Regulation Of Potassium Balance
“What is the regulation of potassium balance?”
Potassium, which is one of the most abundant cations in the body, is critical for many cell functions, including cell volume regulation, intracellular pH regulation, DNA and protein synthesis, growth, enzyme function, resting membrane potential, and cardiac and neuromuscular activity. Despite wide fluctuations in dietary K+ intake, [K+] in cells and extracellular fluid (ECF) remains remarkably constant. Two sets of regulatory mechanisms safeguard K+ homeostasis. First, several mechanisms regulate the [K+] in the ECF. Second, other mechanisms maintain the amount of K+ in the body constant by adjusting renal K+ excretion to match dietary K+ intake. The kidneys regulate K+ excretion.
Overview Of K+ Homeostasis
Total body K+ is 50 mEq/kg of body weight or 3500 mEq for a person weighing 70 kg. A total of 98% of the K+ in the body is located within cells, where its average [K+] is 150 mEq/L. A high intracellular [K+] is required for many cell functions, including cell growth and division and volume regulation. Only 2% of the total body K+ is located in the ECF, where its normal concentration is approximately 4 mEq/L. [K+] in the ECF that exceeds 5.0 mEq/L constitutes hyperkalemia. Conversely, [K+] in the ECF of less than 3.5 mEq/L constitutes hypokalemia. Hypokalemia is one of the most common electrolyte disorders in clinical practice and can be observed as
“Understanding potassium homeostasis in the body”

“How is potassium balance regulated in the human body?”
The effects of variations in plasma K+ concentration on the resting membrane potential of skeletal muscle. Hyperkalemia causes the membrane potential to become less negative and decreases the excitability by inactivating fast Na+ channels, which are responsible for the depolarizing phase of the action potential.
- Hypokalemia hyperpolarizes the membrane potential and thereby reduces excitability because a larger stimulus is required to depolarize the membrane potential to the threshold potential. Resting indicates the “normal” resting membrane potential. Normal threshold indicates the membrane threshold potential.
- As many as 20% of hospitalized patients. The most common causes of hypokalemia include administration of diuretic drugs (see Chapter 10), surreptitious vomiting (i.e., bulimia), and severe diarrhea. Gitelman syndrome (a genetic defect in the Na+-Cl− symporter in the apical membrane of distal tubule cells) also causes hypokalemia (see Chapter 4, Table 4-3).
- Hyperkalemia also is a common electrolyte disorder and is seen in 1% to 10% of hospitalized patients.
- Hyperkalemia often is seen in patients with renal failure, in persons taking drugs such as angiotensin-converting enzyme inhibitors and K+– sparing diuretics (see Chapter 10), in persons with hyperglycemia (i.e., high blood sugar), and the elderly. Pseudohyperkalemia, a falsely high plasma [K+], is caused by traumatic lysis of red blood cells while blood is being drawn.
- Red blood cells, like all cells, contain K+, and the lysis of red blood cells releases K+ into the plasma, artificially elevating the plasma [K+]. The large concentration difference of K+ across cell membranes (approximately 146 mEq/L) is maintained by the operation of sodium-potassium–adenosine triphosphatase (Na+-K+-ATPase). This K+ gradient is important in maintaining the potential difference across cell membranes.
- Thus K+ is critical for the excitability of nerve and muscle cells and for the contractility of cardiac, skeletal, and smooth muscle cells.
- Cardiac arrhythmias are produced by both hypokalemia and hyperkalemia. The electrocardiogram (ECG; monitors the electrical activity of the heart and is a quick and easy way to determine whether changes in plasma [K+] influence the heart and other excitable cells. In contrast, measurements of the plasma [K+] by the clinical laboratory require a blood sample, and values often are not immediately available.
“Importance of potassium balance for overall health”
The first sign of hyperkalemia is the appearance of tall, thin T waves on the ECG. Further increases in the plasma [K+] prolong the PR interval, depress the ST segment, and lengthen the QRS interval on the ECG. Finally, as the plasma [K+] approaches 10 mEq/L, the P wave disappears, the QRS interval broadens, the ECG appears as a sine wave, and the ventricles fibrillate (i.e., manifest rapid, uncoordinated contractions of muscle fibers). Hypokalemia prolongs the QT interval, inverts the T wave, and lowers the ST segment on the ECG.
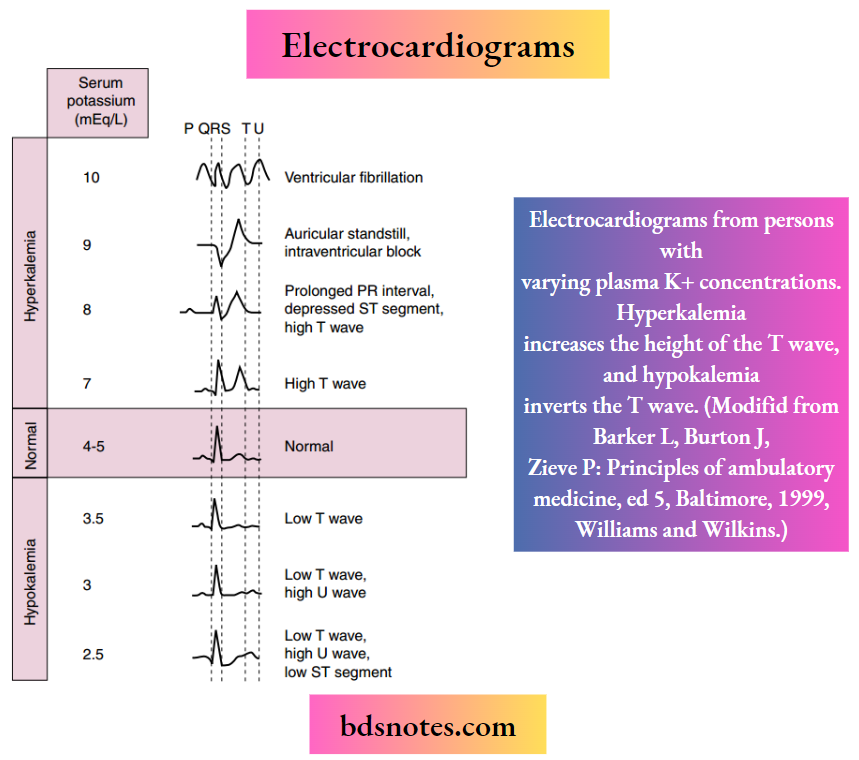
“Common causes of potassium imbalance explained”
Electrocardiograms from persons with varying plasma K+ concentrations. Hyperkalemia increases the height of the T wave, and hypokalemia inverts the T wave. (Modified from Barker L, Burton J, Zieve P: Principles of ambulatory medicine, ed 5, Baltimore, 1999, Williams & Wilkins.)
