Drugs Acting On Central Nervous System General Anaesthetics
General anaesthetics are CNS depressants which cause partial or complete loss of consciousness, sense or pain. The effect is reversible and generally used to produce unconsciousness during painful surgeries.
General anaesthetics bring about the descending depression of the CNS, starting with the cerebral cortex, the basal ganglia, the cerebellum and finally the spinal cord.
Stages of General Anaesthesia
The following are well-defined stages produced by increasing the blood concentration of the anaesthesia. They are:
- Stage 1 (Analgesia): This is the period from mild depression of higher neurons begins when from anaesthetic administration to the loss of consciousness. The patient progressively loses pain. This stage is also called the stage of analgesia (cortical stage) and is suitable for minor surgeries.
- Stage 2 (Delirium): In this, the stage extends from the loss of consciousness to irregular and specific breathing. Respiration may be normal after some time. There may be delirium, urination uncontrolled muscular movement, laughing, vomiting or struggle. Sometimes increases heart rate and blood pressure.
- Stage 3 (Surgical Anaesthesia): In this stage, excitement is lost and skeletal muscle relaxation is produced. Most types of surgeries are done at this stage.
- Stage 4 (Medullary Depression): Overdose of the anaesthetic drugs may cause this stage. Respiratory and circulatory failure occurs due to this.
Characteristics of General Anaesthetic Agents
An ideal general anaesthetic should possess the following characteristic features.
- It should be inert, potent and non-inflammable.
- It should be non-irritating to the mucous membrane and able to produce rapid loss of consciousness along with prompt recovery.
- It should produce analgesia and muscle relaxation in addition to anaesthesia.
- It should not produce severe hypotension, nausea and vomiting.
- It should be compatible with adjuvant drugs used in anaesthesia and should not show any interactions or adverse effects.
- It should be stable to heat, light and alkaline, should be economical.
Mechanism of Action:
Generally acts by depressing the neuronal activity in the brain. Also, exhibits different mechanisms of action by acting on different locations of the brain during various stages explained by lipid and protein theory.
- Lipid Theory: According to this, the more lipid soluble anaesthetics (general) concentrate near hydrophobic regions of neuronal cell membranes and causes swelling of these membranes.
- Due to this swelling, the structure of the membrane alters and thus it blocks the Na+ channels. This inhibits the generation of action potential and produces anaesthesia.
- Meyer and Overton in 1901, correlated this theory of potency of general anaesthetics with their lipid solubility. Its general anaesthetic potency increases with the increase in the partition coefficient of the compound.
- Protein Theory: According to this theory, the anaesthetic moiety binds itself to the hydrophobic sites of protein molecules of neuronal cell membrane which causes alteration in the membrane function and produces anaesthesia.
Classification of General Anaesthetics
- Inhalation anaesthetics: Halothane, Methoxyflurane, Enflurane, Sevoflurane,
Isoflurane, Desflurane. - Ultra short-acting barbiturates: Methohexital sodium, Thiamylal sodium, Thiopental sodium.
- Dissociative anaesthetics: Ketamine hydrochloride.
Inhalation Or Volatile Anaesthetics
They are generally administered by inhalation process and can be further divided into volatile liquids and gases.
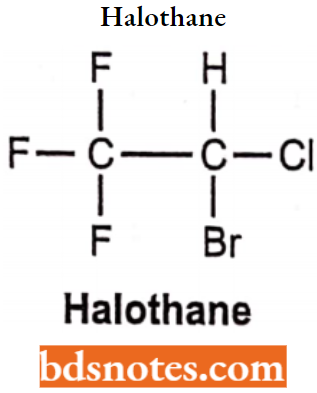
Halothane
Chemically halothane is 2-bromo, 2-chloro, 1,1,1-trifluoroethane. It produces a rapid onset of reaction without causing hypoxia. When given in combination with nitrous oxide the potency of the drug increases and is more potent than chloroform and ether. It is inflammable and thus safe to use and store.
Halothane Synthesis:
1. From trichloro ethylene:
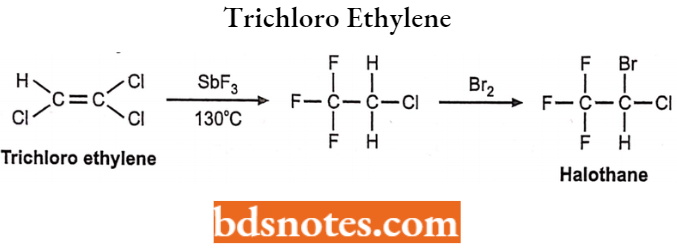
2. From trichloro ethylene:
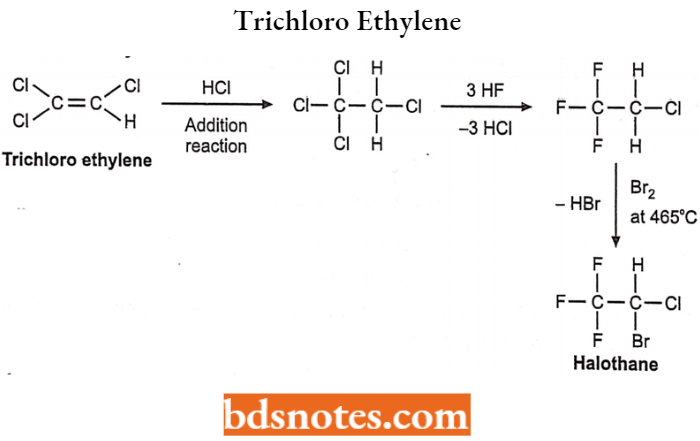
Halothane Side effects:
- It has a narrow margin of safety and a low incidence of hepatic necrosis.
- It produces respiratory depression and hypotension.
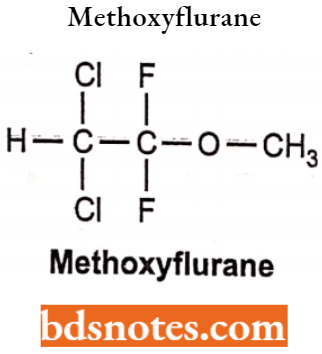
Methoxyflurane
Chemically, methoxyflurane is 2,2-dichloro-l,l-difluoro-l-methoxyethane. It is non-flammable, non-explosive and the most potent of all inhalation anaesthetics. They are lipid-soluble drugs. It is a colourless liquid with a sweet odour.
Methoxyflurane Side Effects: It has a large blood gas partition coefficient and therefore has a slow induction and recovery phase. It may cause renal damage on prolonged use due to fluoride release.
Enflurane
Chemically, Enflurane is (2-chloro-l,l, 2-trifluoromethyl) (trifluoromethyl) ether. It is a clear, colourless, volatile liquid with a pleasant hydrocarbon-like odour. Soluble in water, miscible with organic solvent. Chemically it is extremely soluble.
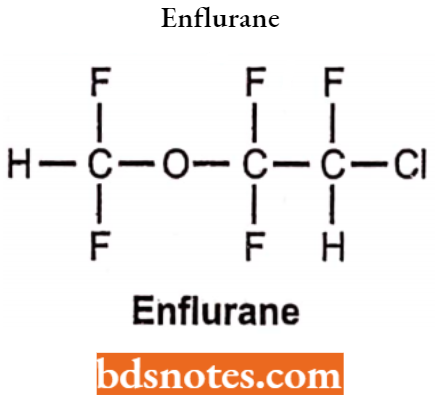
Enflurane Uses: Enflurane is used in the treatment of Sotus Aathmoticas Ventricular Premature Complexes. Used for the induction and maintenance of general anaesthesia during surgery and cesarian section and delivery.
Enflurane Side Effects: (Acute) Nausea, vomiting, irritation of eyes, nose, throat, skin, Headache. (Chronic) Drowsiness, arrhythmias, respiratory depression, and liver or kidney dysfunction.
Sevoflurane
Chemically sevoflurane is 1,1,1,3,3,3-hexafluro-2(fluoromethoxy) propane. It is a low-boiling liquid with a slight odour. Miscible with most organic solvents including fat and oils. Insoluble in water.
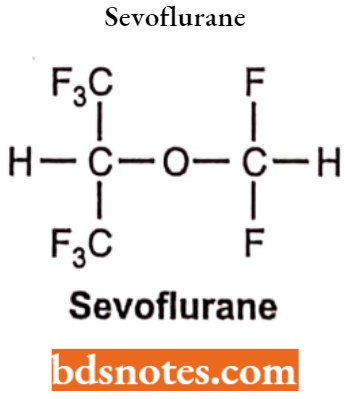
Sevoflurane Uses: It is one of the most commonly used volatile anaesthetic agents. It is often used to put children to sleep for surgery by inhalation.
Sevoflurane Side Effects: It is one of the most commonly used volatile anaesthetic agents. It is often used to put children to sleep for surgery by inhalation.
Isoflurane
Chemically, isoflurane is (l-chloro-2,2,2-trifluroethyl) (difluoro methyl) ether. It is a clear, colourless, heavy liquid. Insoluble in water, soluble in ethanol and trichloroethylene. Also miscible in organic liquids including fats and oils.
- It resembles isomer enflurane in properties.
- It is non-flammable in air or oxygen.
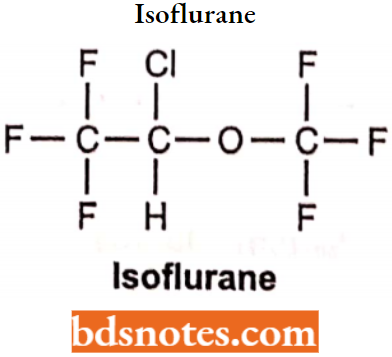
Isoflurane Uses: The depth of anaesthesia can be rapidly adjusted. Used for the induction and maintenance of general anaesthesia.
Isoflurane Side Effects: Respiratory depression, low B.P. and irregular heartbeat.
Desflurane
Chemically, desflurane is 1,2,2,2-tetrafluoroethyl difluoromethyl ether. It has a pungent odour, irritating and unpleasant to inhale and produces an approvable incidence of salivation. Causes breath-holding, coughing or laryngospasm when given to the conscious patient. It has a boiling point of 22.8°C.
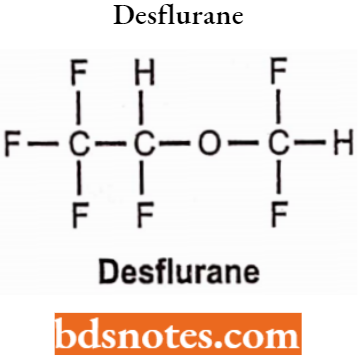
Desflurane Uses:
- It is used to cause general anaesthesia by inhalation before or during the surgery in adults.
- It has the most rapid onset and offset of all the volatile anaesthetic drugs used for general anaesthesia due to its low solubility in blood.
Desflurane Side Effects: Bluish lips or skin, body ache, pain cough, running nose, blurred vision, dizziness, headache.
Ultra Short Acting Barbiturates Or Non-Volatile
They are non-volatile at room temperature and are administered by intravenous route. They are used to produce rapid unconsciousness for surgical and basal anaesthesia.
These drugs induce anaesthesia during surgery which is then maintained by inhalation anaesthetics. Sodium salts of methohexital and thiopental are the most commonly used barbiturates used to produce anaesthesia.
