is rather few.
We only need to consider reaction types according to what is achieved in the conversion, namely substitution, elimination, addition, or rearrangement. In general terms, these may be represented as follows.
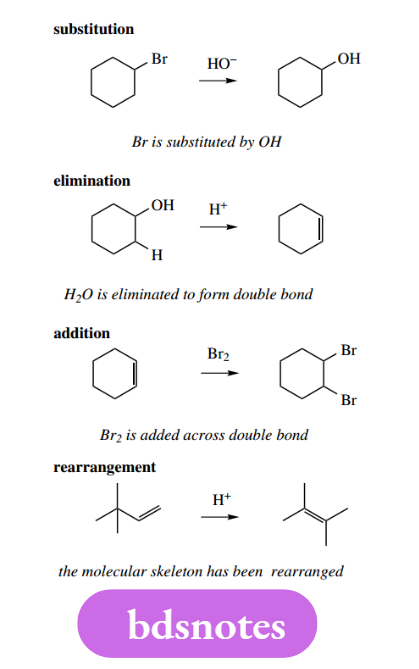
We will then subdivide these reaction types according to the type of reagent that brings about the change, to rationalize typical reactions further.
For example, addition reactions can be subdivided into nucleophilic addition, electrophilic addition, or radical addition.
Whilst this does increase the number of permutations, we shall see that it is necessary to do this, and it is also perfectly logical for our understanding of how reactions occur.
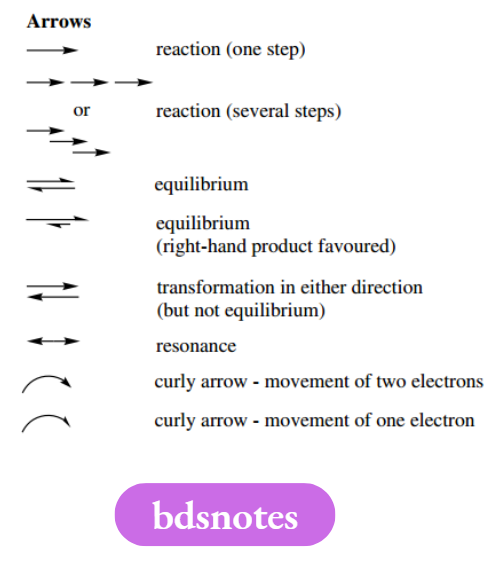
Arrows
We have now encountered several different types of arrows routinely used in chemistry to convey particular meanings. We have met curly arrows used in mechanisms, double-headed resonance arrows, equilibrium arrows, and the simple single arrows used for reactions.
This is a convenient point to bring together the different types and provide a checklist for future reference. We are also showing how additional information about a reaction may be presented with the arrow.

Some Common Mistakes In Drawing Mechanisms
Experience tells us that whilst many students find mechanisms easy and logical, others despair and are completely bewildered. We cannot guarantee success for all, but we hope that by showing a few of the common mistakes we may help some of the latter group join the former.
To make the examples chosen as real as possible, these have all been selected from students’ examination answers.
The mechanisms relate to reactions we have yet to meet, but this is not important. At this stage, it is the manipulation of curly arrows that is under consideration. You may wish to return to this section later.
Mistakes With Valencies
As electrons are moved around via curly arrows, it is imperative to remember how many electrons are associated with a particular atom, and not to exceed the number of bonds permitted.
The usual clanger is five-valent carbon, typically the result of making a new bond to a fully substituted carbon (four bonds, eight electrons) without breaking one of the old bonds. This is the case in the example shown.
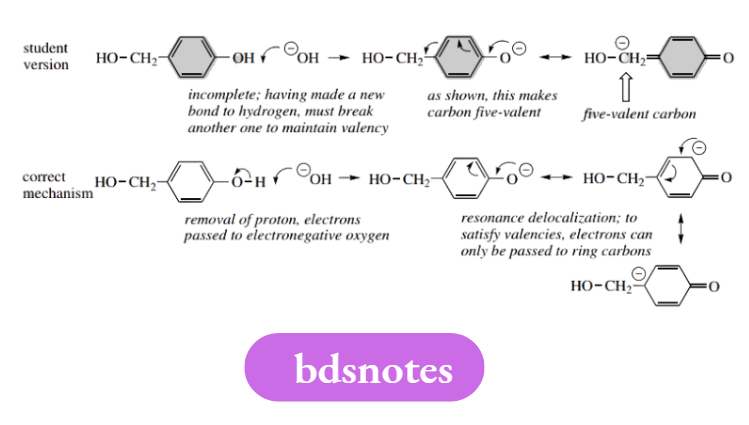
Mistake with formal charges
It is also important when counting electrons to assign any formal charge as necessary. It is all too common to see hydroxide presented with a lone pair but without any charge. Unfortunately, subsequent ionic reactions then just do not ‘balance’.