After a meal, the K+ absorbed by the gastrointestinal tract enters the ECF within minutes (Figure 7-3). If the K+ ingested during a normal meal ( ≈33 mEq) were to remain in the ECF compartment (14 L), the plasma [K+] would increase by a potentially lethal 2.4 mEq/L (33 mEq added to 14 L of ECF):
33 mEq/14 L = 2.4 mEq/L (7-1)
This rise in the plasma [K+] is prevented by the rapid uptake (within minutes) of K+ into cells. Because the excretion of K+ by the kidneys after a meal is relatively slow (within hours), the uptake of K+ by cells is essential to prevent life-threatening hyperkalemia. Maintaining total body K+ constant requires all the K+ absorbed by the gastrointestinal tract to eventually be excreted by the kidneys. This process requires about 6 hours.
“Role of the kidneys in potassium regulation”
Regulation Of Plasma [K+]
As illustrated in Figure 7-3 and Box 7-1, several hormones, including epinephrine, insulin, and aldosterone, increase K+ uptake into skeletal muscle, liver, bone, and red blood cells by stimulating Na+-K+- ATPase, the Na+-K+-2Cl− symporter, and the Na+- Cl− symporter in these cells. Acute stimulation of K+ uptake (i.e., within minutes) is mediated by an increased turnover rate of existing Na+-K+-ATPase, Na+-K+-2Cl−, and Na+-Cl− transporters, whereas the chronic increase in K+ uptake (i.e., within hours to days) is mediated by an increase in the quantity of Na+-K+-ATPase. A rise in the plasma [K+] that follows K+ absorption by the gastrointestinal tract stimulates insulin secretion from the pancreas, aldosterone release from the adrenal cortex, and epinephrine secretion from the adrenal medulla.

“How aldosterone affects potassium balance”
Overview of potassium homeostasis. An increase in plasma insulin, β-adrenergic agonists, or aldosterone stimulates K+ movement into cells and decreases plasma K+ concentration ([K+]), whereas a decrease in the plasma concentration of these hormones moves K+ into cells and increases plasma [K+]. α-Adrenergic agonists have the opposite effect.
- The amount of K+ in the body is determined by the kidneys. A person is in K+ balance when dietary intake and urinary output (plus output by the gastrointestinal tract) are equal. The excretion of K+ by the kidneys is regulated by plasma [K+], aldosterone, and arginine vasopressin.
- In contrast, a decrease in the plasma [K+] inhibits the release of these hormones. Whereas insulin and epinephrine act within a few minutes, aldosterone requires about 1 hour to stimulate K+ uptake into cells.
Epinephrine
Catecholamines affect the distribution of K+ across cell membranes by activating α- and β2-adrenergic receptors. The stimulation of α-adrenoceptors releases K+ from cells, especially in the liver, whereas the stimulation of β2-adrenoceptors promotes K+ uptake by cells. For example, the activation of β2-adrenoceptors after exercise is important in preventing hyperkalemia. The rise in plasma [K+] after a K+-rich meal is greater if the patient has been pretreated with propranolol, a β2– adrenoceptor antagonist. Furthermore, the release of epinephrine during stress (e.g., myocardial ischemia) can lower the plasma [K+] rapidly.
“Impact of insulin on potassium homeostasis”
Insulin
Insulin also stimulates K+ uptake into cells. The importance of insulin is illustrated by two observations. First, the rise in plasma [K+] after a K+-rich meal is greater in patients with diabetes mellitus (i.e., insulin deficiency) than in healthy people. Second, insulin (and glucose to prevent insulin-induced hypoglycemia) can be infused to correct hyperkalemia. Insulin is the most important hormone that shifts K+ into cells after the ingestion of K+ in a meal.
Aldosterone
Aldosterone, like catecholamines and insulin, also promotes K+ uptake into cells. A rise in aldosterone levels (e.g., primary aldosteronism) causes hypokalemia, whereas a fall in aldosterone levels (e.g., in persons with Addison disease) causes hyperkalemia. As discussed later, aldosterone also stimulates urinary K+ excretion. Thus aldosterone alters the plasma [K+] by
Major Factors, Hormones, And Drugs Influencing The Distribution Of K+ Between The Intracellular And Extracellular Fluid Compartments
Physiologic: Keep Plasma [K+] Constant
- Adrenergic receptor agonists
- Insulin
- Aldosterone
Pathophysiologic: Displace Plasma [K+] From Normal
- Acid-base disorders
- Plasma osmolality
- Cell lysis
- Vigorous exercise
“Role of sodium-potassium pumps in potassium regulation”
Drugs That Induce Hyperkalemia
- Dietary potassium supplements
- Angiotensin-converting enzyme inhibitors
- K+-sparing diuretics (see Chapter 10)
- Heparin acts on K+ uptake into cells and by altering urinary
K+ excretion.