Methohexital Sodium: Chemically, it is methohexital sodium-l-methyl-5-allyl-5-(l-methyl-2-pentynyl). It is administered by intravenous route or intramuscular route and synthesis is as follows:
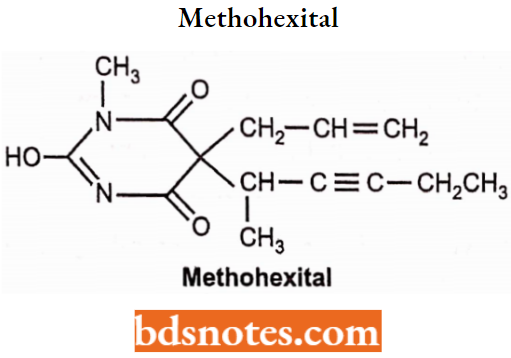
Methohexital Sodium Synthesis:
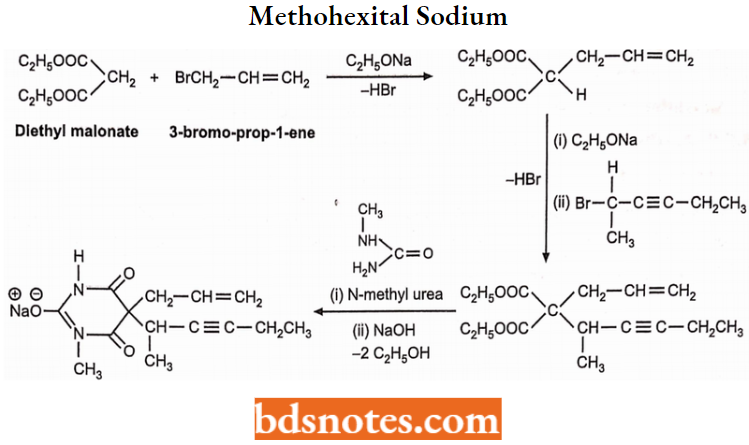
Methohexital Sodium Uses: It is used as a general anaesthetic and hypnotic drug for oral surgery, in gynaecological procedures, genito-urinary investigations and electroconvulsive therapy. It is more potent than thiopentone sodium.
Thiamylal Sodium: Chemically thiamylal sodium is a salt of 5-allyl-5 (1-methyl butyl)-2- thiobarbiturate. It is highly hydrophobic thiobarbiturate having similar structural features related to thiopental.
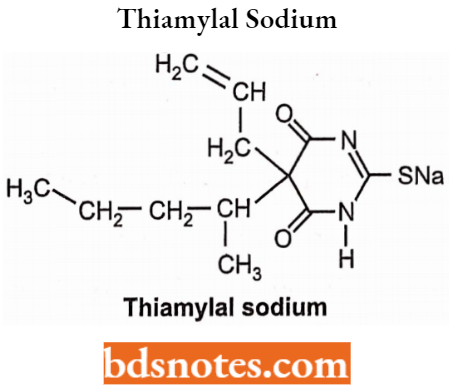
Thiamylal Sodium Uses: It is an intravenous anaesthetic drug used to induce drowsiness sleep or psychological excitement (anxiety).
Thiamylal Sodium Side Effects: Coughing, sneezing, slow breathing, slow heart rate, shivering, cardiac arrhythmia.
Thiopental Sodium: Chemically thiopental sodium is a [5-Ethyl-4,6-dioxo-5- (pentan-2-yl)-l,4,5,6-tetrahydropyrimidin-2-yl]sulfanide sodium. It is used commonly in the induction phase of general anaesthesia, administered by 4 routes. It has a rapid onset of action and causes unconsciousness within 30-45 seconds.
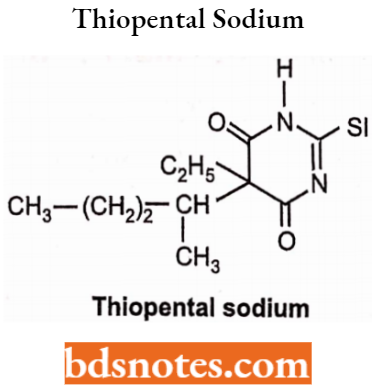
Thiopental Sodium Uses: Used in cesarean delivery. Also used in maintaining anaesthesia during surgery.
Thiopental Sodium Side Effects: Reduce cardiac output, and cause hypotension.
Dissociative Anaesthetic
Dissociative anaesthesia is a state in which a patient feels dissociative completely from the surroundings.
- Dissociative anaesthesia is a class of hallucinogens, which distort the perception of sight and sound and produce feelings of detachment – dissociation from the environment and self.
- This is done by reducing or blocking signals to the conscious mind from other parts of the brain.
- Many dissociative anaesthetics have general depressant effects and can produce sedation, respiratory depression analgesia and anaesthesia. Ketamine hydrochloride is the only drug used presently to produce this effect.
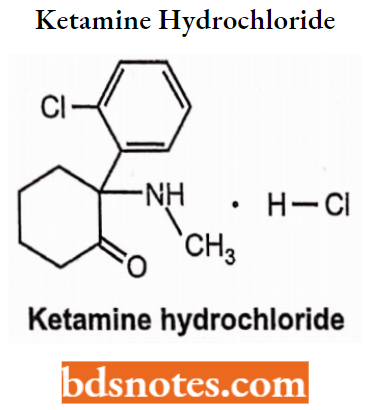
Ketamine Hydrochloride: Chemically, ketamine hydrochloride is (+) 2-(o-chlorophenyl)-2-methylaminocydohexanone). It is a colourless crystalline solid compound with a characteristic odour. Its melting point is 258°C.
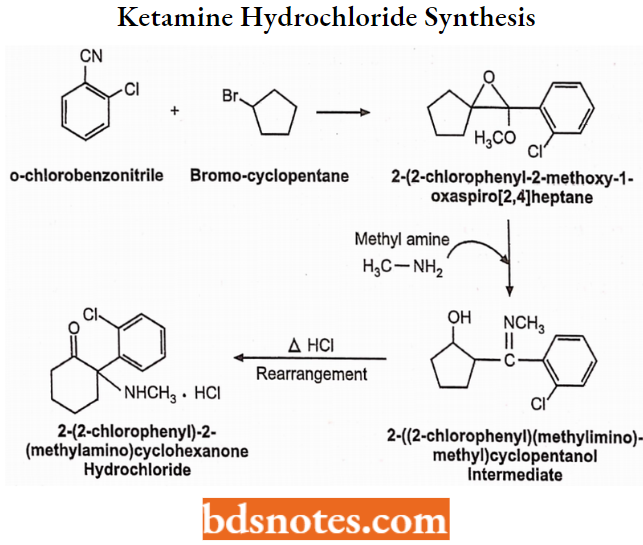
Ketamine Hydrochloride Synthesis: Ketamine is prepared by the Grignard reaction. In the presence of a strong alkali, o-chlorobenzonitrile reacts with bromocyclopentane to give an epoxy compound which converts to an imine in the presence of methylamine. It then rearranges to give ketamine on heating with HCI.
Ketamine Hydrochloride Uses: It can be used as a general anaesthetic and an analgesic. It causes the relaxation of skeletal muscles.
Ketamine Hydrochloride Side Effect: Include agitation, confusion or hallucinations, low blood pressure and muscle tremors. Spasms of the larynx may occur sometimes.
Narcotic And Non-Narcotic Analgesics
Classification of Analgesics:
- Natural compounds: Morphine, codeine, papaverine.
- Semi-synthetic compounds: Diacetylmorphine (Heroin), benzylmorphine,
ethylmorphine. - Synthetic compounds: Fentanyl, pethidine, methadone, tramadol, propoxyphene
hydrochloride (loperamide hydrochloride does not enter the brain and thus lacks
analgesic activity).
Mechanism of Action: All opioid receptors are G-protein coupled receptors and inhibit adenylate cyclase. They are also involved in,
- Postsynaptic hyperpolarization (increasing K+ efflux).
- Reducing presynaptic Ca++ influx thus inhibits neuronal activity.
Opioid receptors:
All opioid receptors are linked through G-proteins for the inhibition of adenylate cyclase. They facilitate the opening of potassium channels (causing hyperpolarisation) and inhibit the opening of calcium channels (inhibiting transmitter release).
There are four types of opioid receptors:
- μ-receptor: μ-receptors are thought to be responsible for most of the analgesic effects of opioids and for some major unwanted effects. Most of the analgesic opioids are p-receptor agonists. They are of two types:
- μ-1 receptor: This is located outside the spinal cord. This receptor is responsible for the central interpretation of the pain.
- μ-2 receptor: This is located throughout the CNS and is responsible for respiratory
depression, spinal analgesia, physical dependence and euphoria.
- σ-receptor: σ-receptors are not true opioid receptors and their activity is not clear. It may regulate p-receptor activity.
- δ-receptor: δ-receptors are more important in the periphery and may also
contribute to analgesia. - k-receptors: k-receptors contribute to analgesia at the spinal level and may elicit sedation and dysphoria, but produce relatively few unwanted effects and do not contribute to dependence.
Morphine And Related Drugs
Morphine is the major analgesic drug contained in crude opium. Morphine may be given by injection (intravenous or intramuscular) or as slow-release tablets. It is metabolized to morphine-6-glucuronide, which is the most potent analgesic.
Analgesia, euphoria and sedation, respiratory depression and suppression of cough, nausea and vomiting, reduce gastrointestinal motility.
Morphine And Related Drugs Side Effects: Addiction to drugs is due to a euphoric effect, overdose causes poisoning, coma and respiratory depression, dryness of mouth, mental clouding, vomiting, headache, fatigue, constipation etc.
The morphine molecule has the following important structural features:
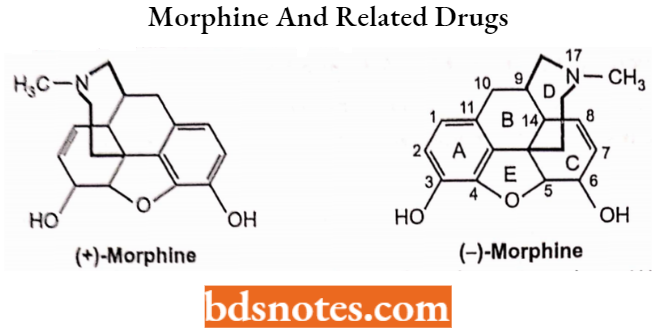
- A rigid pentacyclic structure consisting of a benzene ring (1), two partially unsaturated cyclohexane rings (2 and 3), a piperidine ring (4) and a dihydrofuran ring (5). Rings A, B and C are the phenanthrene ring systems.
- Ring A and its 3-hydroxyl group is an important structural feature for analgesic activity. Removal of the 3-OH group reduces analgesic activity by 10-fold.
- Two hydroxyl functional groups, a C3-phenolic OH (pKa 9.9) and a C6-allylic OH.
- An ether linkage between C4 and C5.
- Unsaturation between C7 and C8.
- A basic, 3c-amine function at position 17.
- Five centres of chirality (C5, C6, C9, C13 and C14) with morphine exhibiting a high degree of stereoselectivity of analgesic action. Only (-)-morphine is active.