If one considers that hydroxide is derived by ionization of NaOH, or by loss of a proton from H2O, this problem should not arise.

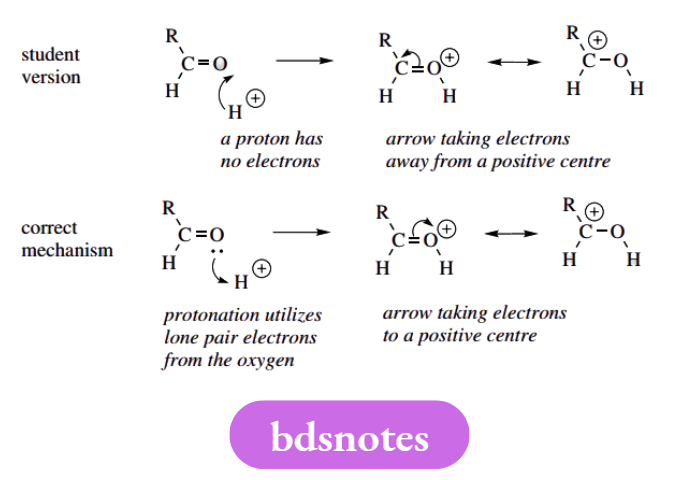
Arrows From Protons
Ask yourself how many electrons are there in a proton. We trust the answer is none, and you will thus realize that arrows representing the movement of electrons can never start from a proton.
It seems that this mistake is usually made because, if one thinks of protonation as the addition of a proton, it is tempting to show the proton being put on via an arrow. With curly arrows, we must always think in terms of electrons.
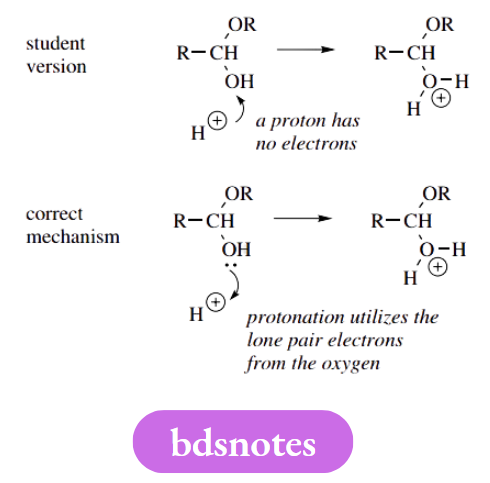
We were even less keen on the second example, where, in the resonance delocalization step, an arrow is shown taking electrons away from a positive charge and creating a new positive centre.
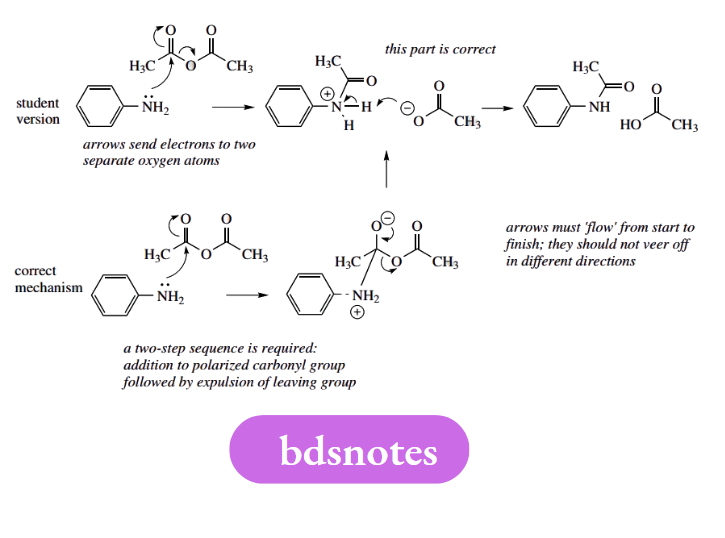
Vague arrows
Some mechanisms have arrows going in all sorts of directions. Arrows must ‘flow’ from start to finish; they should not veer off in different directions. Many of the arrows do not represent electron movements, and it would appear that, as a last resort, students have tried to memorize the mechanism rather than rationalizing it.