Alterations Of Plasma [K+]
Several factors can alter the plasma [K+] (see Box 7-1). These factors are not involved in the regulation of the plasma [K+] but rather alter the movement of K+ between the intracellular fluid and ECF and thus cause the development of hypokalemia or hyperkalemia.
Acid-Base Balance
Metabolic acidosis increases the plasma [K+], whereas metabolic alkalosis decreases it. Respiratory alkalosis causes hypokalemia. Metabolic acidosis produced by the addition of inorganic acids (e.g., HCl and sulfuric acid) increases the plasma [K+] much more than an equivalent acidosis produced by the accumulation of organic acids (e.g., lactic acid, acetic acid, and keto acids).
- The reduced pH—that is, increased [H+]—promotes the movement of H+ into cells and the reciprocal movement of K+ out of cells to maintain electroneutrality.
- This effect of acidosis occurs in part because acidosis inhibits the transporters that accumulate K+ inside cells, including the Na+- K+-ATPase and the Na+-K+-2Cl− symporter. In addition, the movement of H+ into cells occurs as the cells buffer changes in the [H+] of the ECF. As H+ moves across the cell membranes, K+ moves in the opposite direction; thus cations are neither gained nor lost across cell membranes.
- Metabolic alkalosis has the opposite effect; the plasma [K+] decreases as K+ moves into cells and H+ exits. Although organic acids produce metabolic acidosis, they do not cause significant hyperkalemia. Two explanations have been suggested for the reduced ability of organic acids to cause hyperkalemia.
- First, the organic anion may enter the cell with H+ and thereby eliminate the need for K+/H+ exchange across the membrane. Second, organic anions may stimulate insulin secretion, which moves K+ into cells. This movement may counteract the direct effect of acidosis, which moves K+ out of cells.
“Biomechanics of potassium reabsorption and secretion”
Plasma Osmolality
The osmolality of the plasma also influences the distribution of K+ across cell membranes. An increase in the osmolality of the ECF enhances K+ release by cells and thus increases extracellular [K+]. The plasma [K+] may increase by 0.4 to 0.8 mEq/L for an elevation of 10 mOsm/kg H2O in plasma osmolality. In patients with diabetes mellitus who do not take insulin, plasma K+ often is elevated in part because of the lack of insulin and in part because of the increase in the concentration of glucose in plasma (i.e., from a normal value of ~100 mg/dL to as high as ~1200 mg/dL), which increases plasma osmolality. Hypoosmolality has the opposite action. The alterations in plasma [K+] associated with changes in osmolality are related to changes in cell volume. For example, as plasma osmolality increases, water leaves cells because of the osmotic gradient across the plasma membrane (see Chapter 1). Water leaves cells until the intracellular osmolality equals that of the ECF. This loss of water shrinks cells and causes the cell [K+] to rise. The rise in intracellular [K+] provides a driving force for the exit of K+ from cells. This sequence increases plasma [K+]. A fall in plasma osmolality has the opposite effect.
Cell Lysis
Cell lysis causes hyperkalemia, which results from the addition of intracellular K+ to the ECF. Severe trauma (e.g., burns) and some conditions such as tumor lysis syndrome (i.e., chemotherapy-induced destruction of tumor cells) and rhabdomyolysis (i.e., destruction of skeletal muscle) destroy cells and release K+ and other cell solutes into the ECF. In addition, gastric ulcers may cause the seepage of red blood cells into the gastrointestinal tract. The blood cells are digested, and the K+ released from the cells is absorbed and can cause hyperkalemia.
Exercise
During exercise, more K+ is released from skeletal muscle cells than during rest. The ensuing hyperkalemia depends on the degree of exercise. In people walking slowly, the plasma [K+] increases by 0.3 mEq/L. The plasma [K+] may increase by 2.0 mEq/L with vigorous exercise.
- Exercise-induced changes in the plasma [K+] usually do not produce symptoms and are reversed after several minutes of rest. However, vigorous exercise can lead to life-threatening hyperkalemia in persons
- who have endocrine disorders that affect the release of insulin, epinephrine (a β-adrenergic agonist), or aldosterone;
- whose ability to excrete K+ is impaired (e.g., because of renal failure); or
- who take certain medications, such as β2-adrenergic blockers. For example, during vigorous exercise, the plasma [K+] may increase by at least 2 to 4 mEq/L in persons who take β2-adrenergic receptor antagonists for hypertension.
“Mechanisms of potassium excretion by the kidneys”
Because acid-base balance, plasma osmolality, cell lysis, and exercise do not maintain the plasma [K+] at a normal value, they do not contribute to K+ homeostasis (see Box 7-1 ). The extent to which these pathophysiologic states alter the plasma [K+] depends on the integrity of the homeostatic mechanisms that regulate plasma [K+] (e.g., the secretion of epinephrine, insulin, and aldosterone).
K+ Excretion By The Kidneys
The kidneys play a major role in maintaining K+ balance. As illustrated in Figure 7-3, the kidneys excrete 90% to 95% of the K+ ingested in the diet. Excretion equals intake even when intake increases by as much as 10-fold.
- This balance of urinary excretion and dietary intake underscores the importance of the kidneys in maintaining K+ homeostasis. Although small amounts of K+ are lost each day in feces and sweat (approximately 5% to 10% of the K+ ingested in the diet), this amount is essentially constant (except during severe diarrhea), is not regulated, and therefore is relatively less important than the K+ excreted by the kidneys.
- K+ secretion from the blood into the tubular fluid by the cells of the distal tubule and collecting duct system is the key factor in determining urinary K+ excretion (Figure 7-4). Because K+ is not bound to plasma proteins, it is freely filtered by the glomerulus.
- When individuals ingest 100 mEq of K+ per day, urinary K+ excretion is about 15% of the amount filtered. Accordingly, K+ must be reabsorbed along the nephron. When dietary K+ intake increases, however, K+ excretion can, in extreme circumstances, exceed the amount filtered. Thus K+ also can be secreted.