SAR of Morphine Analogues
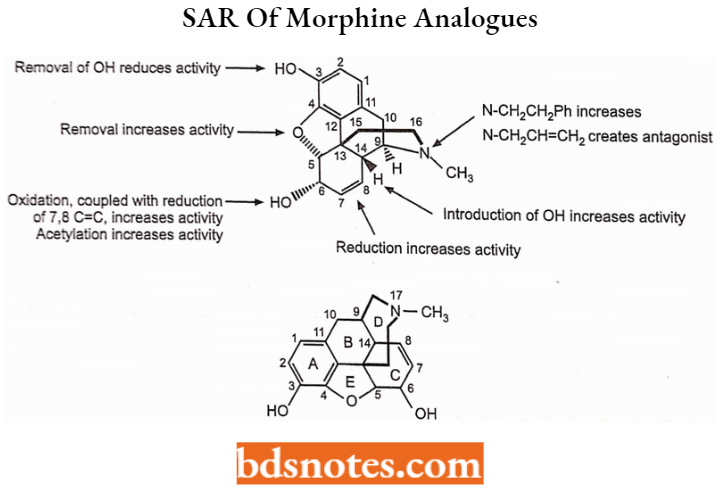
- Phenolic hydroxyl group at C3.
- Alcoholic hydroxyl group at C6.
- Alicyclic unsaturated linkage at C7-C8.
- Tertiary nitrogen.
- C14.
- Ether bridge.
Phenolic Hydroxyl Group at C3 (Ring 1 Analogues): Ring 1 or Aromatic phenyl ring and its 3-hydroxyl group is an important structural feature for analgesic activity. Removal of the 3-OH group reduces analgesic activity by 10-fold.
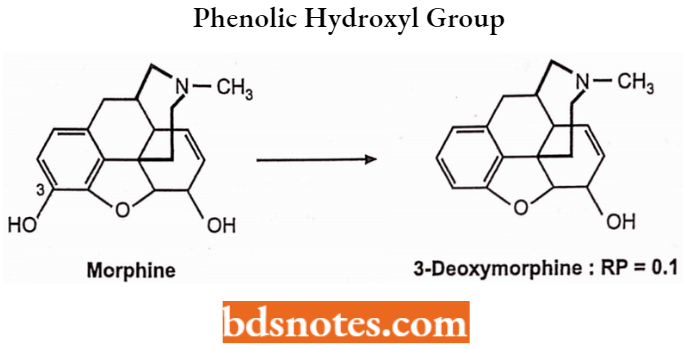
Altering the C-3 OH by etherification as shown by the derivatives below reduces narcotic analgesic activity.
Esterification (acetylation) of both the 3- and 6-OH groups yields heroin, which is more lipophilic and more potent. The primary factor involved in increased analgesic potency is the increased lipophilicity and distribution to the CNS.
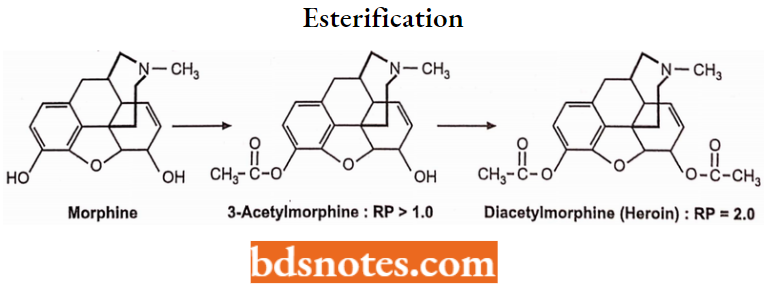
Heroin is much more potent than morphine but without a respiratory depression effect. Acetyl groups are readily removed by metabolism to active morphine. It is not used because of its additive nature.
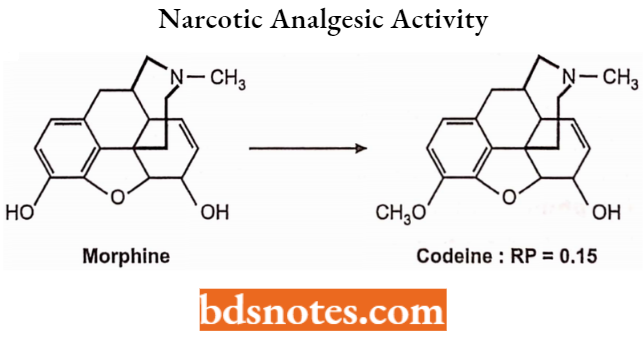
But in the case of codeine conversion of the 3-OH to a 3-OCH3, codeine reduces activity to 15% of morphine.
Alcoholic Hydroxyl Group at C6 (Ring 3 Analogues): The 6-OH of morphine is not required for analgesic activity as indicated by the relative potencies of the following morphine analogues.
Elimination of the 6-OH enhances activity. Etherification of this group with a relatively small alkyl group also increases activity.
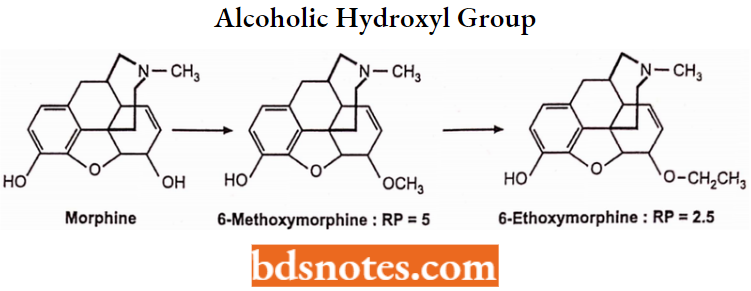
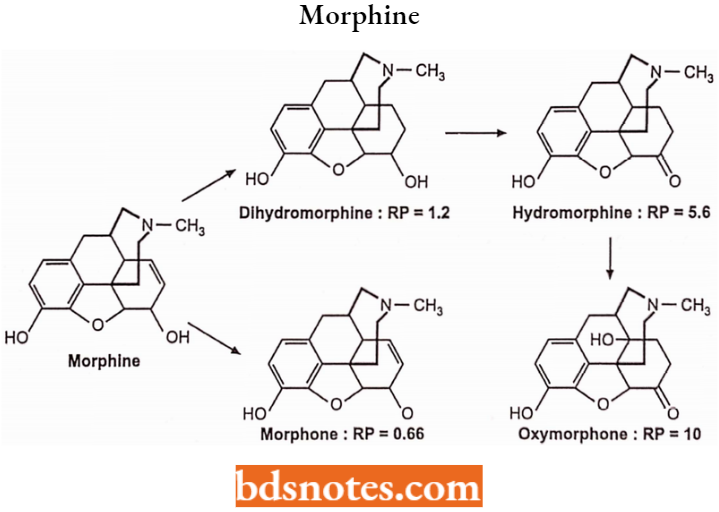
Oxidation of the 6-OH of dihydromorphine to yield hydromorphone further increases activity. Oxidation of the 6-OH of morphine directly as in morphine (without reduction of the 7,8-double bond) does not significantly alter analgesic activity.
In the case of codeine, oxidation of 6-OH of dihydrocodeine as in hydrocodone results in a further increase in activity.
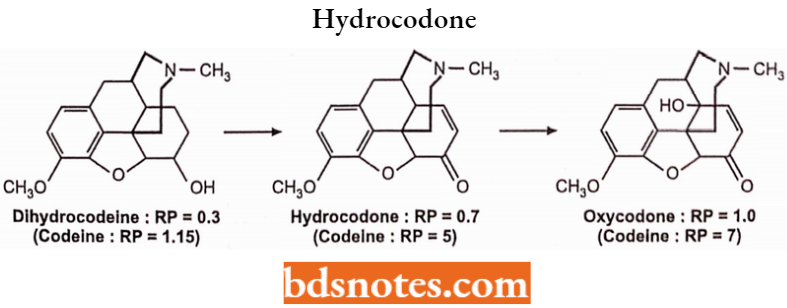
Hydrocodone is available in tablets and syrups and is used as an analgesic and anti-tussive activity. Oxycodone is available as tablets and capsules and is used as an analgesic and antitussive activity.
Alicyclic Unsaturated Linkage at C7-C8: The 7,8-double bond of morphine also is not required for analgesic activity as indicated by the relative analgesic potency of dihydromorphine.
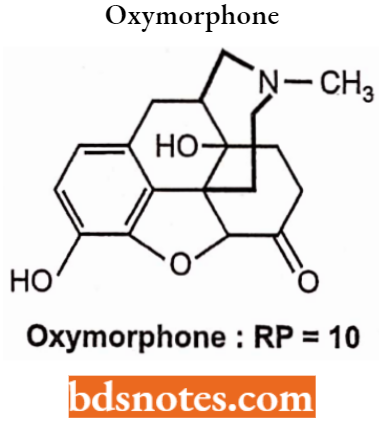
Substitution of a 14-OH group on the hydromorphone structure as in oxymorphone produces a further increase in analgesic activity (RP = 10).
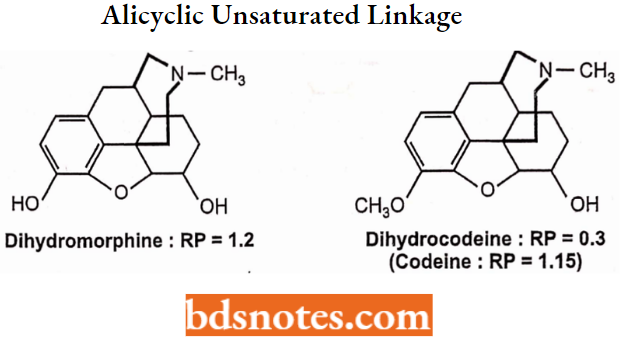
Reduction of codeine’s 7,8-double bond as in dihydrocodeine increases activity relative to codeine. 14-OH substitution produces a further increase in analgesic activity.
Tertiary Nitrogen (Ring 4 Analogues and the Tertiary Amine Function): Replacement of morphine’s N-methyl group by a hydrogen atom as in normorphine reduces analgesic activity to l/8th that of morphine, this decrease is due to increased polarity resulting in reduced blood-brain barrier translocation to the CNS.
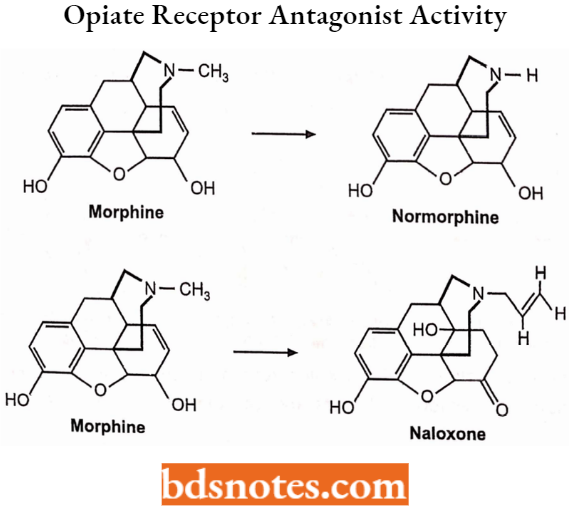
Replacement of morphine’s N-methyl group with an allyl group (-CH2-CH=CH2), a methylcyclopropyl group or a methylcyclobutyl group results in opiate receptor antagonist activity.