This is both dangerous and rather unnecessary. The logical approach gets the right answer, requires relatively little effort, and cuts out the need to learn the mechanism. Mechanisms should not be learnt; they should be deduced.
In this example, the student remembered that a series of curly arrows was required, and they are generally in the right places, but not coming from electron-rich species, and not flowing in the right direction. This is typical of trying to remember a mechanism, which then fails to obey the general rules.
Too many steps at once
It is tempting to draw a mechanism with a series of curly arrows leading to the product via the minimum number of structures. We can often use several curly arrows in the same structure, but only provided we do not destroy the rationale for the mechanism.
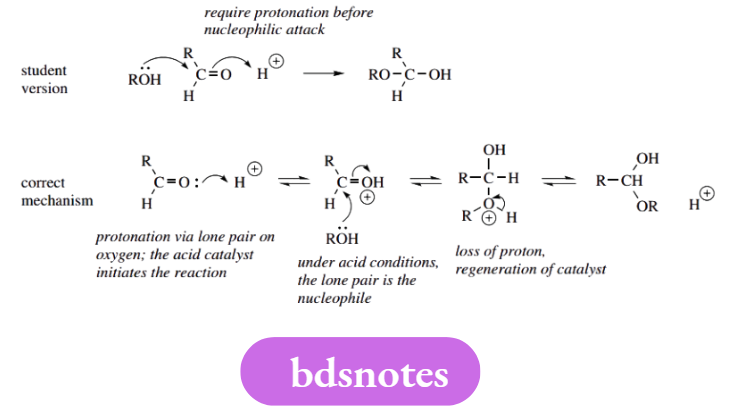
In the example shown, the two curly arrows suggest a concerted interaction of three entities. This is improbable and does not tell us why the reaction should take place. Using the longer sequence, we see that the acid catalyst activates the carbonyl group towards nucleophilic attack, and is later regenerated.
The second example also emphasizes that base is needed to generate the nucleophile, the charged phenoxide being a better nucleophile than the phenol.
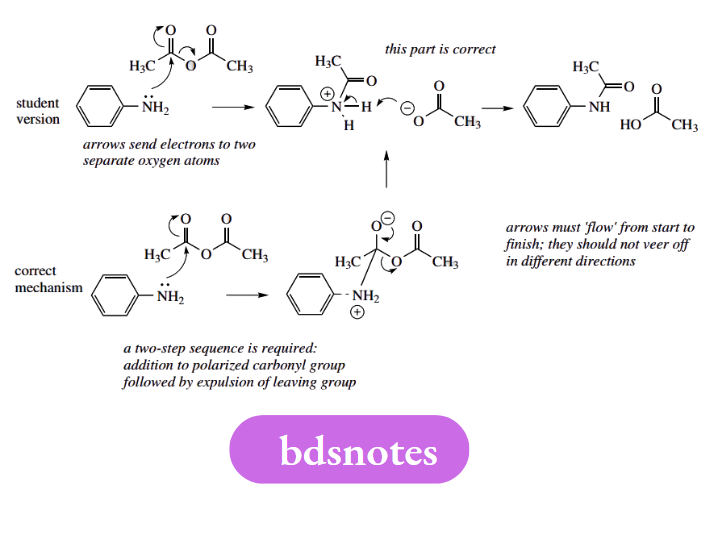
In the third example, arrows veer off in different directions, rather than flowing smoothly from start to finish. This mechanism is wrong in that an intermediate has been omitted.
Unrealistic izations
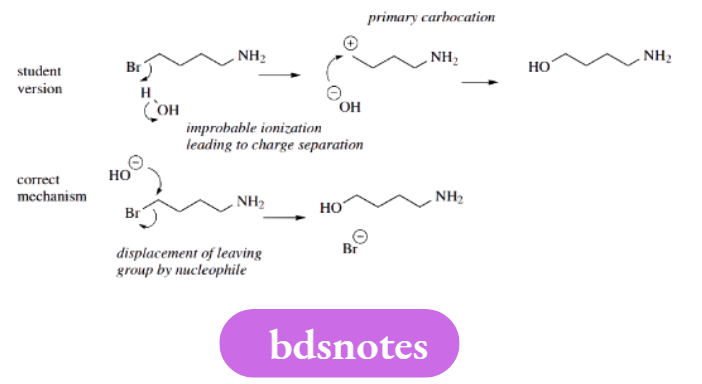
It is often necessary to ionize one of the reagents to initiate a reaction, and this requires careful consideration if the mechanism is to be realistic.