- The proximal tubule reabsorbs about 67% of the filtered K+ under most conditions. Approximately 20% of the filtered K+ is reabsorbed by the loop of Henle, and, as with the proximal tubule, the amount reabsorbed is a constant fraction of the amount filtered.
- In contrast to these nephron segments, which can only reabsorb K+, the distal tubule and collecting duct can reabsorb or secrete K+.
- The rate of K+ reabsorption or secretion by the distal tubule and collecting duct depends on a variety of hormones and factors. When ingesting 100 mEq/day of K+ K+ is secreted by these nephron segments. A rise in dietary K+ intake increases K+ secretion. K+ secretion can increase the amount of K+ that appears in the urine so that it approaches 80% of the amount filtered
In contrast, a low-K+ diet activates K+ reabsorption along the distal tubule and collecting duct so that urinary excretion falls to about 1% of the K+ filtered by the glomerulus (see Figure 7-4). Because the kidneys cannot reduce K+ excretion to the same low levels as they can for Na+ (i.e., 0.2%), hypokalemia can develop
“Role of aldosterone in potassium excretion”

K+ Transport Along The Nephron. K+ Excretion Depends On The Rate And Direction Of K+ Transport By The Distal Tubule And Collecting Duct. Percentages Refer To The Amount Of Fitered K+ Reabsorbed Or Secreted By Each Nephron Segment. Left, Dietary K+ Depletion. An Amount Of K+ Equal To 1% Of The Fitered Load Of K+ Is Excreted. Right, Normal, And Increased Dietary K+ Intake. An Amount Of K+ Equal To 15% To 80% Of The Fitered Load Is Excreted.
“How does the renin-angiotensin-aldosterone system regulate potassium?”
- Ccd, Cortical Collecting Duct; Dt, Distal Tubule; Imcd, Inner Medullary Collecting Duct; Pt, Proximal Tubule; Tal, Thick Ascending Limb in persons who have a K+-deficient diet. Because the magnitude and direction of K+ transport by the distal tubule and collecting duct are variable, the overall rate of urinary K+ excretion is determined by these tubular segments.
- In persons with advanced renal disease, the kidneys are unable to eliminate K+ from the body, and thus the plasma [K+] rises.
- The resulting hyperkalemia reduces the resting membrane potential (i.e., the voltage becomes less negative), which decreases the excitability of neurons, cardiac cells, and muscle cells by inactivating fast Na+ channels, which are critical for the depolarization phase of the action potential.
- Severe, rapid increases in the plasma [K+] can lead to cardiac arrest and death. In contrast, in patients taking diuretic drugs for hypertension, urinary K+ excretion often exceeds dietary Kv intake. Accordingly, the K+ balance is negative, and hypokalemia develops.
- This decline in the extracellular [K+] hyperpolarizes the resting cell membrane (i.e., the voltage becomes more negative) and reduces the excitability of neurons, cardiac cells, and muscle cells.
- Severe hypokalemia can lead to paralysis, cardiac arrhythmias, and death. Hypokalemia also can impair the ability of the kidneys to concentrate the urine and can stimulate the renal production of ammonium, which affects acid-base balance.
- Therefore the maintenance of a high intracellular [K+], a low extracellular [K+], and a high K+ concentration gradient across cell membranes is essential for several cellular functions.

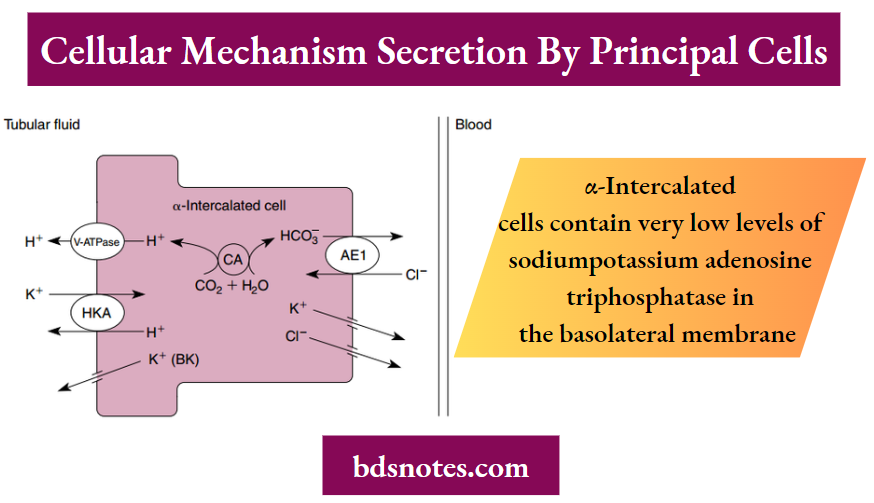
“Impact of antidiuretic hormone (ADH) on potassium balance”
Cellular Mechanism Of K+ Secretion By Principal Cells (A) And Α-Intercalated Cells (B) In The Distal Tubule And Collecting Duct.
- Α-Intercalated Cells Contain Very Low Levels Of Sodiumpotassium Adenosine Triphosphatase In The Basolateral Membrane (Not Shown).
- K+ Depletion Increases K+ Reabsorption By Α-Intercalated Cells By Stimulating H +-K+- Adenosine Triphosphatase (Hka). Ae1, Anion Exchanger 1; Atp, Adenosine Triphosphate; Bk, Ca++-Activated K+; Ca, Carbonic Anhydrase; Hco−3 , Bicarbonate; Kcc1, K+-Cl− Symporter 1; Romk, Renal Outer Medullary K+; V-Atpase, Vacuolar Adenosine Triphosphatase.