C14 (Ring 4 Analogues):
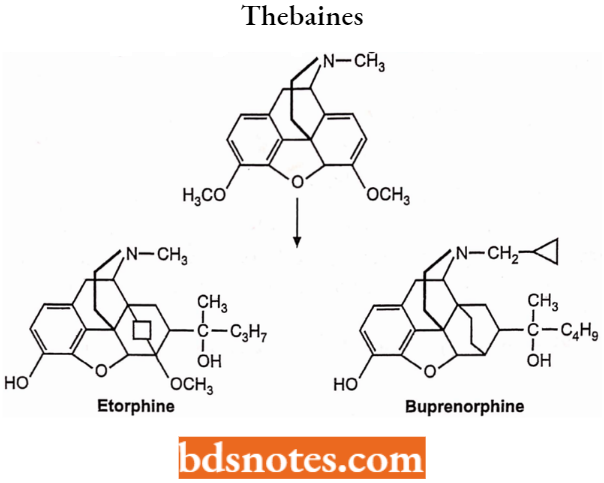
The Thebaines: Adding a sixth ring across carbons 6 and 14 of the C ring of morphine yields thebaine compounds such as etorphine which are extremely potent analgesics.
Replacement of the N-methyl group of the thebaine with a methyl cyclopropyl group yields compounds with mixed agonist or antagonist or partial agonist activity.
Epoxide Bridge: Removal of the 3,4 epoxide bridge in morphine structure is referred to as morphinans, only laevo isomer possesses opioid activity while the dextro isomer has useful antitussive activity.
Morphine Sulphate
Morphine is used primarily to treat both acute and chronic severe pain. It is also used for pain due to myocardial infarction and for labour pains. It acts directly on the central nervous system (CNS) to decrease the feeling of pain.
- It can be administered orally or by IV into a muscle, by injection under the skin, around the spinal cord, or rectally.
- Maximum effect is reached after about 20 minutes when given intravenously and after 60 minutes when given orally, lasting for 3-7 hours.
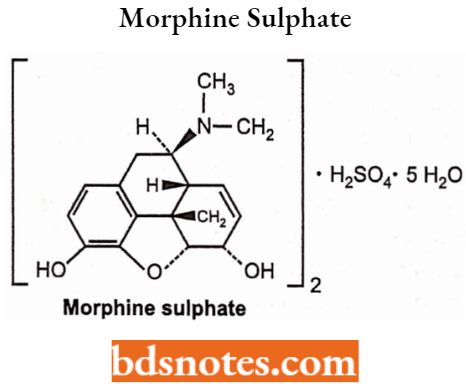
Morphine Sulphate Sife Effects: Constipation, dizziness, perspiration, dry mouth, visual difficulty, itching, euphoria, restlessness, nervousness and excitement, addiction.
Codeine
Codeine is an opiate drug used to treat mild to moderate degrees of pain and also the pain associated with cough and diarrhoea. For more effects, it has been combined with paracetamol or with non-steroidal anti-inflammatory drugs.
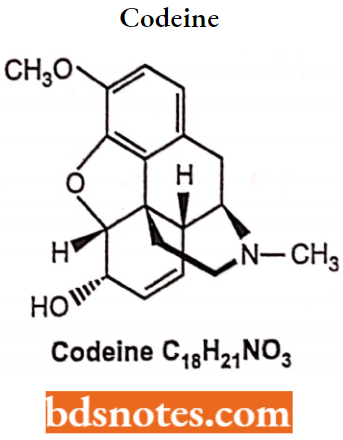
Codeine Side Effects: Vomiting, constipation, itchiness, light-headedness, drowsiness, breathing difficulties and addiction. It is not recommended in children and pregnant women without consultation.
SAR of Phenyl (Ethyl) Piperidines or Meperidine Analogs
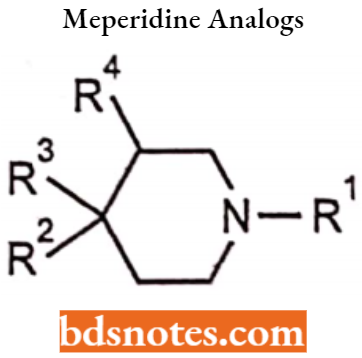
- Replacement of the C-4 phenyl group of meperidine by H, alkyl, aryl, aryl-alkyl and
heterocyclic group decreases the analgesic activity. - The introduction of the m-OH group on the phenyl ring increases the activity.
- The presence of phenyl and ester group at the 4th position of 1-methyl piperidine results in optimum activity.
- Replacement of the carb ethoxy group in Mepiridine by the acyloxy group gave better analgesic as well as spasmolytic.
- Replacement of the phenyl group by phenylmethyl derivative seems to be 3 times as active as Mepiridine.
- The amino congener is 4 times more active (Anileridine). Enlargement of piperidine ring to 7-membered hexahydroazepine is less active but has a low incidence of side effects, (For Example.) Proheptazine.
- Contraction of the piperidine ring to pyrrolidine gives a more active compound but causes abuse liability (For Example) Alphaprodine and Procilidine.
- In fentanyl, the phenyl and acyl groups are separated by nitrogen. It is 50 times stronger than morphine with minimal side effects.
- Its short duration of action makes it well-suited for use in anaesthesia. The C-3 methyl analog with an ester group at C-4 like lofentanil is 8400 times more potent than Meperidine as an analgesic.
p-Chloroanalogloperamide cannot penetrate BBB sufficiently to produce analgesia. Diphenoxylate, a structural hybrid of Meperidine and Methadone is devoid of analgesic activity. It is effective in the treatment of diarrhoea.
Meperidine Hydrochloride
It is a narcotic analgesic used for the relief of most types of moderate to severe pain, including postoperative pain and labour pain. R3– ethyl carboxyl ester substitute increases the activity of morphine.
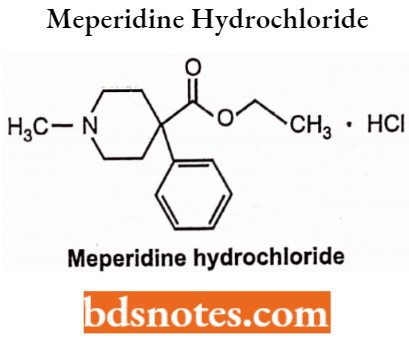
Meperidine Hydrochloride Side Effects: Prolonged use may lead to dependence of drugs similar to morphine.
Anilerdine Hydrochloride
It is a synthetic analgesic drug from the piperidine class of analgesic agents. It differs from pethidine (meperidine) in that the N-methyl group of meperidine is replaced by an N-aminophenyl group, which increases its analgesic activity.
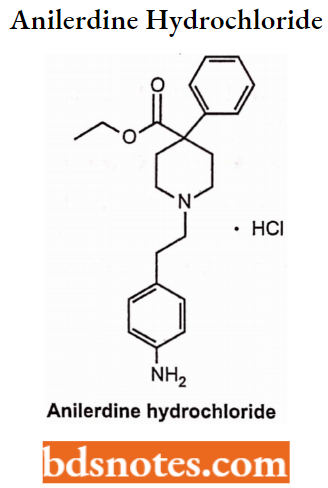
Anilerdine Hydrochloride Side Effects: Dizziness, perspiration, a feeling of warmth, dry mouth, visual difficulty, itching, euphoria, restlessness, nervousness and excitement.
Diphenoxylate Hydrochloride
Diphenoxylate is the derivatives of the phenylpepridines. It is used for the treatment of diarrhoea.
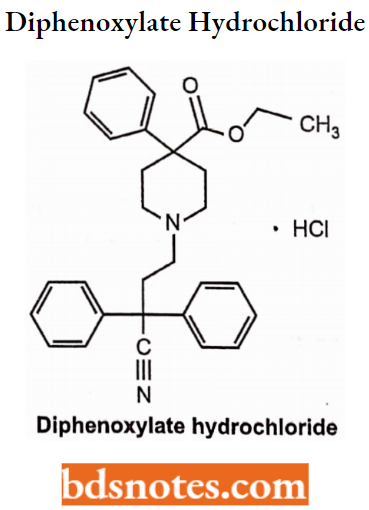
Diphenoxylate Hydrochloride Side Effects: It should not be given to children due to breathlessness. It also includes numbness in the hands and feet, euphoria, depression, lethargy, confusion, drowsiness, dizziness, restlessness, headache, hallucinations, oedema, swollen gums, itchiness, vomiting, nausea, loss of appetite, and stomach pain.
Loperamide Hydrochloride
Loperamide can be taken orally. It is also used to treat diarrhoea in gastroenteritis, inflammatory and short bowel syndrome.
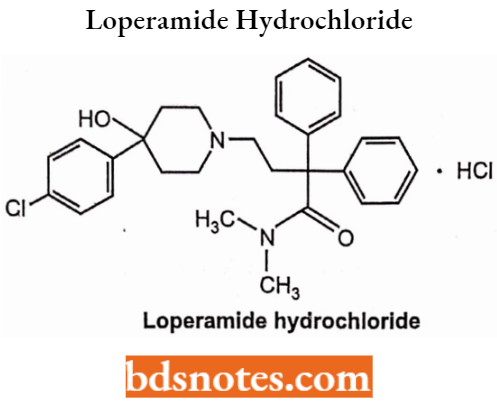
Loperamide Hydrochloride Side Effects: It causes abdominal pain, constipation, sleepiness, vomiting, and a dry mouth.
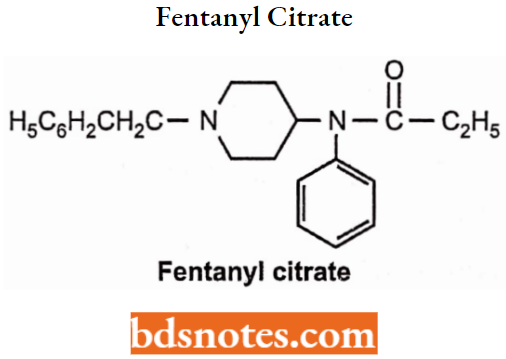
Fentanyl Citrate
Chemically, fentanyl citrate is N- (l-Phenylethyl-4-piperidinyl) propionate-anilide citrate. It is structurally related to phenylpiperidines (For Example. meperidine). R1 is a phenyl substituent group that increases the activity 80 times more than morphine.
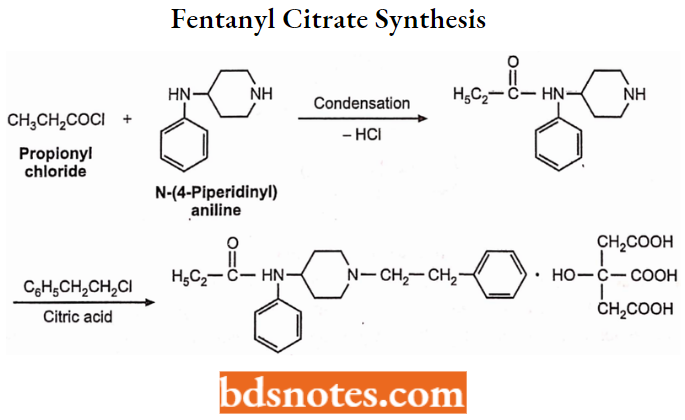
Fentanyl Citrate Synthesis: N-(4-piperidinyl) aniline is prepared by reductive amination of 4-piperidone and aniline, which is condensed with propionyl chloride to form an amide. On N-alkylation with phenyl ethyl chloride, it gives fentanyl.
Fentanyl Citrate Use: Fentanyl citrate relieves moderate to severe breakthrough pain.
Methadone Hydrochloride: Chemically, Methadone hydrochloride is 6-(dimethylamino)-4,4-diphenyl-3-heptanone hydrochloride which is widely used as a narcotic analgesic, particularly in clinical treatment for the withdrawal of heroin addiction.