For example, we should not attempt to protonate substrates under basic conditions, and we are unlikely to generate anionic species under acidic conditions. These are fairly obvious limitations but are frequent mistakes.
Some thought about relative acidity and basicity is also sensible; ionization of alcohols does not occur without a strong base, as suggested in the example.
Primary Carbocations
Should you wish to use carbocations in a reaction mechanism, you must consider the relative stability of these entities. Tertiary carbocations are OK, and in many cases so are secondary carbocations.
Primary carbocations are just not stable enough, unless there is the added effect of resonance, as in benzylic or allylic systems.
Arrows curled the wrong way Can arrows curl the wrong way? Yes, they can, as the example shows. You should always understand that the arrowhead is depositing electrons between the start of the arrow and the atom it is approaching so that the new bond is formed at the inside of the curl.
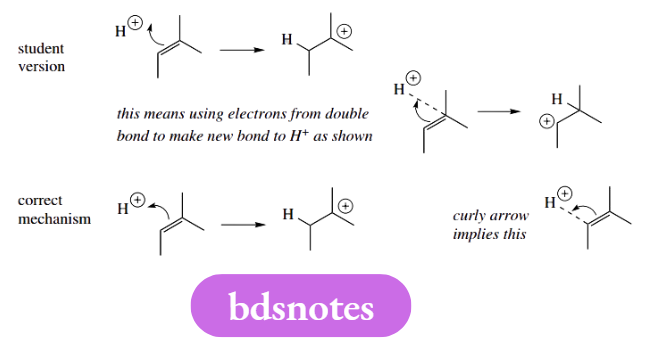
The electrophilic addition to alkenes is one of the occasions when the direction of the curl matters and can convey the formation of different products. Although the product shown is correct, the curly arrow is wrong.
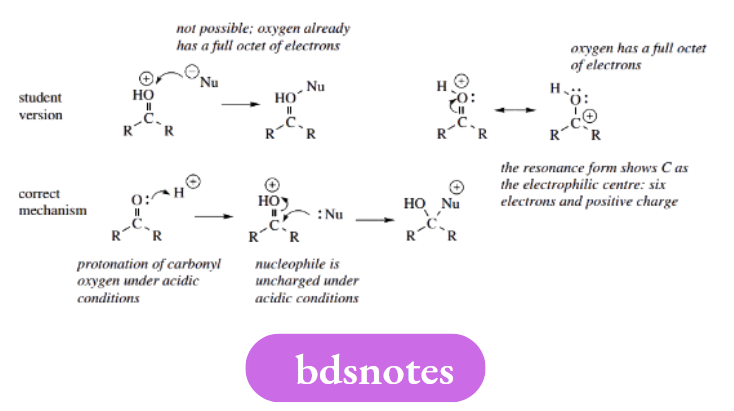
Making bonds to O+ or N+
It is tempting to consider O+ and N+ as electron-deficient species and, therefore, open to attack by nucleophiles. Here, we must count electrons to appreciate the true nature of these charged systems.
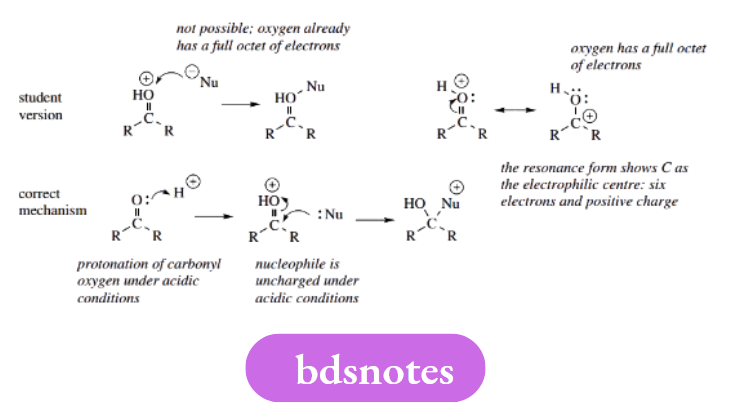
Both O+ bonded to three atoms and N+ bonded to four atoms are isoelectronic with tetravalent carbon, in other words, they have a full octet of electrons. Despite the positive charge, these atoms are not electron-deficient and are unable to make a new bond with the electron-rich nucleophiles.
Leave a Reply