Cellular Mechanisms Of K+ Transport By Principal Cells And Intercalated Cells In The Distal Tubule And Collecting Duct
A illustrates the cellular mechanism of K+ secretion by principal cells in the distal tubule and collecting duct. Secretion from the blood into the tubule lumen is a two-step process: (1) K+ uptake from the blood across the basolateral membrane by Na+-K+- ATPase and (2) diffusion of K+ from the cell into the tubular fluid through K+ channels (the renal outer medullary K+ channel and the Ca++-activated K+ [BK] channel).
- A K+-Cl− symporter in the apical plasma membrane also secretes K+. Na+-K+-ATPase creates a high intracellular [K+], which provides the chemical driving force for K+exit across the apical membrane through K+ channels. Although K+ channels also are present in the basolateral membrane, K+ preferentially leaves the cell across the apical membrane and enters the tubular fluid. K+ transport follows this route for two reasons.
- First, the electrochemical gradient of K+ across the apical membrane favors its downhill movement into the tubular fluid. Second, the permeability of the apical membrane to K+ is greater than that of the basolateral membrane.
- Therefore K+ preferentially diffuses across the apical membrane into the tubular fluid. K+ secretion across the apical membrane via the K+-Cl− symporter is driven by the favorable concentration gradient of K+ between the cell and tubular fluid.
- The three major factors that control the rate of K+ secretion by the distal tubule and the collecting duct are:
- The activity of Na+-K++-ATPase
- The driving force (electrochemical gradient for the K+ channel and the chemical concentration gradient for the K+-Cl− symporter) for K+ movement across the apical membrane
- The permeability of the apical membrane to K+
“Steps in renal potassium handling explained”
Every change in K+ secretion by principal cells results from an alteration in one or more of these factors.
α-Intercalated cells reabsorb K+ by an H+-K+- ATPase transport mechanism located in the apical membrane (see Figure 7-5, B, and Chapter 4).
- This transporter mediates K+ uptake across the apical plasma membrane in exchange for H+. K+ exit from intercalated cells into the blood is mediated by a K+ channel.
- The reabsorption of K+ is activated by a low K+ diet. Intercalated cells also express the Ca++- activated, BK channels in the apical plasma membrane.
- K+ secretion by BK channels in intercalated cells (most likely α-intercalated cells) is activated by increased tubule flow rate, which enhances Ca++ uptake across the apical plasma membrane by activating a transient receptor potential channel also located in the apical plasma membrane (not shown in Figure 7-5, B).
- Increased intracellular Ca++ stimulates protein kinase C, which activates BK channels.
Regulation Of K+ Secretion By The Distal Tubule And Collecting Duct
The regulation of K+ excretion is achieved mainly by alterations in K+ secretion by principal cells of the distal tubule and collecting duct. Plasma [K+] and aldosterone are the major physiologic regulators of K+ secretion. Ingestion of a K+-rich meal also activates renal K+ excretion by a mechanism involving an unknown gut-dependent mechanism. Arginine vasopressin (AVP) also stimulates K+secretion; however, it is less important than the plasma [K+] and aldosterone.
Major Factors And Hormones Influencing K+ Excretion
Physiologic: Keep K+ Balance Constant
- Plasma [K+]
- Aldosterone
- Arginine vasopressin
Pathophysiologic: Displace K+ Balance
- The flow rate of tubule fluid
- Acid-base disorders
- Glucocorticoids
Other factors, including the flow rate of tubular fluid and acid-base balance, influence K+ secretion by the distal tubule and collecting duct. However, they are not homeostatic mechanisms because they disturb the K+ balance (Box 7-2).
“Causes and effects of hypokalemia (low potassium)”
Plasma [K+]
Plasma [K+] is an important determinant of K+ secretion by the distal tubule and collecting duct. Hyperkalemia (e.g., resulting from a high-K+ diet or from rhabdomyolysis) stimulates K+ secretion within minutes. Several mechanisms are involved.
- First, hyperkalemia stimulates Na+-K+- ATPase and thereby increases K+ uptake across the basolateral membrane. This uptake raises the intracellular [K+] and increases the electrochemical driving force for K+ exit across the apical membrane.
- Second, hyperkalemia also increases the permeability of the apical membrane to K+. Third, hyperkalemia stimulates aldosterone secretion by the adrenal cortex, which acts synergistically with the plasma [K+] to stimulate K+ secretion.
- Fourth, hyperkalemia also increases the flow rate of tubular fluid, which stimulates K+ secretion by the distal tubule and collecting duct.
- Hypokalemia (e.g., caused by a low-K+ diet or K+ loss in diarrhea) decreases K+ secretion by actions opposite to those described for hyperkalemia.
- Hence hypokalemia inhibits Na+-K+-ATPase, decreases the electrochemical driving force for K+ efflux across the apical membrane, reduces the permeability of the apical membrane to K+, and reduces plasma aldosterone levels.
“Symptoms and treatment of hyperkalemia (high potassium)”

The relationship between plasma K+ concentration ([K+]) and K+ secretion by the distal tubule and the cortical collecting duct.
- Chronic hypokalemia—that is, plasma K+ concentration ([K+]) <3.5 mEq/L—occurs most often in patients who receive diuretics for hypertension.
- Thus the excretion of K+ by the kidneys exceeds the dietary intake of K+. Hypokalemia also occurs in patients who vomit, have nasogastric suction, have diarrhea, abuse laxatives, or have hyperaldosteronism.
- Vomiting, nasogastric suction, diuretics, and diarrhea all can decrease the extracellular fluid volume, which in turn stimulates aldosterone secretion (see Chapter 6). Because aldosterone stimulates K+ excretion by the kidneys, its action contributes to the development of hypokalemia.
- Chronic hyperkalemia (plasma [K+] >5.0 mEq/L) occurs most frequently in persons with reduced urine flow, low plasma aldosterone levels, and renal disease in which the glomerular filtration rate falls below 20% of normal.