It is a synthetic drug and can be given orally. Its potency is similar to morphine but induces less euphoria and has a longer duration of action. The drug is bio-transformed into inactive metabolites in the liver and is excreted in the urine.

Methadone Hydrochloride Synthesis: Methadone is prepared by reaction of 2,2-diphenyl-4-(dimethylamino)- pentane nitrile with an ethylmagnesium halide.
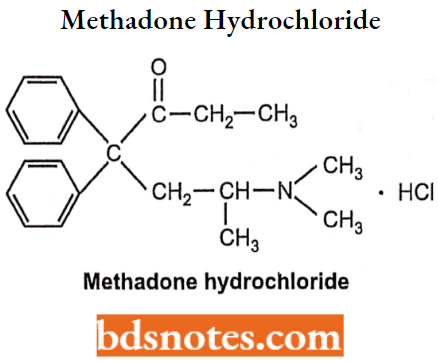
Methadone Hydrochloride Side Effects: It can produce physical dependence like morphine, restlessness, nausea or vomiting, slow breathing, itchy skin, heavy sweating, and constipation.
Propoxyphene Hydrochloride: Propoxyphene is a narcotic pain reliever. It is used to relieve mild to moderate pain.
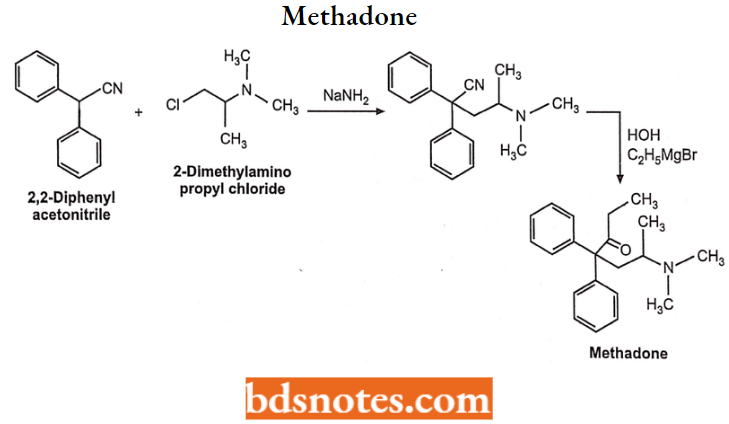
Propoxyphene Hydrochloride Side Effects: Propoxyphene may be habit-forming. Not given to the patients having a history of addiction without consultation. It also causes confusion, hallucinations, vomiting, and slow breathing.
Pentazocine
Pentazocine is used to treat moderate to severe pain. Chemically it is classed as a benzomorphan and it comes in two enantiomers.
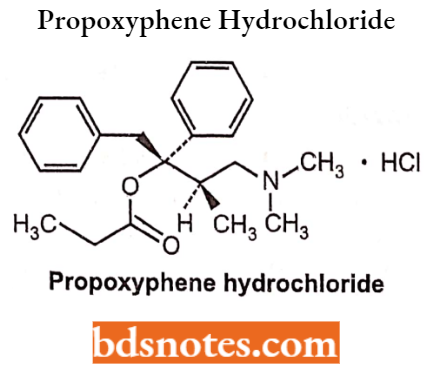
Pentazocine Side Effects: High blood pressure, myocardial infarction, and respiratory depression.
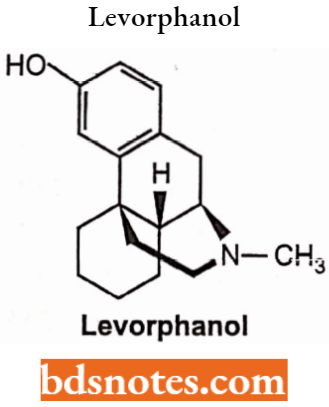
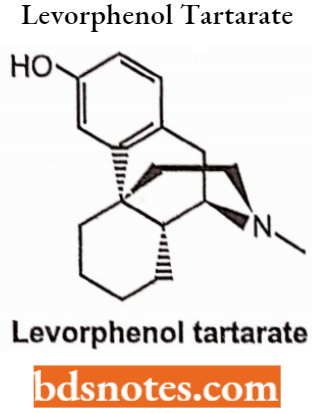
Levorphanol Tartrate
Chemically, levorphanol is 3-hydroxy-N-methyl-morphinan. It is used to treat moderate to severe pain. It can be taken orally and has morphine-like analgesic activity.
Levorphenol Tartarate Side Effects: Hallucinations, drowsiness, dizziness, restlessness, headache, nausea.
Narcotic Antagonists
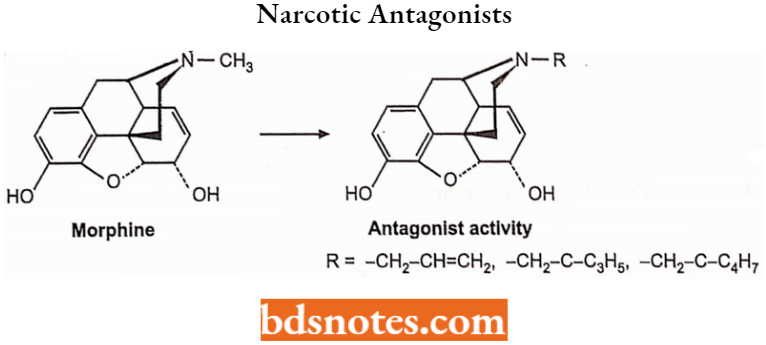
Replacement of morphine’s N-methyl group with an allyl group (-CH2-CH=CH2), a methylcyclopropyl group or a methylcyclobutyl group results in the emergence of opiate receptor antagonist activity like naloxone HCI, naltrexone HCl and nalmefene respectively.
Nalorphine Hydrochloride: Nalorphine is N-allylmorphine. In morphine, tertiary nitrogen is attached to an allyl (-CH2CH=CH2) group. It is a white-coloured, odourless, crystalline powder.
- It darkens on exposure to light, so it has to be kept in a tightly closed container. It is soluble in water, dilute alkali hydroxide solution but insoluble in chloroform and ether.
- It was used as an antidote to reverse opioid overdose and in a challenge test to determine opioid dependence.
- Nalorphine is a mixed opioid agonist-antagonist with opioid antagonist and analgesic properties, used to treat narcotic-induced respiratory depression.
It is administered by intravenous injection to treat the overdosage of morphine, pethidine, methadone and levorphanol.
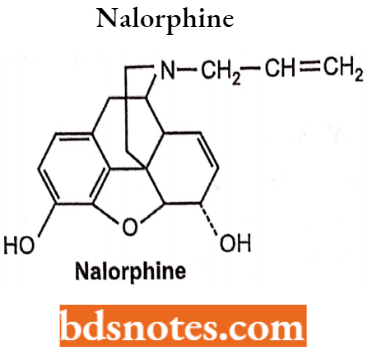
Nalorphine Hydrochloride Side Effects: Dysphoria, anxiety, confusion, and hallucinations and therefore no longer used medically.
Levallorphan Tartarate
Levallorphan is available as tartrate salt, it is a white-coloured, odourless, crystalline powder with a melting point of around 175°C. It is slightly soluble in water but insoluble in ether and chloroform.
- It acts as an antagonist of the μ-opioid receptor and as an agonist of the K-opioid receptor.
- It blocks the effects of stronger agents like morphine with greater intrinsic activity. It is
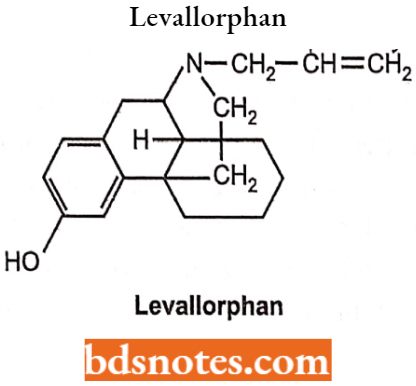
Levallorphan Tartarate Side Effects: Hallucinations, psychotomimetic effects, dysphoria, dizziness, disorientation, anxiety, confusion.
Naloxone Hydrochloride: Naloxone is N-allyl-4, 5-epoxy-3, 14-dihydroxymorphinan-6-one. Naloxone is a derivative of 7, 8-dihydro-14-hydroxymorphinone having an allyl group at the nitrogen. Naloxone is administered by IV or IM and has a relatively short half-life (1 hour).
Replacement of the potent narcotic agonist oxymorphone’s N-methyl group with an allyl group (-CH2-CH=CH2) gives naloxone. Naloxone is a pure antagonist without morphine-like effects. It blocks the euphoric effect of heroin when is given before heroin.
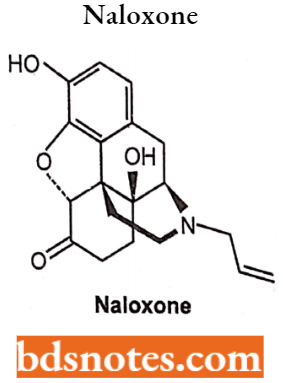
Naloxone Hydrochloride Uses: Naloxone is a pure antagonist with no morphine-like effects. It blocks the euphoric effect of heroin when given before heroin.
Naloxone Hydrochloride Side effects: Nausea, vomiting, diarrhoea, stomach pain, fever, sweating, body aches, weakness, tremors or shivering, fast heart rate, pounding heartbeats, increased blood pressure, restlessness.
Anti-Inflammatory Agents
Anti-inflammatory agents are the drugs used to reduce inflammation and pain for the management of oedema and tissue damage. They are also called non-steroidal anti-inflammatory drugs (NSAIDs) and non-narcotic analgesics.
- It can be used topically, orally, and parenterally for treating inflammation. A large number of drugs have antipyretic, anti-inflammatory, and analgesic properties and thus they can be used for the treatment of fever.
- NSAIDs are used in the treatment of rheumatoid arthritis, osteoarthritis (OA), acute gouty arthritis, ankylosing, spondylitis, dysmenorrhea and tissue damage resulting from inflammatory joint disease (arthritis). It has a wide range of therapeutic activities.
Characteristic Features of Anti-inflammatory Agents:
- NSAIDs were also known as non-opioid analgesics, and non-narcotic drugs as they do not interact with opioid receptors.
- They do not have a steroidal ring.
- Their structure consists of an acidic moiety (carboxylic or ends) attached to the planner aromatic functional group. The acidic group is essential for COX inhibitory activity and is the major binding group with plasma protein. Some contain a polar linking group which is attached to planner moiety as an additional group.
- Certain drugs have varying degrees of activity against thrombocytes.
- They act by exerting an inhibitory effect on cyclooxygenases (COX-1 and COX-2) and thus inhibit the generation of various prostaglandins (PGs).
- These drugs were metabolised and transferred by glucuronidation and then excreted via kidneys.
Classification of Anti-inflammatory Agents:

Mechanism of action of NSAIDs: The major mechanism by which the NSAIDs show their therapeutic effects (antipyretic analgesic, and inflammatory activities) is the inhibition of prostaglandin (PG) synthesis.