- In these persons, hyperkalemia occurs because the excretion of K+ by the kidneys is less than the dietary intake of K+. Less common causes for hyperkalemia occur in people with deficiencies of insulin, epinephrine, and aldosterone secretion or people with metabolic acidosis caused by inorganic acids.
“How do potassium imbalances affect heart function?”
Aldosterone
A chronic (i.e., 24 hours or more) elevation in the plasma aldosterone concentration enhances K+ secretion across principal cells in the distal tubule and collecting duct (Figure 7-7) by five mechanisms:
- Increasing the amount of Na+-K+-ATPase in the basolateral membrane;
- Increasing the expression of the sodium channel (ENaC) in the apical cell membrane;
- Elevating serum glucocorticoid stimulated kinase (Sgk1) levels, which also increases the expression of ENaC in the apical membrane and activates K+ channels;
- Stimulating channel activating protease 1 (CAP1, also called prostate), which directly activates ENaC; and
- Stimulating the permeability of the apical membrane to K+.
The cellular mechanisms by which aldosterone affects the expression and activity of Na+-K+-ATPase and ENaC (preceding actions 1 to 4) have been described (see Chapter 4). Aldosterone increases the apical membrane K+ permeability by increasing the number of K+ channels in the membrane.
“Role of potassium balance in managing hypertension”
- However, the cellular mechanisms involved in this response are not completely known. Increased expression of Na+- K+-ATPase facilitates K+ uptake across the basolateral membrane into cells and thereby elevates intracellular [K+].
- The increase in the number and activity of Na+ channels enhances Na+ entry into the cell from the tubule fluid, an effect that depolarizes the apical membrane voltage. The depolarization of the apical membrane and increased intracellular [Kv] enhance the electrochemical driving force for K+ secretion from the cell into the tubule fluid.
- Taken together, these actions increase the cell [K+] and enhance the driving force for K+ exit across the apical membrane. Aldosterone secretion is increased by hyperkalemia and by angiotensin II (after activation of the renin-angiotensin system).
- Aldosterone secretion is decreased by hypokalemia and natriuretic peptides released from the heart.
- Although an acute increase in aldosterone levels (i.e., within hours) enhances the activity of Na+-K+– ATPase, K+ excretion does not increase.
The reason for this phenomenon is related to the effect of aldosterone on Na+ reabsorption and tubular flow. Aldosterone stimulates Na+ reabsorption and water reabsorption and thus decreases tubular flow. The decrease in flow in turn decreases K+ secretion (discussed in more detail later in this chapter). However, chronic stimulation of
“Complications of untreated potassium imbalances”

The Relationship Between Plasma Aldosterone And K+ Secretion By The Distal Tubule And The Cortical Collecting Duct. Note That K+ Secretion Is Increased Further When The Plasma K+ Concentration ([K] P) Is Increased.
Na+ reabsorption expands the ECF and thereby returns tubular flow to normal. These actions allow the direct stimulatory effect of aldosterone on the distal tubule and collecting duct to enhance K+ excretion.
Arginine Vasopressin
Although AVP does not affect net urinary K+ excretion, this hormone does stimulate K+ secretion by the distal tubule and collecting duct (Figure 7-8). AVP increases the electrochemical driving force for K+ exit across the apical membrane of principal cells by stimulating Na+ uptake across the apical membrane of principal cells.
- The increased Na+ uptake reduces the electrochemical driving force for K+ exit across the apical membrane (i.e., the interior of the cell becomes less negatively charged). Despite this effect, AVP does not change K+ secretion by these nephron segments.
- The reason for this phenomenon is related to the effect of AVP on tubular fluid. AVP decreases tubular fluid flow by stimulating water reabsorption.
- The decrease in tubular flow in turn reduces K+ secretion (explained later in this chapter). The inhibitory effect of decreased flow of tubular fluid offsets the stimulatory effect of AVP on the electrochemical driving force for K+ exit across the apical membrane.
- If AVP did not increase the electrochemical driving force favoring K+ secretion, urinary K+ excretion would decrease as AVP levels increase and urinary flow rates decrease. Hence K+ balance would change in response to alterations in water balance.
- Thus the effects of AVP on the electrochemical driving force for K+ exit across the apical membrane and tubule flow enable urinary K+ excretion to be maintained constant despite wide fluctuations in water excretion.
“Pathophysiology of potassium imbalances explained”
Factors That Perturb K+ Excretion
Whereas plasma [K+], aldosterone, and AVP play important roles in regulating K+ balance, the factors and hormones discussed next perturb K+ balance
Flow of Tubular Fluid
A rise in the flow of tubular fluid (e.g., with diuretic treatment and ECF volume expansion) stimulates K+ secretion within minutes, whereas a fall (e.g., ECF
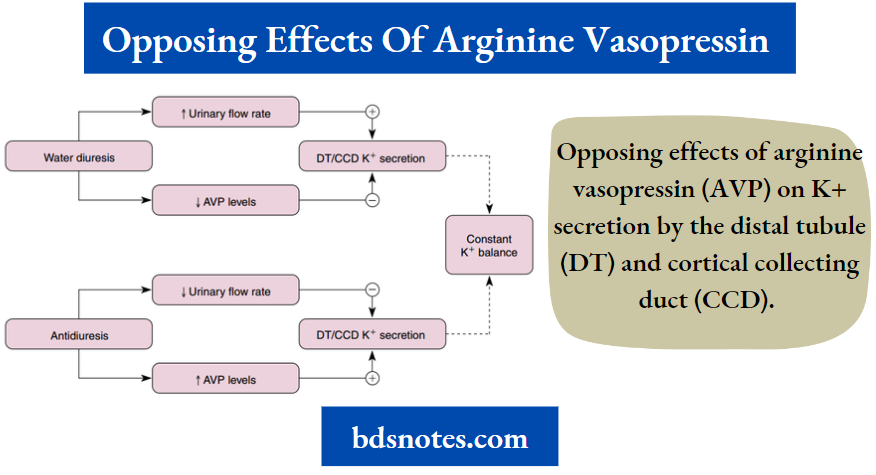
“Emerging research on potassium regulation mechanisms”
Opposing Effects Of Arginine Vasopressin (AVP) On K+ Secretion By The Distal Tubule (Dt) And Cortical Collecting Duct (Ccd). Secretion Is Stimulated By An Increase In The Electrochemical Gradient For K+ Across The Apical Membrane And By An Increase In The K+ Permeability Of The Apical Membrane. In Contrast, Secretion Is Reduced By A Fall In The Flow Rate Of Tubular Fluid. Because These Effects Oppose Each Other, Net K+ Secretion Is Not Affected By Avp.