Specifically, NSAIDs competitively inhibit cyclooxygenases (prostaglandin synthetase), the enzymes that catalyze the synthesis of cyclic endoperoxides from arachidonic acid to form prostaglandins. Generally, the NSAIDs inhibit both COX-1 and COX-2
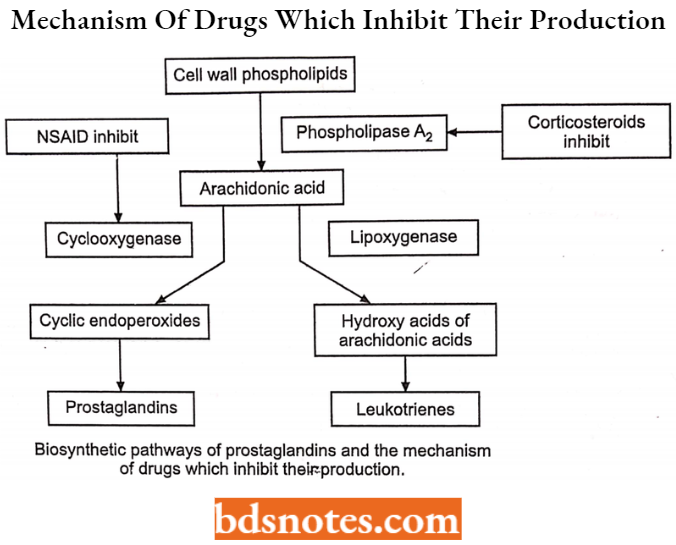
Further NSAIDs can be classified based on their chemical structure:
- Salicylates: aspirin, sodium salicylate.
- Propionic acids (Profens).
- Aryl and heteroarylacetic acids.
- Anthranilates (Fenamates).
- Oxicams (“Enol Acids”).
- Phenylpyrazolones.
- Anilides.
Salicylates
They are the derivatives of 2-hydroxybenzoic acid and used as a sodium salt. It has analgesic, antipyretic and anti-inflammatory activity.
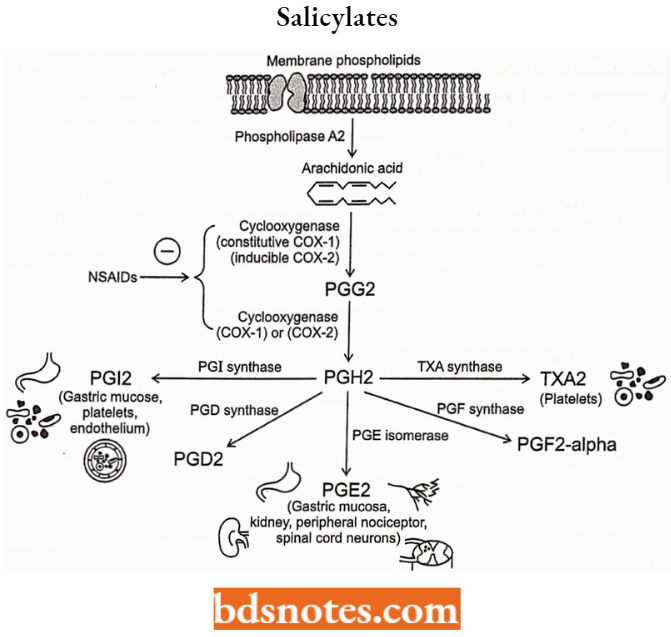
Salicylates Mechanism of Action: They act by inhibiting the biosynthesis of prostaglandins at the cyclooxygenase stage, thus blocking the formation of a potent platelet aggregating factor, thromboxane A2 (TX A2).
These drugs are mainly COX-1 selective and bound with higher affinity to COX-1. Some of the therapeutic actions of aspirin and related drugs as they inhibit COX 1 used to treat headache, discomfort, fever, muscle pain aches and the common cold.
Salicylates SAR:
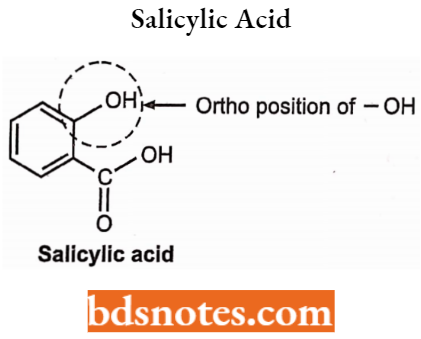
COOH functional group at position -Cl and -OR group at -C2 is essential for the anti-inflammatory activity of salicylic acid derivatives (salicylates).
- Replacement or substitution of the COOH group may decrease or abolish the anti-inflammatory activity. It may show some analgesic activity, For Example, Salicylamide.
- Substitution of the -OR group by the -OH group at the -C2 position increases the activity but decreases if placed at -C3, -C5 (meta) and -C4 (para) positions.
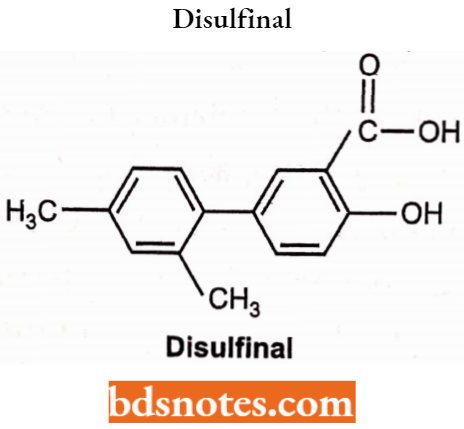
Aryl group at -C5 position increases activity. Substitution by halogen or alkyl groups on the aryl ring also increases the activity, For Example. Disulfiram.
Sodium Salicylate: Chemically, it is 2-hydroxy benzoate.
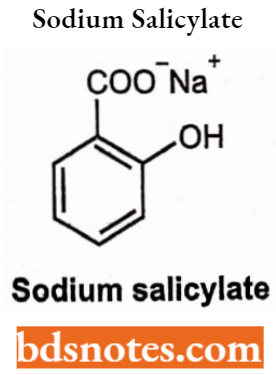
Sodium salicylate is sodium 2-hydroxybenzenecarboxylate. It occurs as a white, crystalline powder or small, colourless crystals or shiny flakes. It is freely soluble in water, sparingly soluble in alcohol and practically insoluble in ether. It should be stored in an airtight container and protected from light.
Sodium Salicylate Uses: Sodium salicylate is employed for the relief of pain, rheumatic fever and symptomatic treatment of gout.
Aspirin (Acetylsalicylic Acid): It is an acetyl derivative of salicylic acid and was introduced by Dreser in 1899. Aspirin occurs as colourless crystals or powder. It is slightly soluble in water and soluble in alcohol, chloroform, ether and glycerine.
Aspirin is stable in dry air but in the presence of moisture, it hydrolyses slowly into salicylic acid and acetic acid. Aspirin is acidic and produces effervescence with carbonates and bicarbonates.
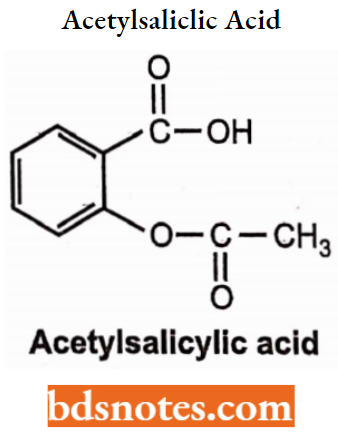
Aspirin Use: Aspirin is used as an antipyretic, analgesic and anti-rheumatic.
Aspirin Synthesis: It is an esterification reaction, in which the reaction between the hydroxyl groups of salicylic acid reacts with acetic anhydride to form an ester in the presence of an acid as a catalyst.

Propionic Acids Derivatives (Profens): Some NSAIDs were derived from arylacetic acids, known as the “profits” For Example., ibuprofen. These agents are all strong organic acids and form water-soluble salts with alkaline reagents.
The arylpropionic acids are characterized by the general structure Ar-CH(CH3)-COOH which confirms the required general structure.
Propionic Acids Derivatives Mechanism of Action: They are COX-1 inhibitors. Some compounds like naproxen appear to be more COX-2 inhibitors as compared to others.
Propionic Acids Derivatives Uses: For rheumatoid arthritis, osteoarthritis, analgesic and antipyretic. They should not be used during pregnancy or nursing. They produce less GI ulceration than the salicylates.
Propionic Acids Derivatives SAR:
- α-CH3 substituent present in the structure increases cyclooxygenase inhibitory activity and reduces toxicity of the profens.
- The α-carbon in these compounds is chiral and the S-(+)-enantiomer of profits is a more potent cyclooxygenase inhibitor.
Propionic Acids Derivatives General structure:
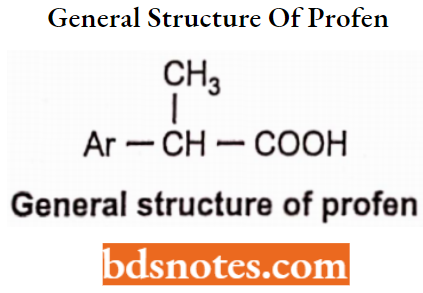
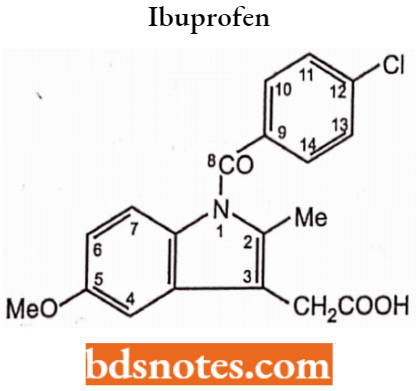
Ibuprofen
Ibuprofen: It is a non-steroidal anti-inflammatory drug (NSAID), used for treating pain, fever, and inflammation.
Ibuprofen Mechanism of Action: Ibuprofen is a non-selective, reversible inhibition of the cyclooxygenase enzymes COX-1 and COX-2 (coded for by PTGS1 and PTGS2, respectively).
Ibuprofen SAR
- The carboxyl group is essential for anti-inflammatory activity.
- The presence of the methoxy (position 5) group on the ring (5 or 6), methyl (2), and dimethyl amino group (5) in the indole moiety of indomethacin exhibit activity.
- The presence of chlorine fluorine or CF3 groups at the para position of the phenyl group also exhibits anti-inflammatory activity.
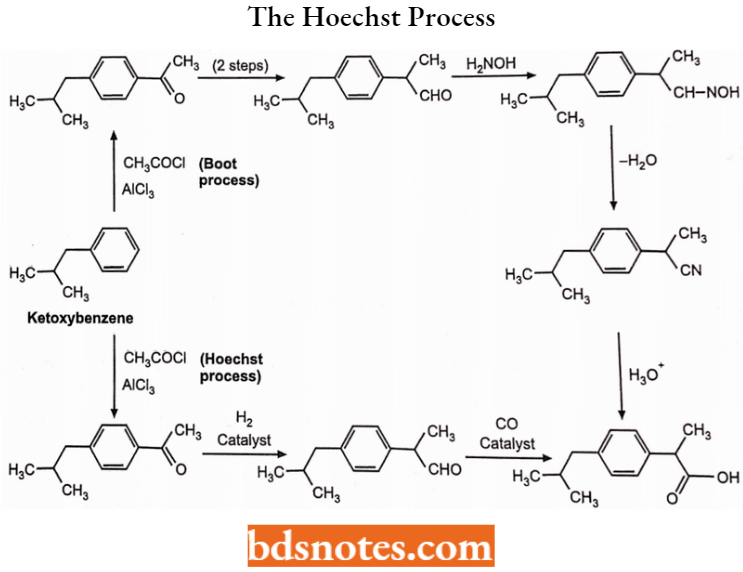
Synthesis of Ibuprofen: Two methods to obtain ibuprofen
- The Boot process: The Boot process is an older commercial process developed by the Boot Pure Drug Company.