“Case studies on potassium balance outcomes”
Relationship Between Tubular Flow Rate And K+ Secretion By The Distal Tubule And Cortical Collecting Duct. A Diet High In K+ Increases The Slope Of The Relationship Between Flw Rate And Secretion And Increases.
- The Maximum Rate Of Secretion. A Diet Low In K+ Has The Opposite Effects. The Shaded Bar Indicates The Flw Rate Under Most Physiologic Conditions.
- volume contraction caused by hemorrhage, severe vomiting, or diarrhea) reduces K+ secretion by the distal tubule and collecting duct. Increments in tubular fluid are more effective in stimulating K+ secretion as dietary K+ intake is increased. Studies of the primary cilium in principal cells have elucidated some of the mechanisms whereby increased flow stimulates K+ secretion.
- As described in Chapter 2, increased flow bends the primary cilium in principal cells, which activates the PKD1/PKD2 Ca++ conducting channel complex.
- This mechanism allows more Ca++ to enter principal cells and increases intracellular [Ca++]. The increase in [Ca++] activates BK channels in the apical plasma membrane, which enhances K+ secretion from the cell into the tubule fluid. Increased flow also activates BK-mediated K+ secretion by intercalated cells.
- Increased flow also may stimulate K+ secretion by other mechanisms. As flow increases, for example, following the administration of diuretics or as the result of an increase in the ECF volume, so does the Na+ concentration of tubule fluid.
- This increase in Na+ concentration ([Na+]) facilitates Na+ entry across the apical membrane of the distal tubule and collecting duct cells, thereby decreasing the interior negative membrane potential of the cell.
- This depolarization of the cell membrane potential increases the electrochemical driving force that promotes K+ secretion across the apical cell membrane into the tubule fluid. In addition, increased Na+ uptake into cells activates the Na+-K+-ATPase in the basolateral membrane, thereby increasing K+ uptake across the basolateral membrane and elevating cells [K+].
- However, it is important to note that an increase in flow rate during a water diuresis does not have a significant effect on K+ excretion (see Figure 7-9), most likely because, during a water diuresis, the [Na+] of tubule fluid does not increase as flow rises.
“Complications of ignoring potassium imbalances”
Acid-Base Balance
Another factor that modulates K+ secretion is the [H+] of the ECF (Figure 7-10). Acute alterations (within minutes to hours) in the pH of the plasma influence K+ secretion by the distal tubule and collecting duct. Alkalosis (i.e., a plasma pH above normal) increases K+ secretion, whereas acidosis (i.e., a plasma pH below normal) decreases it. An acute acidosis reduces K+ secretion by two mechanisms:
- It inhibits Na+-K+-ATPase and thereby reduces the cell [K+] and the electrochemical driving force for K+ exit across the apical membrane, and
- It reduces the permeability of the apical membrane to K+. Alkalosis has the opposite effect.
The effect of metabolic acidosis on K+ excretion is time-dependent. When metabolic acidosis lasts for several days, urinary K+ excretion is stimulated (Figure 7-11). This stimulation occurs because chronic metabolic acidosis decreases the reabsorption of water and solutes (e.g., sodium chloride [NaCl]) by the
- Renal outer medullary K+ (ROMK) (KCNJ1) channels in the apical membrane of principal cells mediate K+ secretion. Four ROMK subunits make up a single channel. Interestingly, the knockout of the KCNJ1 gene (ROMK) causes increased sodium chloride (NaCl) and K+ excretion by the kidneys, leading to reduced extracellular fluid volume and hypokalemia.
- Although this effect is somewhat perplexing, it should be noted that ROMK also is expressed in the apical membrane of the thick ascending limb of Henle’s loop, where it plays an important role in K+ recycling across the apical membrane, an effect that is critical for the operation of the Na+-K+-2Cl− symporter (see Chapter 4).
- In the absence of ROMK, NaCl reabsorption by the thick ascending limb is reduced, which leads to NaCl loss in the urine. Reduction of NaCl reabsorption by the thick ascending limb also reduces the lumen-positive transepithelial voltage, which is the driving force for K+ reabsorption by this nephron segment.
- Thus the reduction in paracellular K+ reabsorption by the thick ascending limb increases urinary K+ excretion, even when the cortical collecting duct is unable to secrete the normal amount of K+ because of a lack of ROMK channels.
- The cortical collecting duct, however, does secrete K+ even in ROMK knockout mice through the flow and Ca++-dependent BK channels and by the operation of a K+-Cl− symporter expressed in the apical membrane of principal cells.
“Advances in understanding potassium homeostasis”

Effect Of Plasma Ph On The Relationship Between Plasma K+ Concentration ([K+]) And K+ Secretion By The Distal Tubule And Collecting Duct.