- The Hoechst process: The Hoechst process is a newer process developed by the
Hoechst Company. While the Hoechst process, with the assistance of catalysts.
Ibuprofen synthesis begins with isobutylbenzene and use Friedel-Craft’s acylation.
Ibuprofen Uses: In the treatment of rheumatoid arthritis, and osteoarthritis, analgesic and antipyretic. It should not be used during pregnancy or nursing as it can enter fetal circulation and breast milk. It has less GI ulceration than the salicylates.
Naproxen: Chemically, Naproxen is naphthyl 6-methoxyisopropionic acid. It is a non-steroidal anti-inflammatory, antirheumatic, analgesic, antidysmenorrhoeal and vascular headache suppressant.
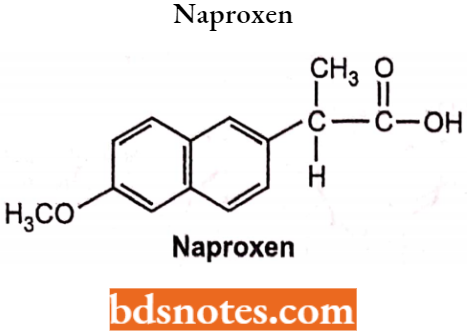
Naproxen has (partly) the ability to inhibit COX-1 and COX-2. It irreversibly blocks the enzyme cyclooxygenase (prostaglandin synthase), which catalyzes the conversion of arachidonic acid to endoperoxide compounds and decreases the formation of prostaglandins.
Aryl and Heteroarylacetic Acid Derivatives: These compounds are the derivatives of acetic acid, but substituent at the -C2 position is a heterocycle or carbocycle. This does not alter the acidic properties of compounds of this group.
These NSAIDs can be further sub-classified as:
- Indene and indoles
- Pyrroles
- Oxazoles
Aryl and Heteroarylacetic Acid Derivatives General Structure:
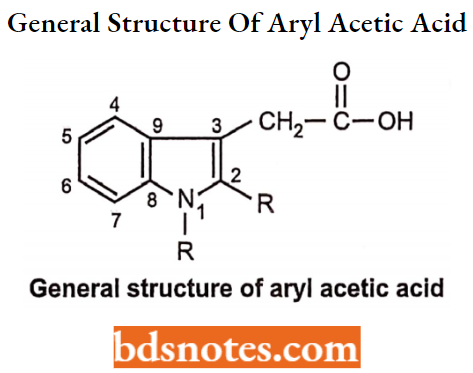
Aryl and Heteroarylacetic Acid Derivatives SAR:
The COOH group is essential for the activity. Replacement of this group diminishes or abolishes the activity.
- An increase in acidity of the COOH group will increase the anti-inflammatory activity. A decrease in COOH acidity decreases the activity.
- The presence of an indole ring is necessary for the activity. Substitution at the C5 position (R2) by fluro, methyl, dimethylamino, alkoxy or acetyl group increases the activity. The Alkyl group at the C2 position increases activity as compared to aryl groups. Substitution at the acetic acid chain gives compounds of greater activity, For Example, Indomethacin, sulindac, ketorolac,
- The presence of methoxy at the -C5 position group on the ring, methyl at the C2 position, and dimethyl amino group at the C5- position in the indole moiety of indomethacin exhibit activity.
- The presence of chlorine fluorine or CF3 groups at the para position of the phenyl group also gives anti-inflammatory activity.
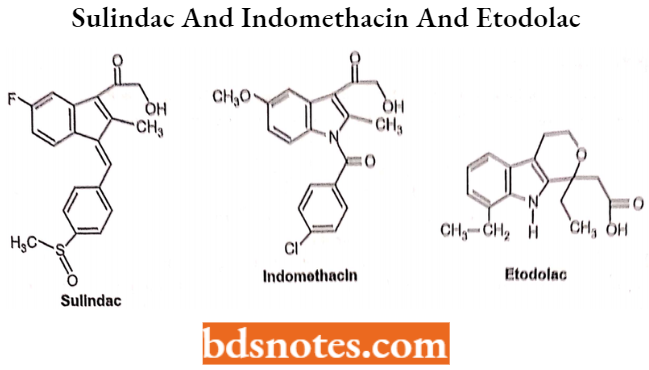
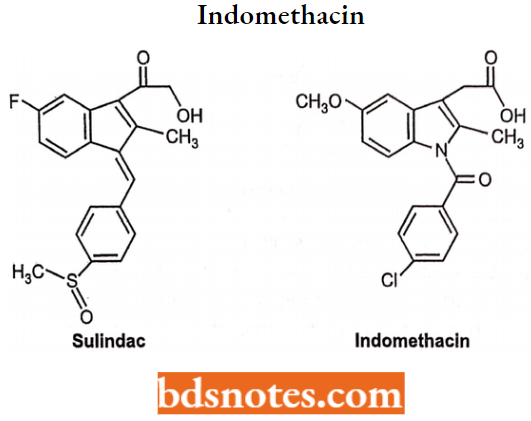
Indomethacin: Chemically, indomethacin is l-(p-chlorobenzoyl)-5-mothoxy-2-mothyllndolo-3-acetlc acid. It consists of a benzene ring fused to an indole ring. Indomethacin Is available as white to yellow crystalline powder. It is practically soluble in water.
Indomethacin Structure-activity Relationship (SAR):
- The carboxyl group is essential for anti-inflammatory activity.
- The presence of the methoxy (position 5) group on the ring (5 or 6), methyl (2), and dimethyl amino group (5) in the indole moiety of indomethacin exhibit activity.
- The presence of chlorine fluorine or CF3 groups at the para position of the phenyl group also exhibits anti-inflammatory activity.
Indomethacin Side Effects: The most frequent side effects are peptic ulcer, blood disorders and gastrointestinal.
Sulidac
(Arylalkanoic acid Derivative): Chemically, it is 5-fluro-2 methyl [(4 methyl sulphinyl) phenyl methylene] indene-3-acetic acid. It is a yellowish crystalline powder. Very slightly soluble in water, soluble in methylene chloride and dilute solution of alkali hydroxides, sparingly soluble in alcohol.
Sulidac Uses: It has an analgesic, and anti-inflammatory activity; it is usually employed in the treatment of rheumatic and muscular skeletal disorder, acute gout arthritis and osteoarthritis.
Sulidac Side Effects: Affect CNS and gastrointestinal irritations.
Tolmetin
It is a non-steroidal anti-inflammatory drug (NSAID) of the heterocyclic acetic acid derivative class. It acts by reducing the levels of prostaglandins and thus reduces inflammation.
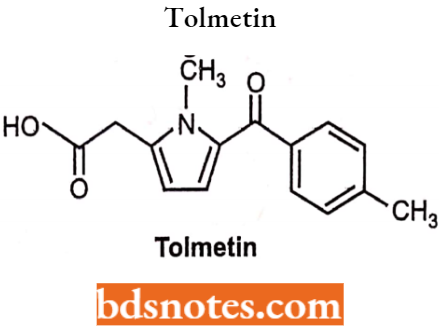
Tolmetin Uses: To reduce hormones that cause pain, swelling, tenderness, and stiffness in conditions such as osteoarthritis and rheumatoid arthritis, including juvenile rheumatoid arthritis.
Tolmetin Side Effects: GI irritations, ulcer.
Pyrrolesarylacetic Acids
Ketorolac: It does not have a benzylic methyl group, thus its half-life is about (4-6 hours) as it is not susceptible to oxidation. It is formulated for oral and IM administration showing good oral activity with primarily analgesic activity along with anti-inflammatory activity and antipyretic actions.
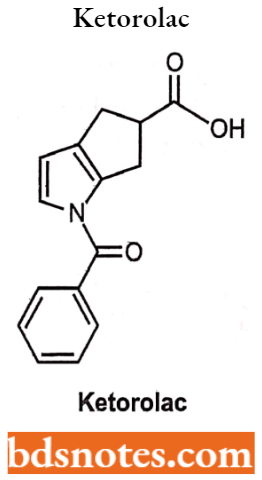
Pyrrolesarylacetic Acids Uses: In the management of post-operative pain
Pyrrolesarylacetic Acids Side Effects: Stomach pain, GI irritations, ulcer.
Oxazoles Arylacetic Acids: In 1993, oxaprozin was introduced having a non-selective COX inhibition activity. It differs from indomethacin and other compounds in the substitution of the propionic acid at the C3-position rather than at the C2-position.
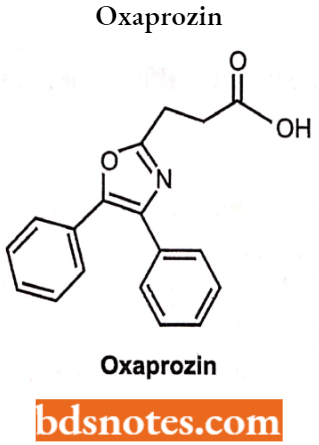
Oxazoles Arylacetic Acids Uses: In the treatment of pain associated with surgery.
Oxazoles Arylacetic Acids Side Effects: Vomiting, nausea, GI irritations, ulcer.
Anthranilates (Fenamates): (Anthranilic Acid Derivatives)
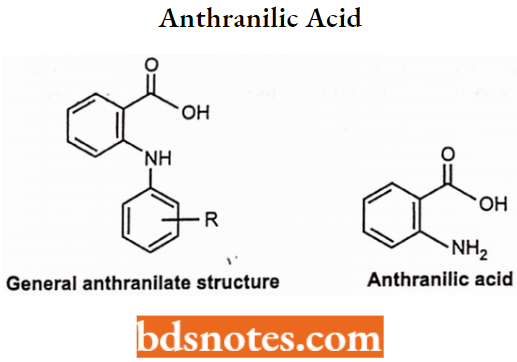
Anthranilates (Fenamates) SAR:
- They are N-aryl substituted derivatives of anthranilic acid.
- These compounds have small alkyl or halogen substituents at the C2′, C3′ and or C6′ position of the N-aryl moiety and are 25 times more potent than mefenamate.
- The presence of -NH moiety is essential for the activity. Substitution by ether, thioether, ketonic, methylene or sulphur group decreases the activity.
Anthranilates (Fenamates) Mechanism of Action: They are non-COX selective and have anti-inflammatory with some analgesic and antipyretic activity. The anthranilates are used as mild analgesics and occasionally to treat inflammatory disorders.
Diclofenac is used for rheumatoid arthritis, osteoarthritis and post-operative pain and the utility of this class of agents is limited by several adverse reactions including nausea, vomiting, diarrhoea, ulceration, headache, drowsiness and hematopoietic toxicity.
Mefanamic Acid: Chemically, it is N-2,3-xylyl anthranilic acid. It produces analgesic activity centrally and peripherally.
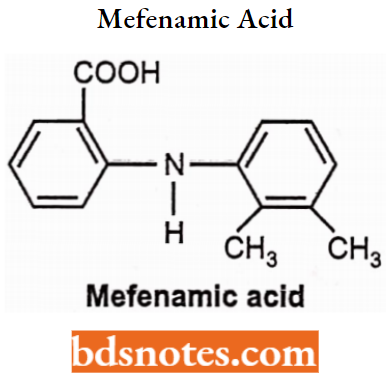
Mefenamic Acid Synthesis: Mefenamic acid can be prepared by condensing 2,3-xylidine with
2-chlorobenzoic acid under acidic conditions.
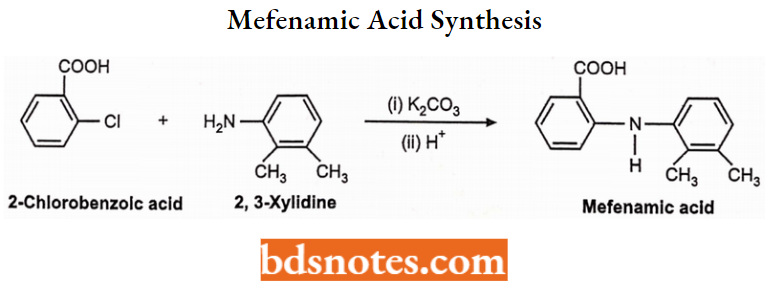
Mefenamic Acid Uses: Mefenamic acid is used as an analgesic for short-term relief from dysmenorrhoea.
Mefanamic Acid Side Effects: Nausea vomiting, diarrhoea, ulceration, headache, drowsiness and hematopoietic toxicity.
Meclofenamate sodium: it is an anthranilic acid derivative. Chemically it is sodium (2,6-dichloro-3-methyl phenylamino) benzoate. It is an anti-inflammatory drug for oral administration. Freely soluble in water. Meclofenamate capsule contains 50 mg or 100 mg.
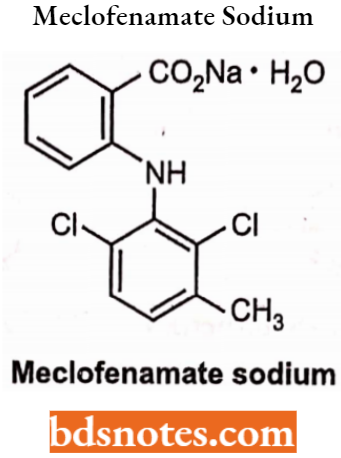
Meclofenamate sodium Uses: Meclofenamate is given for the relief of mild to moderate pain in acute and chronic rheumatoid, arthritis, osteo-artheritis. In the primary treatment of dysmenorrhoea and for the treatment of idiopathic heavy menstrual blood loss.
Contraindication: Meclofenamate sodium should not be used in those patients who have previously exhibited hypersensitivity to these drugs. Greater potential for cross-sensitivity to aspirin or other non-steroidal anti-inflammatory drugs. It should not be given to those patients who show drug-induced symptoms of bronchospasm, allergic rhinitis or urticarial.
Meclofenamate sodium Side Effects: Abdominal pain, oedema, urticaria, pruritis, headache, nausea, dizziness, tinnitus, blurred vision.
Diclofenac Sodium: Chemically, it is a sodium [0-(2,6-dichloroanilino) phenyl] acetate. It acts (COX-2) as an inhibitor with more potency than COX-1 inhibitors and has lower GI irritations than aspirin.
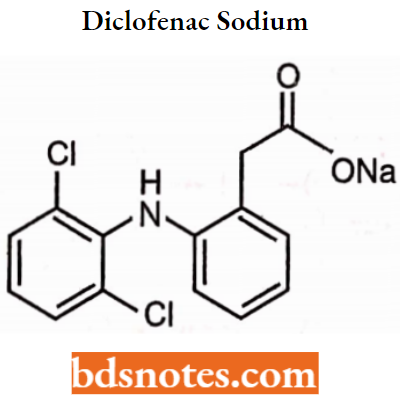
Diclofenac Sodium Uses: As an anti-inflammatory, antipyretic, and analgesic agent.
Diclofenac Sodium Side Effects: Nausea, vomiting, ulceration, headache.
Oxicams (Enol Acid): They are the 4-hydroxybenzothiazine. The acidity of the compound is because of the 4-OH which was stabilized by intramolecular hydrogen bonding to the amide N-H group, they were ionized at physiologic pH, which is required for COX inhibitory activity.
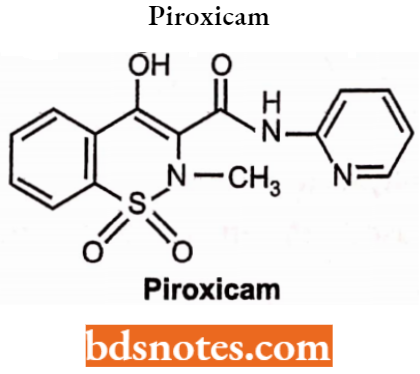
Oxicams (Enol Acid) Uses: In the treatment of rheumatoid, arthritis and osteoarthritis.
Oxicams (Enol Acid) Side Effects: Nausea, vomiting, ulceration, headache.
Phenylpyrazolones: Chemically, it is l-aryl-3,5 pyrazolidinedione structure. The presence of a proton which is situated to two electron-withdrawing carbonyl groups renders these compounds acidic. The pKa for phenylbutazone is 4.5. Oxyphenbutazone is a hydroxylated metabolite of phenylbutazone.
Phenylbutazone: They are anti-inflammatory drugs, and also possess some analgesic and antipyretic activities. They also have mild uricosuric
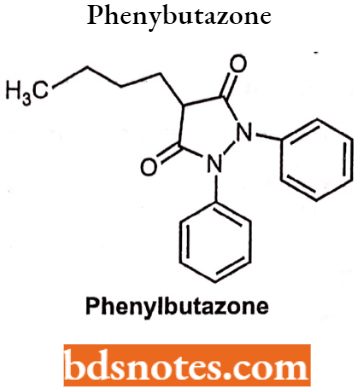
Phenylbutazone Uses: Phenylbutazone is used in the treatment of rheumatoid arthritis and osteoarthritis.
Phenylbutazone Side Effects: Include G1 irritation, Na+ and H20 retention and blood dyscariasis. Therapy should be limited to 7-10 days due to the development of bone marrow depression.
Antipyrine: Chemically it is 2, 3-dimethyl-l-phenyl-3-pyrazolin-5-one. It was one of the first synthetic compounds to be used in medicine. It is a colourless, crystalline white powder. It is odourless and has a slightly bitter taste. It is freely soluble in water, alcohol, and chloroform.
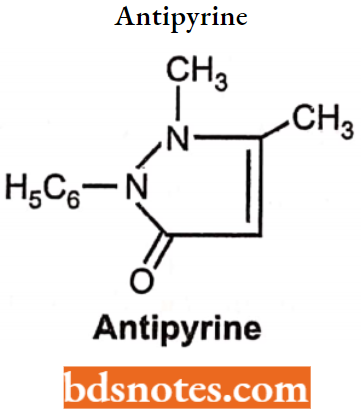
Antipyrine Uses: Analgesic, anti-inflammatory and antipyretic activities.
Antipyrine Side Effects: Include GI irritation, swelling, and vomiting.
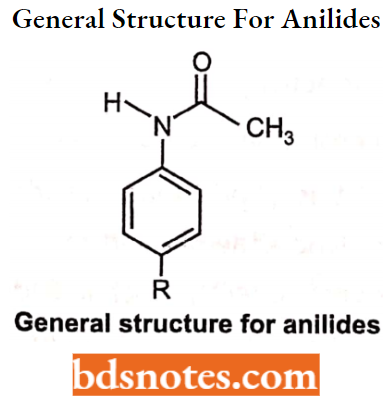
Anilides: The anilides are simple acetamides of aniline. It may or may not have 4-hydroxy or 4-alkoxy group. They do not possess carboxylic acid functionality and therefore they are neutral drugs and possess little inhibitory activity against cyclooxygenase.
Anilides SAR:
- Esterification of phenolic groups using methyl or propyl groups produces more side effects as compared to ethyl derivatives.
- The presence of substituents reduces the basicity of nitrogen and thus decreases the pharmacological activity of the drug.
Anilides Mechanism of Action: They are different from other NSAIDs in their mechanism of action. They act as scavengers of hydro-peroxide radicals (hydro-peroxide radicals are generated after injury).
- These radicals have a stimulating effect on cyclooxygenase. The concentration of hydro-peroxides at the sight of injury and inflammation is high.
- Thus they do not have anti-inflammatory action. They are only capable of suppressing cyclooxygenase activity in areas which are not inflamed.
- The lack of an acidic group and COX inhibitory activity made anilides have less gastric irritation, ulceration, and respiratory effects and little effect on platelets (no increase in clotting).
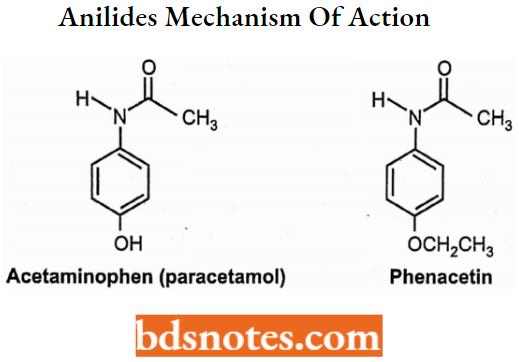
Acetaminophen: (Paracetamol): Chemically, it is N-acetyl-p-aminophen. It is a metabolite of phenacetin. It inhibits the cyclooxygenase enzyme centrally but has a lesser effect peripherally.
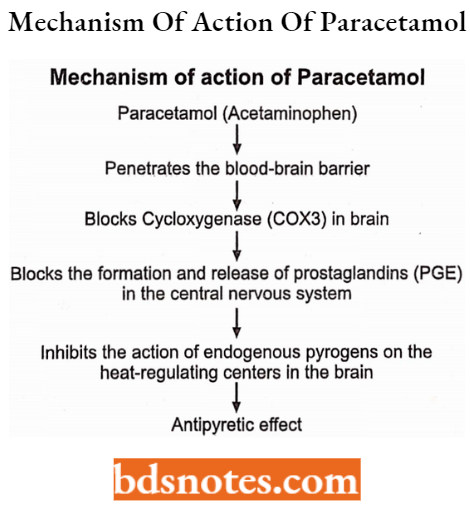
Acetaminophen Uses: Analgesic and antipyretic activity
Acetaminophen Side Effects: Liver toxicity in chronic alcoholics.
Phenacetin: Chemically, it is p-ethoxyacetanilide. It was introduced in 1885 to medicine. It causes haemolytic anaemia and meth-haemoglobin anaemia. Its use has declined because of its adverse effects, which include increased risk of certain cancers and kidney damage. It is metabolized as paracetamol (acetaminophen).
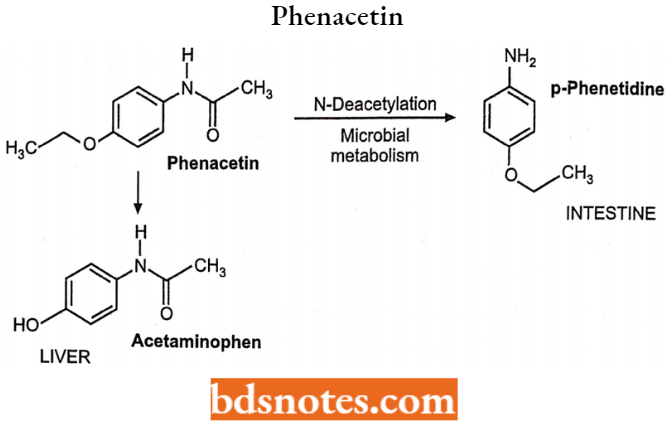
Phenacetin Uses: Analgesic and antipyretic activity.
Phenacetin Effects: Include GI irritation.
Leave a Reply