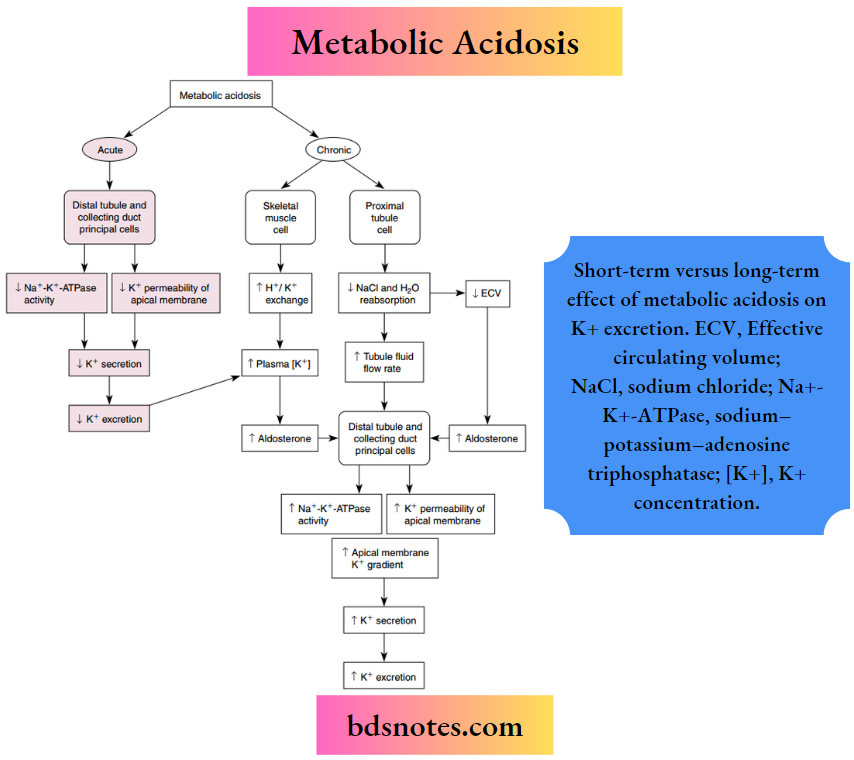
Short-term versus long-term effect of metabolic acidosis on K+ excretion. ECV, Effective circulating volume; NaCl, sodium chloride; Na+-K+-ATPase, sodium–potassium–adenosine triphosphatase; [K+], K+ concentration.
Proximal Tubule By Inhibiting Na+-K+-Atpase. Hence The Flw Of Tubular Flid Is Augmented Along The Distal Tubule And Collecting Duct. The Inhibition Of Proximal Tubular Water And Nacl Reabsorption Also Decreases The Ecf Volume And Thereby Stimulates Aldosterone Secretion.
- In Addition, Chronic Acidosis, Caused By Inorganic Acids, Increases Plasma [K+], Which Stimulates Aldosterone Secretion. The Rise In Tubular Flid Flw, Plasma [K+], And Aldosterone Levels Offsets The Effects Of Acidosis On The Cell [K+] And Apical Membrane Permeability, And K+ Secretion Rises.
- Thus Metabolic Acidosis May Either Inhibit Or Stimulate K+ Excretion, Depending On The Duration Of The Disturbance.
- The cellular mechanisms whereby changes in the K+ content of the diet and acid-base balance regulate K+ secretion by the distal tubule and collecting duct have been elucidated.
- Elevated K+ intake increases K+ secretion by several mechanisms, all related to increased serum K+ concentration. Hyperkalemia increases the activity of the renal outer medullary K+ (ROMK) channel in the apical plasma membrane of principal cells.
- Moreover, hyperkalemia inhibits proximal tubule sodium chloride (NaCl) and water reabsorption, thereby increasing distal tubule and collecting duct flow rate, a potent stimulus to K+ secretion.
- Hyperkalemia also enhances aldosterone concentration, which increases K+ secretion by three mechanisms. First, aldosterone increases the number of K+ channels in the apical plasma membrane. Second, aldosterone stimulates K+ uptake across the basolateral membrane by enhancing the number of Na+-K+-ATPase pumps, thereby enhancing the electrochemical gradient driving K+ secretion across the apical membrane.
- Third, aldosterone increases Na+ entry across the apical membrane, which depolarizes the apical plasma membrane voltage, thereby increasing the electrochemical gradient, and promoting K+ secretion. A low-K+ diet dramatically reduces K+ secretion by the distal tubule and collecting duct by increasing the activity of protein tyrosine kinase, which causes
- ROMK channels are to be removed from the apical plasma membrane, thereby reducing K+ secretion. Acidosis decreases K+ secretion by inhibiting the activity of ROMK channels, whereas alkalosis stimulates K+ secretion by enhancing ROMK channel activity.
“Global prevalence of potassium disorders”
As noted, acute metabolic alkalosis stimulates K+ excretion. Chronic metabolic alkalosis, especially in association with ECF volume contraction, significantly increases renal K+ excretion because of the associated increased levels of aldosterone.
Glucocorticoids
Glucocorticoids increase urinary K+ excretion. This effect is in part mediated by an increase in the glomerular filtration rate, which enhances urinary flow rate, a potent stimulus of K+ excretion, and by stimulating Sgk1 activity (discussed in a previous section).
- As discussed earlier, the rate of urinary K+ excretion is frequently determined by simultaneous changes in hormone levels, acid-base balance, or the flow rate of tubule fluid (Table 7-1).
- The powerful effect of flow often enhances or opposes the response of the distal tubule and collecting duct to hormones and changes in acid-base balance. This interaction can be beneficial in the case of hyperkalemia, in which the change in flow enhances K+ excretion and thereby facilitates K+ homeostasis.
- However, this interaction also can be detrimental, as in the case of alkalosis, in which changes in flow and acid-base status alter K+ homeostasis.
Table 7-1
Effects Of Hormones And Other Factors On K+ Secretion By The Distal Tubule And Collecting Duct And On Urinary K+ Excretion

Leave a Reply